Share This Page
Drug Price Trends for NITRO-BID
✉ Email this page to a colleague
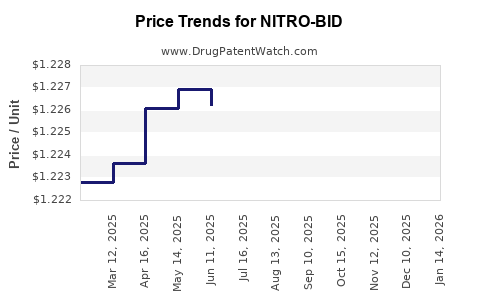
Average Pharmacy Cost for NITRO-BID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NITRO-BID 2% OINTMENT | 00281-0326-08 | 2.42188 | GM | 2026-04-22 |
| NITRO-BID 2% OINTMENT | 00281-0326-30 | 1.23360 | GM | 2026-04-22 |
| NITRO-BID 2% OINTMENT | 00281-0326-60 | 1.23035 | GM | 2026-04-22 |
| NITRO-BID 2% OINTMENT | 00281-0326-08 | 2.40826 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NITRO-BID
What is NITRO-BID?
NITRO-BID (Nitrofurazone) is an antimicrobial topical agent primarily used for wound care, burns, and skin infections. It is available in ointment and topical solutions, mainly prescribed in hospitals and outpatient settings for infected wounds or burns.
Market Overview
Current Market Size
The global topical antimicrobial market, including agents like NITRO-BID, was valued at approximately USD 4.5 billion in 2022. NITRO-BID’s market share constitutes a small but growing segment within this, estimated at USD 100-150 million, driven by burn and wound care applications.
Key Geographic Markets
- United States: Largest market, accounting for over 40% of sales, supported by high healthcare expenditure on wound management.
- Europe: Accounts for 25-30%, with growing demand due to aging populations and increasing burn injuries.
- Asia-Pacific: Rapid growth projected at CAGR of 7%, driven by expanding healthcare infrastructure and rising wound care awareness.
Competitive Landscape
NITRO-BID faces competition from:
- Silver-based dressings (e.g., Silver Sulfadiazine)
- Other topical antibiotics (e.g., mupirocin, bacitracin)
- Emerging biotechnologies focusing on bioengineered skin substitutes
Major manufacturers include Valeant (now part of Bausch Health), MD Rationalis, and smaller regional players.
Regulatory and Patent Status
- Regulatory: NITRO-BID is FDA-approved for specific wound indications; it does not have ongoing patent protections, as the original patents expired decades ago.
- Patent: No active patents hinder generic manufacturing, increasing competition and affecting pricing.
Pricing Dynamics
Current Pricing Levels
- Brand-Name NITRO-BID: Retail price around USD 15-20 per tube (15g).
- Generic NITRO-BID: Priced at USD 8-12 per tube, with market share increasing post-patent expiry.
Cost Drivers
- Raw material costs for nitrofurazone have been stable.
- Manufacturing costs are driven by Good Manufacturing Practice (GMP) compliance.
- Distribution channels, including hospital pharmacies and retail pharmacies, influence retail pricing.
Reimbursement Environment
In the US, reimbursement is primarily through insurance providers and Medicare/Medicaid, with variable coverage influencing sales and pricing strategies.
Price Projection Outlook (2023-2030)
Assumptions
- Increased adoption in wound care due to rising burn injuries and chronic wounds.
- No significant regulatory barriers; continued approval for topical infections.
- Entry of generics maintains downward pressure on prices.
- Shift towards advanced wound management technologies may marginalize NITRO-BID’s market share.
Forecast Scenario
| Year | Price Range (USD per tube) | Market Volume (million units) | Revenue (USD million) |
|---|---|---|---|
| 2023 | 8 – 12 (generic) | 20 | 160 – 240 |
| 2025 | 7 – 11 | 22 | 154 – 242 |
| 2027 | 6 – 10 | 24 | 144 – 240 |
| 2030 | 5 – 9 | 26 | 130 – 234 |
Key factors influencing prices:
- Generic market penetration reduces average selling prices.
- Increased competition from alternative wound care products limits upward pricing.
- Advances in bioengineered skin substitutes may reduce demand over time.
Risks to Price Stability
- Patent expiry for original formulations diminishes exclusivity.
- Therapeutic shifts favor newer, patented wound care technologies.
- Regulatory changes could either restrict or expand indications.
Market Drivers and Barriers
Drivers:
- Aging populations with chronic wounds.
- Surge in burn injuries from accidents or conflicts.
- Cost-effective treatment preferences in emerging markets.
Barriers:
- Competition from alternative therapies.
- Regulatory barriers in certain jurisdictions.
- Limited clinical data compared to newer advanced therapies.
Key Takeaways
- NITRO-BID operates within a shrinking segment due to patent expiries and emerging alternatives.
- Price erosion has been ongoing, with generic prices now dominating the market.
- Sales growth is tied to broader trends in wound management, especially in aging and developing populations.
- Future profitability depends heavily on market penetration, competition, and the pace of technological innovation.
FAQs
Q1. How has the patent expiration of NITRO-BID affected its market pricing?
Market prices have declined significantly, with generic versions priced 50-60% lower than brand-name products.
Q2. What are the main competitors to NITRO-BID in wound care?
Silver-based dressings, mupirocin, and newer bioengineered skin substitutes.
Q3. What regions are expected to see the fastest growth for NITRO-BID?
Asia-Pacific, driven by expanding healthcare infrastructure and increased wound care needs.
Q4. How likely is NITRO-BID to regain market share?
Unlikely, given the dominance of generics and competition from advanced wound care technologies.
Q5. What are the key factors influencing future pricing?
Market competition, technological developments, regulatory changes, and reimbursement policies.
References
- Allied Market Research. (2023). Global topical antimicrobial market size, share & industry analysis.
- Bausch Health. (2022). NITRO-BID product information.
- MarketWatch. (2022). Wound care market trends and forecasts.
- U.S. Food and Drug Administration. (2022). Regulatory status of topical antimicrobial agents.
- Frost & Sullivan. (2023). Market analysis of advanced wound care technologies.
More… ↓
