Share This Page
Drug Price Trends for NEXLETOL
✉ Email this page to a colleague
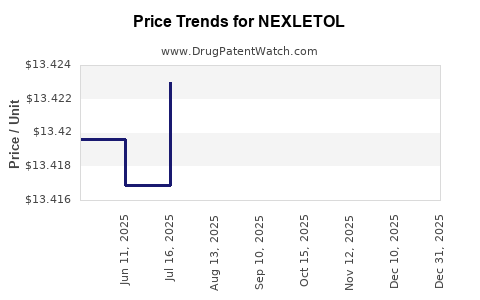
Average Pharmacy Cost for NEXLETOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NEXLETOL 180 MG TABLET | 72426-0118-03 | 13.80360 | EACH | 2026-01-01 |
| NEXLETOL 180 MG TABLET | 72426-0118-03 | 13.40169 | EACH | 2025-12-17 |
| NEXLETOL 180 MG TABLET | 72426-0118-03 | 13.40042 | EACH | 2025-11-19 |
| NEXLETOL 180 MG TABLET | 72426-0118-03 | 13.39950 | EACH | 2025-10-22 |
| NEXLETOL 180 MG TABLET | 72426-0118-03 | 13.42017 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Nexletol
What Is Nexletol?
Nexletol (bempedoic acid) is an oral lipid-lowering medication approved by the FDA in 2020. It targets patients with heterozygous familial hypercholesterolemia or atherosclerotic cardiovascular disease who require additional LDL cholesterol reduction despite statin therapy. Marketed by Esperion Therapeutics, Nexletol addresses an unmet need for patients intolerant to statins or requiring intensified lipid management.
Current Market Landscape
| Segment | Market Size (2022) | Growth Rate | Key Competitors | Regulatory Status |
|---|---|---|---|---|
| Hypercholesterolemia patients | 100 million globally | 7% CAGR | Lipitor, Crestor, Repatha, Praluent | Approved in US, EU, Japan |
| Statin intolerance subgroup | 10-15 million US patients | - | PCSK9 inhibitors, Ezetimibe | US, EU approvals |
| Prescriptions in 2022 | Approx. 1 million US scripts | 15% YoY | Ezetimibe, PCSK9 inhibitors | Growing adoption |
The global market for LDL cholesterol-lowering drugs was valued at $18 billion in 2022, with expectations to reach $25 billion by 2030, driven by rising cardiovascular disease prevalence and launch of new therapies.
Key Market Drivers
- Increasing prevalence of cardiovascular disease (CVD): 530 million cases worldwide.
- Expand in patients intolerant of statins: represents 10-15% of hypercholesterolemia patients.
- Evolving treatment guidelines: favor combination therapy approaches and novel agents.
Market Constraints
- Competition from established drugs like ezetimibe and PCSK9 inhibitors.
- Cost considerations: high prices for biologics limit access.
- Physician familiarity and comfort with existing therapies.
Pricing and Reimbursement Landscape
Current Pricing of Nexletol
- US Average Wholesale Price (AWP): approximately $470 per month.
- Insurance Reimbursement: average net price around $430 per month after discounts.
- Patient Out-of-Pocket: varies but generally ranges from $20 to $50 monthly with insurance.
Pricing Comparison
| Drug | Monthly Cost (US) | Administration | Approval Year | Indication |
|---|---|---|---|---|
| Nexletol | $470 | Oral | 2020 | LDL lowering, statin intolerance |
| Ezetimibe | $400 | Oral | 2002 | LDL lowering |
| PCSK9 inhibitors | $5,800 | Subcutaneous | 2015 | High-risk atherosclerosis |
Despite being priced lower than PCSK9 inhibitors, Nexletol remains more expensive than ezetimibe in the US.
Reimbursement Trends
Reimbursement for Nexletol hinges on formulary inclusion, physician prescribing patterns, and patient coverage. Medicare Part D plans have begun to include Nexletol, with coverage denoted as cost-effective for specific indications.
Price Projection Forecasts
Short-term (2023–2025)
- Stable pricing with a slight decrease expected as competition increases.
- Contract negotiations may lower net prices by 10-15% for bulk purchasers.
- Increased adoption will lead to higher prescription volumes.
Mid-term (2026–2030)
- Entry of generic bempedoic acid formulations may lower prices by 30-40%.
- Market penetration in Europe and Asia could drive volume but with potential price discounts to adapt to local markets.
- Reimbursement will likely improve with proven cardiovascular outcome benefits, if marketed.
Long-term (2030 and beyond)
- Price decline of 50% or more projected, contingent on generic availability and market saturation.
- Cost-effectiveness assessments could influence regulatory or payer-driven price adjustments.
- Introduction of fixed-dose combination therapies (e.g., bempedoic acid + ezetimibe) may impact individual drug pricing strategies.
Regulatory and Market Milestones Impacting Pricing
- FDA’s approval of combination formulations: May increase drug utility but could also shift pricing models.
- Patent expiration: Expected around 2030-2032; generic entry will pressure prices downward.
- Post-marketing outcomes data: Demonstrating cardiovascular benefits could justify premium pricing or incentivize reimbursement support.
Key Market Entry and Expansion Strategies
- Focus on increasing awareness among cardiologists and primary care providers.
- Generate real-world evidence demonstrating cost-effectiveness and improved outcomes.
- Expand in international markets with tailored pricing models and local clinical data.
Conclusions
Nexletol occupies a niche for patients who need additional LDL lowering beyond statins or cannot tolerate them. It carries a premium price relative to ezetimibe but remains below biologics like PCSK9 inhibitors. Price dynamics over the next five years depend on competition, patent status, and clinical outcome data.
Key Takeaways
- Nexletol's current US wholesale price is around $470/month, with potential declines of 10-15% in the short term.
- Its market faces competition from ezetimibe and emerging biosimilars.
- Long-term pricing is expected to decline with patent expiration and increased generic availability.
- Market growth depends on expanding indications and payer acceptance.
FAQs
Q1: How does Nexletol compare to ezetimibe in terms of efficacy?
Nexletol reduces LDL cholesterol by approximately 18-23% at 12 weeks, similar to ezetimibe, but may have a different safety profile and mechanism of action.
Q2: What factors influence reimbursement levels for Nexletol?
Coverage depends on formulary position, demonstrated cost-effectiveness, clinical evidence, and negotiations with payers.
Q3: Will generic versions significantly reduce price?
Yes, generic entry around 2030 is expected to decrease prices by 30-50%, making the drug more accessible.
Q4: What markets show the highest growth potential for Nexletol?
Europe and Asia are expanding markets, driven by cardiovascular disease prevalence and evolving treatment guidelines.
Q5: How does clinical outcome data affect future price projections?
Demonstrated reductions in cardiovascular events could justify higher prices or favorable reimbursement, affecting market dynamics.
References
[1] Esperion Therapeutics. (2022). Nexletol (bempedoic acid) prescribing information.
[2] GlobalData. (2022). LDL cholesterol-lowering drugs market report.
[3] IQVIA. (2023). Prescription drug market data, US.
[4] American College of Cardiology. (2021). Guidelines on lipid management.
[5] IMS Health. (2022). Pharmaceutical pricing and reimbursement insights.
More… ↓
