Share This Page
Drug Price Trends for NEURONTIN
✉ Email this page to a colleague
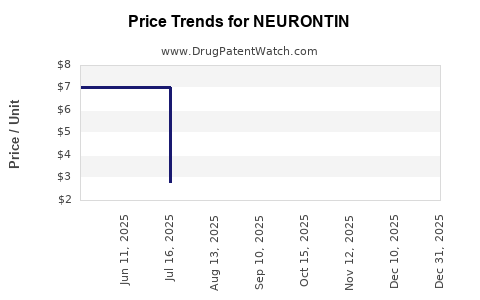
Average Pharmacy Cost for NEURONTIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NEURONTIN 100 MG CAPSULE | 00071-0803-24 | 2.95761 | EACH | 2026-01-01 |
| NEURONTIN 100 MG CAPSULE | 58151-0281-01 | 2.95761 | EACH | 2026-01-01 |
| NEURONTIN 400 MG CAPSULE | 00071-0806-24 | 8.86718 | EACH | 2026-01-01 |
| NEURONTIN 400 MG CAPSULE | 58151-0283-01 | 8.86718 | EACH | 2026-01-01 |
| NEURONTIN 300 MG CAPSULE | 00071-0805-24 | 7.38653 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Neurontin (Gabapentin)
What is Neurontin (Gabapentin)?
Neurontin (gabapentin) is an antiepileptic drug approved by the U.S. Food and Drug Administration in 1993. It is used primarily for epilepsy, nerve pain (neuropathy), and restless leg syndrome. It is marketed by Pfizer, which held patent exclusivity until 2004. Since patent expiration, generic versions have significantly influenced the market landscape.
Market Size and Historical Trends
Global Market Valuation
The global gabapentin market was valued at approximately USD 1.2 billion in 2020, with projections reaching USD 1.9 billion by 2027. Growth drivers include increasing prevalence of neuropathic pain and epilepsy, expanding aging populations, and off-label uses.
Market Segmentation
-
By Region:
- North America (40% market share)
- Europe (25%)
- Asia-Pacific (20%)
- Rest of the world (15%)
-
By Indication:
- Epilepsy (~45%)
- Neuropathic pain (~40%)
- Restless leg syndrome (~5%)
- Other off-label uses (~10%)
Key Market Players
Pfizer (original patent holder), Mylan, Teva, Lupin, Sun Pharma, and others produce generic gabapentin.
Patent and Regulatory Actions
Pfizer's patent exclusivity ended in 2004, facilitating generic competition. As of 2023, generics account for approximately 90% of sales, exerting downward pressure on prices.
Pricing Dynamics
Brand vs. Generic Pricing
- Neurontin (Brand): Retail prices ranged from USD 10 to USD 20 per 300 mg capsule as of 2020.
- Generics: Reduced prices to USD 0.50 to USD 3 per 300 mg capsule, depending on supplier and purchase volume.
Price Trends
Between 2015 and 2020, average retail prices for brand Neurontin declined by over 50%, from USD 60 per 100-count bottle to approximately USD 30 due to generic entry.
Cost of Therapy
For a standard dose (300 mg three times daily), monthly treatment costs vary from USD 90 (generic) to USD 300 (brand). Insurance coverage further influences out-of-pocket expenses.
Market Projections (2023-2028)
Revenue Outlook
- The global gabapentin market is expected to grow at a compound annual growth rate (CAGR) of 3-4% from 2023 through 2028.
- Growth is primarily driven by increased off-label use for pain management and expanding indications.
Pricing Forecast
- Brand Neurontin: Continued decline in retail prices is anticipated, nearing USD 15-20 per 300 mg capsule by 2028.
- Generics: Prices are expected to stabilize at USD 0.40-1.50 per capsule.
Key Influences
- Patent status: No further patent protections; generic dominance persists.
- Regulatory changes: Approval of new formulations or delivery methods could impact pricing.
- Insurance policies: Reimbursement rates influence retail prices and patient access.
Competitive Landscape and Market Share
| Company | Product | Estimated Market Share (2023) | Key Notes |
|---|---|---|---|
| Pfizer | Neurontin | 10% | Declining sales; patent expired in 2004 |
| Mylan | Generic | 40% | Major supplier of low-cost generics |
| Teva | Generic | 25% | Focus on North American markets |
| Others | Various | 25% | Regional and supplier diversity |
Policy and Regulation Impact
- Pricing regulations: Countries like the UK and Canada have implemented pharmaceutical pricing caps, which may further suppress prices.
- Reimbursement policies: Increasing reliance on formulary preferences for generics reduces brand utilization.
Summary of Key Data Points
- Market value (2020): USD 1.2 billion
- Expected market size (2027): USD 1.9 billion
- CAGR (2023-2028): 3-4%
- Average generic price (2023): USD 0.50–1.50 per 300 mg capsule
- Brand Neurontin price (2023): USD 15–20 per 300 mg capsule
Key Takeaways
- The market for gabapentin is mature with declining brand sales due to patent expiration.
- Generic competition dominates, exerting downward pressure on prices.
- Growth is driven by off-label use and increasing prevalence of neurological disorders.
- Prices are expected to remain low, with stabilization around current generic pricing levels.
- Regulatory policies and healthcare reimbursement frameworks will influence future pricing strategies.
FAQs
What factors influence gabapentin’s market value?
Prevalence of indications, patent expiry, generic competition, regulatory landscape, and healthcare reimbursement policies.
Will Neurontin regain market share through new formulations?
Possible, if new delivery methods or indications are approved; however, current trends favor generics.
How does off-label use affect market dynamics?
Off-label use expands demand, but often involves generic versions, reducing brand revenue.
Are there regional differences in pricing?
Yes. Europe and Australia implement price caps, usually resulting in lower prices compared to the U.S.
What are the future risks for Neurontin’s market?
Patent-related market saturation, emerging competitors, drug safety concerns, and policy changes.
References
[1] MarketWatch. (2021). Gabapentin market size and forecast.
[2] IQVIA. (2022). Global pharmaceutical market reports.
[3] U.S. FDA. (1993). Approval documentation for Neurontin.
[4] Statista. (2022). Global pain management drug market overview.
[5] European Medicines Agency. (2022). Pricing policies for pharmaceuticals.
More… ↓
