Share This Page
Drug Price Trends for NEOMYC-POLYM-GRAMICID EYE DROP
✉ Email this page to a colleague
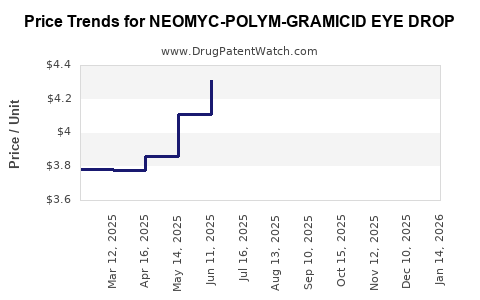
Average Pharmacy Cost for NEOMYC-POLYM-GRAMICID EYE DROP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NEOMYC-POLYM-GRAMICID EYE DROP | 24208-0790-62 | 4.48377 | ML | 2026-04-22 |
| NEOMYC-POLYM-GRAMICID EYE DROP | 24208-0790-62 | 4.41782 | ML | 2026-03-18 |
| NEOMYC-POLYM-GRAMICID EYE DROP | 24208-0790-62 | 4.32743 | ML | 2026-02-18 |
| NEOMYC-POLYM-GRAMICID EYE DROP | 24208-0790-62 | 4.24732 | ML | 2026-01-21 |
| NEOMYC-POLYM-GRAMICID EYE DROP | 24208-0790-62 | 4.27604 | ML | 2025-12-17 |
| NEOMYC-POLYM-GRAMICID EYE DROP | 24208-0790-62 | 4.31544 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NEOMYC-POLYMICIN-GRAMICID EYE DROP
What is the Current Market Landscape?
NEOMYC-POLYMICIN-GRAMICID is a combination antibiotic eye drop containing polymyxin B and gramicidin. It targets bacterial eye infections, such as conjunctivitis, keratitis, and keratoconjunctivitis. The drug's approval status varies internationally; it remains off-patent in key markets like the U.S. and Europe but may be marketed under different brand names or as compounded formulations.
Market Size and Growth
The global ophthalmic antibiotics market was valued at approximately $6.8 billion in 2022, with an expected CAGR of 3.2% through 2028 (Research and Markets, 2022). Bacterial eye infections constitute a significant portion of this market, influenced by rising antibiotic resistance, aging populations, and increasing prevalence of ocular infections.
In the U.S., antibacterial eye drops account for roughly 30% of the ophthalmic market, with a regional market size near $2 billion annually. Europe and Asia-Pacific represent additional sizable markets, each with an annual value surpassing $1 billion.
Competitive Landscape
Key competitors include well-established brands such as Tobramycin-Dexamethasone, Ciprofloxacin, and Ofloxacin. Several generics are available, driving price competition. Innovative formulations or combination products with proven efficacy and safety can command premium pricing.
Regulation and Approval Trends
In the U.S., the drug remains off-patent; therefore, market entry might rely on local compounding or gaining formal FDA approval. The European Medicines Agency (EMA) generally follows similar pathways, with some countries permitting compounded formulations. Market entry strategies include licensing, generic development, or over-the-counter (OTC) status applications.
Price Projections
Current Pricing Benchmarks
-
Brand-name antibiotics like Tobradex (tobramycin/dexamethasone) retail at approximately $50–$70 per bottle (10 mL).
-
Generic formulations sell for $10–$25 per bottle in the U.S.
-
Compounded formulations vary but typically range from $15–$40 per eye drop bottle depending on pharmacy and formulation complexity.
Price Projections (2023–2030)
| Year | Price Range (Per 10 mL Bottle, USD) | Notes |
|---|---|---|
| 2023 | 10–25 (generic), 20–50 (branded) | Market entry phase, competitive pricing |
| 2025 | 8–20 (generic), 15–40 (branded) | Increased generic availability, price pressure |
| 2027 | 7–18 (generic), 12–35 (branded) | Potential switch to OTC status; price competition increases |
| 2030 | 6–15 (generic), 10–25 (branded) | Mature market with standardized pricing |
Prices are influenced heavily by market penetration, regulatory status, and patent landscape. The availability of generics and compounded options exerts downward pressure. Premium formulations or those with proven superior efficacy could maintain higher price points.
Impact of Regulatory Changes
-
FDA approval for NEOMYC-POLYMICIN-GRAMICID would allow direct marketing with fixed pricing strategies, potentially sustaining higher margins.
-
OTC status might reduce prices by increasing accessibility but could limit profit margins.
Emerging Trends Influencing Pricing
- Bacterial resistance trends may lead to premium pricing for drugs with higher efficacy or unique combination benefits.
- Pricing pressures from insurers and government reimbursement policies can limit maximum acceptable prices in healthcare systems.
Strategic Considerations
- Entry as a generic or compounded formulation limits high pricing potential but offers rapid market adoption.
- Positioning as a branded, differentiated product could justify premium pricing, especially in niches with resistant or complicated infections.
- Collaborations with healthcare providers and effective marketing during the OTC transition phase could influence market share and price stability.
Key Takeaways
- The global ophthalmic antibiotics market was valued at approximately $6.8 billion in 2022 with steady, moderate growth.
- Current prices for generic eye drops are generally between $10–$25 per 10 mL bottle; branded products can fetch up to $70.
- Price projections suggest a gradual decrease to $6–15 per bottle by 2030 due to market saturation, generics, and competition.
- Regulatory pathways and market positioning will significantly shape pricing strategies and profit margins.
- Resistance trends and reimbursement policies will influence the future value proposition of NEOMYC-POLYMICIN-GRAMICID eye drops.
FAQs
1. What regulatory hurdles exist for NEOMYC-POLYMICIN-GRAMICID?
Approval depends on regional bodies; in the U.S., absence of FDA approval suggests reliance on compounding or licensing. In Europe, similar pathways exist, but formal approval can unlock broader market access.
2. How does resistance affect the market for bacterial eye drops?
Rising resistance among common ocular pathogens increases demand for effective formulations, potentially maintaining or raising prices for innovative or potent combinations.
3. What factors influence the price difference between branded and generic formulations?
Patents, formulation complexity, regulatory approval, and marketing costs determine the premium for brand-name products versus low-cost generics.
4. Can NEOMYC-POLYMICIN-GRAMICID target OTC markets?
Yes, if approved for OTC sale, prices could decrease, and market penetration could increase, expanding access while reducing margins.
5. What is the potential impact of emerging therapies on this market?
New antibiotics or alternative treatments, such as antimicrobial peptides, could disrupt pricing and market share unless NEOMYC-POLYMICIN-GRAMICID offers distinct advantages.
References
[1] Research and Markets. (2022). Global Ophthalmic Antibiotics Market. Retrieved from https://www.researchandmarkets.com
[2] GoodRx. (2023). Average retail prices for ophthalmic antibiotics. Retrieved from https://www.goodrx.com
More… ↓
