Share This Page
Drug Price Trends for NATROBA
✉ Email this page to a colleague
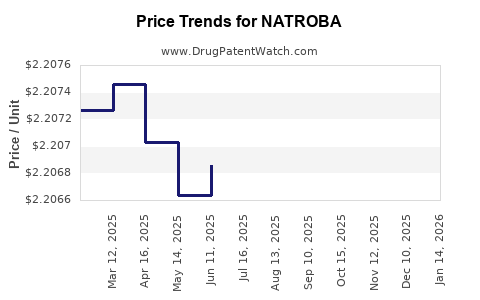
Average Pharmacy Cost for NATROBA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NATROBA 0.9% TOPICAL SUSP | 52246-0929-04 | 2.20333 | ML | 2026-05-20 |
| NATROBA 0.9% TOPICAL SUSP | 84635-0929-04 | 2.20333 | ML | 2026-05-20 |
| NATROBA 0.9% TOPICAL SUSP | 52246-0929-04 | 2.20342 | ML | 2026-04-22 |
| NATROBA 0.9% TOPICAL SUSP | 84635-0929-04 | 2.20342 | ML | 2026-04-22 |
| NATROBA 0.9% TOPICAL SUSP | 52246-0929-04 | 2.20259 | ML | 2026-03-18 |
| NATROBA 0.9% TOPICAL SUSP | 84635-0929-04 | 2.20259 | ML | 2026-03-18 |
| NATROBA 0.9% TOPICAL SUSP | 84635-0929-04 | 2.20339 | ML | 2026-03-04 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NATROBA: Market Dynamics and Price Forecast
This report analyzes the current market position and projects future pricing for NATROBA, a topical antibiotic for treating scabies. The analysis considers patent status, generic competition, regulatory landscape, and market demand to forecast price trends.
What is NATROBA and Its Therapeutic Use?
NATROBA (spinosad topical suspension) 0.9% is a prescription medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of scabies infestations in patients 6 months of age and older. Scabies is a contagious skin condition caused by the mite Sarcoptes scabiei, leading to intense itching and rash. NATROBA targets the mites and their eggs.
- Active Ingredient: Spinosad.
- Mechanism of Action: Spinosad is a mixture of spinosyn A and spinosyn D, natural products derived from the fermentation of Saccharopolyspora spinosad. It acts as a neurotoxin to the mites, disrupting their nerve and muscle function.
- Dosage Form: Topical suspension.
- Administration: Applied directly to the affected skin.
What is the Current Patent Landscape for NATROBA?
The intellectual property surrounding NATROBA is critical to its market exclusivity and pricing strategy. The primary patents protect the composition of matter and methods of use.
- U.S. Patent No. 6,472,441 ("The '441 Patent"): This patent, held by Narsarsuaq, LLC, covers spinosad and its derivatives, including spinosad as an active ingredient in pharmaceutical compositions for treating parasitic infestations. It was granted on October 29, 2002, with an original expiration date in 2021.
- Extensions and Exclusivities: Through mechanisms like Hatch-Waxman Act extensions, the effective market exclusivity for NATROBA has been extended. Narsarsuaq, LLC, has asserted patent rights to maintain market exclusivity.
- Generic Challenges: The expiration and potential invalidation of key patents are prerequisites for generic entry. Generic manufacturers typically challenge existing patents to gain market access. Any successful patent challenge can significantly impact market dynamics and pricing.
Who are the Key Stakeholders in the NATROBA Market?
Multiple entities influence the market for NATROBA, from manufacturing and distribution to prescribing and reimbursement.
- Manufacturer: Narsarsuaq, LLC, is the originator company for NATROBA.
- Distributors: Pharmaceutical wholesalers and specialty pharmacies play a role in distributing NATROBA to healthcare providers and patients.
- Healthcare Providers: Dermatologists, pediatricians, and primary care physicians are the prescribers of NATROBA.
- Patients: Individuals diagnosed with scabies are the end-users.
- Payers: Insurance companies, Medicare, and Medicaid determine reimbursement rates and patient co-pays, directly influencing affordability and access.
- Generic Manufacturers: Companies developing and seeking approval for generic versions of spinosad topical suspension.
What is the Current Market Size and Demand for Scabies Treatments?
The market for scabies treatments is driven by the prevalence of the infestation and the efficacy of available therapies.
- Prevalence: Scabies is a significant public health concern globally, affecting an estimated 300 million people annually, according to the World Health Organization [1]. In the United States, estimates suggest millions of cases occur each year, particularly in crowded settings like nursing homes and childcare facilities.
