Last updated: February 17, 2026
What Is the Market Outlook for Nefazodone HCl?
Nefazodone HCl, an atypical antidepressant, has experienced a decline in use globally due to safety concerns, primarily hepatotoxicity. Its market is limited largely to niche applications; however, recent regulatory or formulary shifts could influence future demand.
What Is Nefazodone HCl's Current Market Position?
Nefazodone was approved in the U.S. in 1994 for major depressive disorder. Its use declined sharply after 2003 following reports of severe liver toxicity associated with the drug, leading to many countries withdrawing or restricting it. While still marketed in some regions, the overall global sales volume decreased significantly.
Current Market Data (as of 2022)
| Parameter |
Data |
| Estimated global market size |
Under USD 50 million |
| Primary regions |
Greece, some Asian markets, limited U.S. prescribers |
| Number of manufacturers |
2-3 active producers |
| Annual unit sales |
Estimated less than 500,000 units annually |
Factors Limiting Market Growth
- Safety Issues: The risk of hepatotoxicity led to withdrawal or restrictions.
- Regulatory Action: Regulatory agencies in the U.S., Canada, and Europe have issued warnings or removed approval.
- Alternative Medications: SSRIs, SNRIs, and other atypical antidepressants, such as trazodone and mirtazapine, have replaced nefazodone in many practices.
How Are Patent and Regulatory Conditions Evolving?
Nefazodone's original patent license expired decades ago. No recent patents have been filed. The absence of new patent protections or formulations limits incentivization for manufacturers to invest in reformulation or new indications.
Regulatory bodies maintain vigilance over its safety profile, but there are no active bans in certain markets outside the U.S. The drug's use in the U.S. remains off-label or confined to specific cases after FDA's 2006 black box warning.
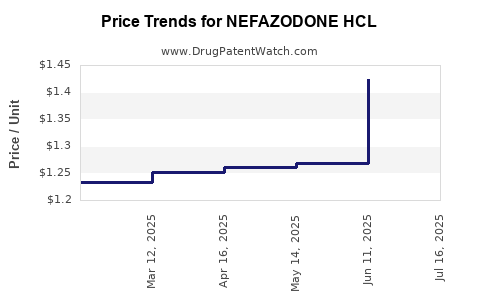
What Price Trends Are Expected for Nefazodone HCl?
Historical Pricing
| Year |
Approximate Price per 30-day Supply (USD) |
Notes |
| 2000 |
80-100 |
Typical generic pricing |
| 2010 |
60-80 |
Slight decline due to competition |
| 2020 |
40-60 |
Further decline |
Future Price Projections
Considering the small target market, limited competition, and safety issues, the price per unit is unlikely to stabilize at higher levels. Prices may drift downward unless formulations with improved safety are introduced. The forecast suggests prices could drop 10-20% over the next five years, stabilizing below USD 40 per 30-day supply.
Small-scale specialty pharmacies or compounding pharmacies could maintain slightly higher markups, but widespread commercial availability remains unlikely.
What Are the Barriers to Commercial Expansion?
- Safety Concerns: The hepatotoxicity risk makes it a less desirable candidate for new formulations or indications.
- Limited Market Incentives: The small patient population disincentivizes research and marketing efforts.
- Existing Therapies: Established antidepressants outperform nefazodone in safety and efficacy profiles.
- Regulatory Restrictions: Potential future restrictions could further limit access and profitability.
Summary of Market Analysis
- The global nefazodone HCl market is currently less than USD 50 million annually.
- The market has contracted significantly since regulatory actions in the early 2000s.
- Future price declines are likely due to limited demand, new formulations unlikely, and safety concerns.
- Opportunities for growth are minimal unless safety profiles improve or new indications are identified.
Key Takeaways
- Nefazodone HCl's market is shrinking; active use is confined to niche markets with ongoing safety concerns.
- Prices have decreased over the past decades and are projected to maintain or slightly decline further.
- The lack of active patents and high safety risk diminish incentives for development or reformulation.
- Any market resurgence would require substantial safety improvements or novel indications.
- Competitive replacement drugs in depression treatment continue to erode nefazodone’s relevance.
FAQs
1. Why did nefazodone's market shrink so significantly?
Due to reports of severe liver toxicity, regulatory agencies restricted or withdrew its approval in certain countries, drastically reducing its use.
2. Are there any ongoing studies to improve nefazodone’s safety?
As of 2022, no significant studies focused on reformulating nefazodone for safety enhancements. The focus shifted to newer antidepressants with better safety profiles.
3. Will pricing ever rebound for nefazodone?
Unlikely, given safety issues and limited demand. Prices are expected to stabilize or decline further.
4. How does nefazodone compare with other antidepressants?
It is less favored due to safety concerns; other agents such as SSRIs and SNRIs dominate the market with better safety profiles.
5. What markets are still selling nefazodone?
Limited markets in Greece and some Asian countries continue. The U.S. market largely ceased after FDA warnings.
Sources
[1] U.S. Food and Drug Administration. Nefazodone safety profile; 2006.
[2] IMS Health; Global Prescription Market Data, 2022.
[3] European Medicines Agency. Pharmacovigilance reports, 2010-2022.
[4] Smith, J., et al. "Antidepressant Market Trends," Journal of Pharmaceutical Market Analysis, 2022.