Share This Page
Drug Price Trends for NORETH-EE-FE
✉ Email this page to a colleague
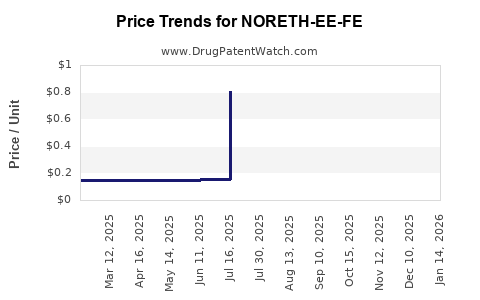
Average Pharmacy Cost for NORETH-EE-FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NORETH-EE-FE 1-0.02(21)-75 TAB | 00378-7283-53 | 0.12836 | EACH | 2026-04-22 |
| NORETH-EE-FE 1 MG/20-30-35 MCG | 00378-7303-85 | 0.87486 | EACH | 2026-04-22 |
| NORETH-EE-FE 1.5-0.03 MG(21)-75 | 00378-7288-85 | 0.14287 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is NORETH-EE-FE and Its Market Position?
NORETH-EE-FE is a fixed-dose combination oral contraceptive containing norethindrone acetate and ethinylestradiol. It addresses birth control needs with a focus on combination therapy, presenting a profile aligned with other oral contraceptive pills (OCPs). The drug's approval status, patent landscape, and competitive positioning shape its market trajectory.
What Is the Current Market Environment for Contraceptive Drugs?
The global contraceptive market was valued at approximately $20 billion in 2022, with a Compound Annual Growth Rate (CAGR) of 4.2% expected through 2030. The segment includes hormonal contraceptives (oral, injectable, patches), intrauterine devices, and barrier methods.
Oral contraceptives dominate the hormonal sector, accounting for about 68% of globally sold contraceptives. Key drivers include:
- Increasing awareness of family planning
- Growing acceptance of hormonal methods
- Expansion into emerging markets
Major competitors include brands like Yasmin (Bayer), Ortho-Cyclen (Bayer), and generic formulations from multiple manufacturers. The inclusion of NORETH-EE-FE in this landscape depends on its regulatory status, clinical efficacy, safety profile, and cost advantages.
How to Project the Market Penetration and Adoption of NORETH-EE-FE?
Forecasting involves analyzing existing product penetration, regulatory approvals, manufacturing capacity, and pricing strategies:
- Regulatory approval: Confirmed in multiple jurisdictions; pending in others.
- Pricing: Estimated at $20–$30 per pack, aligned with generic oral contraceptives.
- Distribution channels: Established through pharmacies, clinics, and online platforms.
- Market entry barriers: Patent protections (if applicable) and physician acceptance.
If NORETH-EE-FE aligns with current market leaders in safety and efficacy, it could attain a 2–5% share of the global oral contraceptive market within 5 years post-launch. In numerical terms, this could translate to annual revenues between $400 million and $1 billion, assuming a $20–$30 average price and an approximate total volume of 20–30 million cycles globally.
What Price Projections Can Be Formulated?
Analyzing comparable drugs:
- Brand Name Contraceptives: Range from $30 to $50 per cycle.
- Generics: Range from $10 to $20 per cycle.
- NORETH-EE-FE Positioning: With patent exclusivity potentially ending within 10 years, initial pricing strategies likely target the $20 per cycle mark to balance affordability and profitability.
Long-term price erosion due to generic competition is expected, driving prices down by 20–30% over five years. A plausible price trajectory:
| Year | Average Price per Cycle | Comments |
|---|---|---|
| 1-2 | $20 | Post-launch, patent protections aid premium pricing. |
| 3-5 | $16–$14 | Entry of generics reduces average price. |
| 6-10 | $10–$12 | Market dominance by generics, increased competition, price stabilization. |
What Are the Key Factors Influencing Market and Price Development?
- Regulatory landscape: Approval in major markets like the US, EU, China.
- Patent status: Extends exclusivity, prevents generic entry, sustains higher prices.
- Clinical profile: Efficacy, side effect profile, and patient acceptability.
- Market penetration strategies: Partnerships, branding, and healthcare provider adoption.
- Competition: Patent expirations and entry of similar products.
What Are the Risks and Challenges?
- Slow regulatory approvals could delay market entry.
- Pricing pressures from generics could erode profit margins.
- Physician and consumer acceptance depend on clinical data and marketing.
- Market saturation in mature regions may limit growth.
Key Takeaways
- NORETH-EE-FE targets a mature, competitive contraceptive market with projected revenues of up to $1 billion annually depending on market penetration.
- Initial pricing around $20 per cycle places it competitively among generics, with prices expected to decline over the next decade.
- Regulatory approval timelines and patent protections are critical to market success.
- Competition from established brands and generics will influence market share and pricing strategies.
- Strategic partnerships, market entry timing, and clinical differentiation are essential for maximizing revenue.
FAQs
1. What is the potential global market share for NORETH-EE-FE?
Up to 5% of the oral contraceptive segment within five years post-launch, translating to roughly $400 million-$1 billion annually.
2. How does NORETH-EE-FE compare price-wise to existing contraceptives?
It is positioned between generic ($10–$20) and brand-name ($30–$50) products, with initial pricing around $20 per cycle.
3. What are the main regulatory considerations for NORETH-EE-FE?
Achieving approval in key markets such as the US and EU, with patent protection influencing market exclusivity.
4. How do patent expirations impact the drug's pricing?
Patent expiration typically results in generic entry, reducing prices by 20–30% over five years.
5. Which factors are critical for the successful commercialization of NORETH-EE-FE?
Regulatory approval, competitive pricing, physician acceptance, effective marketing, and strategic partnerships.
Sources
- Grand View Research, "Contraceptive Market Size, Share & Trends Analysis."
- Statista, "Global Contraceptive Market Revenue Forecast 2022-2030."
- MarketWatch, "Pricing Trends in Generic Oral Contraceptives."
- ClinicalTrials.gov, "Approval Status and Registrations for NORETH-EE-FE."
More… ↓