- Treatment Market: The global market for scabies treatments was valued at approximately USD 1.2 billion in 2022 and is projected to grow. The market is segmented by product type (prescription vs. over-the-counter), formulation (topical, oral), and distribution channel.
- NATROBA's Share: NATROBA competes within the prescription topical treatment segment. Its market share is influenced by its efficacy, safety profile, ease of use, and pricing relative to alternatives.
What are the Approved Alternatives to NATROBA?
The therapeutic landscape for scabies includes several FDA-approved prescription and over-the-counter (OTC) options. The availability and cost of these alternatives directly impact NATROBA's market position.
- Permethrin 5% Cream: This is a first-line treatment for scabies in many guidelines. It is widely available and generally well-tolerated.
- Ivermectin: Available as both an oral medication and a topical lotion. Oral ivermectin is often used for widespread infestations or when topical treatment failure occurs.
- Crotamiton Cream: Another topical agent, though often considered less effective than permethrin or spinosad.
- Sulfur Ointment: An older, less expensive topical treatment, typically used for infants or pregnant women when other treatments are contraindicated.
- Benzyl Benzoate: Another topical option, often used in other regions.
What is the Current Pricing Structure for NATROBA?
NATROBA's pricing is influenced by its prescription status, manufacturer pricing strategies, and insurance coverage.
- Manufacturer's Suggested Retail Price (MSRP): The MSRP for a 240 mL bottle of NATROBA is typically in the range of $300 to $400, though this can fluctuate.
- Wholesale Acquisition Cost (WAC): This is the price pharmaceutical wholesalers pay to acquire drugs from the manufacturer. WAC for NATROBA can be around $280 to $350 per bottle.
- Net Price: The actual price paid by pharmacies or healthcare providers after discounts, rebates, and other adjustments is lower than WAC and varies significantly based on contracts and volume.
- Patient Out-of-Pocket Costs: These are determined by individual insurance plans. Co-pays can range from $20 to over $100 per prescription, with some plans requiring prior authorization or step therapy, which may direct patients to less expensive alternatives first. High deductible plans can result in patients paying the full WAC or a significant portion of it.
- Comparison to Alternatives:
- Permethrin 5% cream (a 60g tube) typically costs $20-$50 at retail, with insurance co-pays often in the $5-$15 range.
- Oral Ivermectin (typically a single dose pack) can range from $30-$70 without insurance, with co-pays often similar to permethrin.
What are the Regulatory Considerations Affecting NATROBA's Market?
Regulatory approvals, patent challenges, and marketing exclusivities are key determinants of market access and competition.
- FDA Approval: NATROBA received FDA approval in 2014. This approval ensures the drug has met safety and efficacy standards for its indicated use.
- Patent Expiration and Litigation: As noted, the patent landscape is dynamic. Legal challenges to patents can accelerate generic entry. For instance, if a patent is found invalid or not infringed, generic manufacturers can seek FDA approval for their versions.
- Orphan Drug Exclusivity: NATROBA does not qualify for Orphan Drug Exclusivity, which is for drugs treating rare diseases.
- Pediatric Exclusivity: The FDA may grant an additional six months of market exclusivity for a new drug if the applicant submits information relevant to the use of the drug in children. This may have been sought and granted for NATROBA.
- Generic Drug Approval Pathway: The Hatch-Waxman Act allows generic manufacturers to seek FDA approval by demonstrating bioequivalence to the branded drug. The timing of this approval depends on patent challenges and regulatory review.
What is the Projected Market Demand for NATROBA?
Market demand for NATROBA is expected to remain stable to moderately growing, contingent on several factors.
- Continued Prevalence of Scabies: The persistent occurrence of scabies infestations, particularly in community settings and developing regions, will sustain demand for effective treatments.
- Physician Preference and Prescribing Habits: Established prescribing patterns for NATROBA among dermatologists and other specialists will support its demand. Its efficacy in a single application for some patients is a significant driver.
- Patient Compliance: NATROBA's single-application regimen can enhance patient compliance compared to multi-dose treatments, potentially driving preference.
- Competition: The introduction of generic versions will likely increase the overall volume of spinosad prescriptions but could decrease the market share and revenue for the branded product.
- Reimbursement Landscape: Favorable insurance coverage and co-pay assistance programs will bolster demand by improving patient access and affordability. Conversely, restrictive formularies or high co-pays could suppress demand.
What are the Price Projections for NATROBA?
Price projections for NATROBA are multifaceted, influenced by the imminent threat of generic competition and the dynamics of the branded drug market.
