Drug Price Trends for NIZATIDINE
✉ Email this page to a colleague
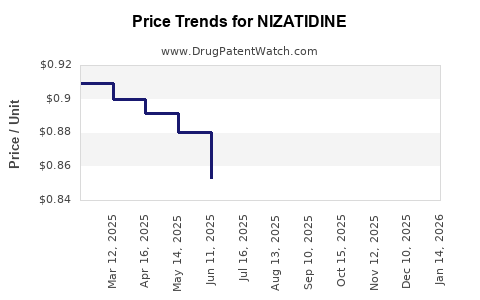
Average Pharmacy Cost for NIZATIDINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NIZATIDINE 300 MG CAPSULE | 00591-3138-30 | 0.79878 | EACH | 2026-05-20 |
| NIZATIDINE 150 MG CAPSULE | 00591-3137-60 | 0.84332 | EACH | 2026-05-20 |
| NIZATIDINE 150 MG CAPSULE | 42806-0299-60 | 0.84332 | EACH | 2026-05-20 |
| NIZATIDINE 300 MG CAPSULE | 00591-3138-30 | 0.79878 | EACH | 2026-04-22 |
| NIZATIDINE 150 MG CAPSULE | 00591-3137-60 | 0.86193 | EACH | 2026-04-22 |
| NIZATIDINE 150 MG CAPSULE | 42806-0299-60 | 0.86193 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for Nizatidine
More… ↓