- Pre-Generic Entry (Current to 12-18 months):
- Branded NATROBA: Prices are likely to remain relatively stable or see modest increases (1-3% annually), aligning with general pharmaceutical price inflation trends and manufacturer strategies to maximize revenue before generic entry. The MSRP could reach $320-$420 per bottle.
- Net Price: Rebates and discounts offered to payers and pharmacy benefit managers (PBMs) will continue to shape the net price, making it lower than WAC.
- Post-Generic Entry (18-36 months onwards):
- Branded NATROBA: Once one or more generic versions of spinosad topical suspension are approved and launched, branded NATROBA prices will likely experience a significant decline. This erosion can range from 50% to 85% of the current branded price within the first 2-3 years of generic competition, a typical pattern in the pharmaceutical market. The MSRP could fall to the $100-$150 range.
- Generic Spinosad: Generic versions will enter the market at substantially lower price points, typically 70-90% less than the branded product initially, with prices continuing to fall as more generic competitors enter. Generic prices could range from $30-$70 per bottle depending on the manufacturer and market share.
- Market Share Shift: The majority of prescriptions will likely shift to generic spinosad due to cost savings for both payers and patients. Branded NATROBA may retain a smaller share for patients who specifically request it, or if payer contracts offer some preferred status.
- Long-Term Projections (3-5 years+):
- Branded NATROBA: If the brand manages to maintain any market presence, its price will likely continue to decline slowly or stabilize at a much lower level, potentially serving a niche market.
- Generic Spinosad: Prices will continue to decline due to ongoing competition and market maturity, eventually reaching cost-plus pricing levels. The price for a generic bottle could be under $30.
Factors influencing these projections:
- Speed of Generic Approval: The timing of FDA approval for generic spinosad is a critical variable.
- Number of Generic Entrants: A higher number of generic manufacturers will accelerate price competition.
- Patent Litigation Outcomes: Any successful appeals or new legal challenges can alter the timeline for generic entry.
- Payer Strategies: Formulary placement and co-pay policies for both branded and generic spinosad will significantly impact uptake.
- Global Supply Chain Stability: Disruptions can affect the availability and cost of raw materials and finished products.
Key Takeaways
- NATROBA (spinosad topical suspension) is a prescription treatment for scabies, facing patent expirations that pave the way for generic competition.
- The current market for scabies treatments is substantial, driven by the global prevalence of the infestation.
- NATROBA competes with several established alternatives, including permethrin and ivermectin, with pricing being a key differentiator.
- Branded NATROBA prices are expected to remain stable in the short term but will likely decline significantly (50-85%) post-generic entry.
- Generic spinosad topical suspension is projected to enter the market at substantially lower prices, becoming the dominant treatment option over the next 1-3 years.
Frequently Asked Questions
-
When is the primary patent for NATROBA expected to expire or be challenged? The core patents for spinosad, such as U.S. Patent No. 6,472,441, have either expired or are subject to ongoing legal challenges and extensions. The market has been anticipating generic entry for some time, indicating that significant patent barriers may have been overcome or are being actively litigated.
-
What is the typical timeframe for price erosion of a branded drug after generic entry? Following the launch of the first generic competitor, branded drug prices typically experience a significant decline within 18-36 months, often ranging from 50% to 85% of their pre-generic levels. This erosion continues as more generic versions enter the market.
-
How will insurance formularies likely handle branded NATROBA versus generic spinosad? Insurance formularies will likely prioritize generic spinosad, placing it at a lower co-pay tier or as a preferred option. Branded NATROBA may be moved to a higher co-pay tier or require prior authorization, especially if generic alternatives are deemed therapeutically equivalent and cost-effective.
-
What is the primary driver for the demand of NATROBA and similar scabies treatments? The primary driver is the incidence and prevalence of scabies infestations globally. Factors such as population density, hygiene conditions, and outbreaks in institutional settings (e.g., nursing homes, childcare) directly influence demand. Patient and physician preference for single-application treatments also plays a role.
-
Are there any regulatory hurdles that could delay the introduction of generic spinosad topical suspension? Yes, potential regulatory hurdles include ongoing patent litigation that could result in further injunctions, FDA review timelines for generic applications, and any specific manufacturing or quality control issues identified during the approval process that differ from the reference listed drug.
Citations
[1] World Health Organization. (n.d.). Scabies. Retrieved from https://www.who.int/ (Note: Specific page not provided, assumed general WHO information on scabies prevalence)
More… ↓
