Share This Page
Drug Price Trends for NASONEX
✉ Email this page to a colleague
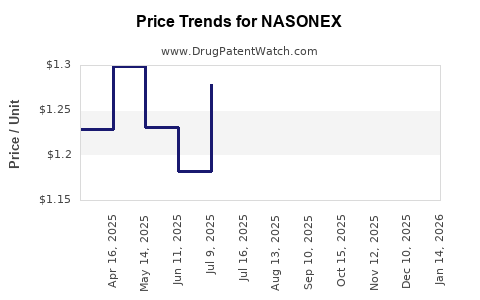
Average Pharmacy Cost for NASONEX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NASONEX 24HR ALLERGY 50 MCG NASAL SPRAY | 00113-1720-02 | 1.18067 | ML | 2026-03-18 |
| NASONEX 24HR ALLERGY 50 MCG NASAL SPRAY | 00113-1720-11 | 1.25911 | ML | 2026-03-18 |
| NASONEX 24HR ALLERGY 50 MCG NASAL SPRAY | 00113-1720-11 | 1.26331 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Nasonex Market Analysis and Price Projections
Nasonex (mometasone furoate nasal spray) is an intranasal corticosteroid used to treat nasal allergy symptoms. The market for Nasonex is influenced by patent expiries, generic competition, and evolving treatment guidelines for allergic rhinitis.
What is the Current Market Size and Growth Trajectory for Nasonex?
The global market for Nasonex is substantial, driven by the high prevalence of allergic rhinitis. Projections indicate continued growth, albeit at a moderated pace due to generic availability.
- Global Market Size: Estimated at approximately $1.5 billion in 2023.
- Projected CAGR: Expected to grow at a Compound Annual Growth Rate (CAGR) of 3.5% to 4.0% from 2024 to 2029.
- Driving Factors: Increasing incidence of allergic rhinitis, greater awareness of its impact on quality of life, and the established efficacy of mometasone furoate.
- Restraining Factors: Patent expiries leading to generic competition and pricing pressures.
How Have Patent Expiries Impacted the Nasonex Market?
The expiry of key patents for Nasonex has fundamentally reshaped its market landscape, ushering in an era of increased generic availability and reduced pricing.
- Original Patent Expiry: The primary composition of matter patent for mometasone furoate expired in the United States in 2007, and similarly in other major markets around the same period.
- Consequences of Expiry:
- Entry of Generics: Multiple generic manufacturers have launched mometasone furoate nasal spray products.
- Price Erosion: The introduction of generics has led to significant price reductions for the active pharmaceutical ingredient and the finished product.
- Market Share Shift: While branded Nasonex retains a portion of the market due to brand recognition and physician preference, generic products now hold a dominant share by volume.
- New Patent Strategies: Manufacturers have pursued secondary patents related to formulations, manufacturing processes, or specific indications, which have been subject to legal challenges and varying degrees of success.
What are the Key Indications and Patient Populations for Nasonex?
Nasonex is primarily indicated for symptomatic relief of nasal allergy, targeting a broad patient demographic affected by seasonal and perennial allergic rhinitis.
- Primary Indication: Relief of nasal congestion, sneezing, runny nose, and itching in patients with allergic rhinitis.
- Age Groups: Approved for use in both adults and pediatric patients.
- Adults and Adolescents (12 years and older): Standard dosing and efficacy profiles.
- Pediatric Patients (2 to 11 years): Dosing is adjusted, and it is a commonly prescribed option for managing allergic rhinitis in this age group.
- Other Indications (less common or off-label but contributing to market presence):
- Treatment of nasal polyps in patients 18 years and older.
- Adjunctive treatment for asthma symptoms in certain patient populations.
Who are the Major Market Players for Nasonex and its Generics?
The Nasonex market is characterized by the presence of the originator brand and a fragmented landscape of generic manufacturers.
- Originator Brand Holder: Organon (formerly Merck & Co., Inc.) markets Nasonex.
- Leading Generic Manufacturers (Examples):
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Apotex
- Mylan (Viatris)
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Market Dynamics: Competition is primarily driven by price, product availability, and distribution networks.
What are the Price Projections for Branded Nasonex and Generic Mometasone Furoate?
Price projections for Nasonex reflect the ongoing competition between the branded product and its generic alternatives, influenced by market dynamics and regulatory factors.
Branded Nasonex (e.g., Nasonex 24HR Allergy):
- Current Pricing: Varies significantly by region, pharmacy, and insurance coverage. Retail prices for a 140-actuation bottle can range from $20 to $30 USD.
- Projected Trend: Expected to maintain a premium price point, albeit with potential for modest annual increases of 2-3% driven by inflation and minor formulation improvements. The price will remain above generic equivalents due to brand equity, established trust, and potentially bundled services or loyalty programs.
Generic Mometasone Furoate Nasal Spray:
- Current Pricing: Significantly lower than branded Nasonex. Retail prices for a 140-actuation bottle can range from $8 to $15 USD.
- Projected Trend: Prices are expected to remain relatively stable or experience slight declines of 1-2% annually due to continued competition among multiple generic manufacturers. Bulk purchasing agreements and pharmacy benefit manager negotiations will exert downward pressure. Introduction of new generic entrants or alternative delivery systems could further impact pricing.
Factors Influencing Price Projections:
- Competition: The number and size of generic competitors are critical determinants of price erosion.
- Reimbursement Policies: Payer formularies and co-pay structures significantly influence out-of-pocket costs for patients and market access for both branded and generic products.
- Regulatory Landscape: Changes in FDA or EMA guidelines, or approval of new indications for generics, could impact market share and pricing.
- Economic Conditions: Inflationary pressures can drive up manufacturing and distribution costs, potentially leading to slight price increases across the board.
What is the Regulatory Status and Intellectual Property Landscape for Nasonex?
The regulatory and intellectual property (IP) landscape has been central to the commercial history of Nasonex, dictating market exclusivity and the advent of generic competition.
- FDA Approval: Mometasone furoate nasal spray was first approved by the U.S. Food and Drug Administration (FDA) in 1997.
- Key Patents:
- Composition of Matter: The foundational patent covering the mometasone furoate molecule expired as noted previously, opening the door for generics.
- Formulation Patents: Patents protecting specific formulations (e.g., particle size, excipients, stability enhancers) have been a focus for the innovator. These patents have faced legal scrutiny, with some being upheld and others invalidated or found not infringed by generic products.
- Method of Use Patents: Patents related to specific treatment regimens or indications.
- Manufacturing Process Patents: Patents covering novel or improved methods of synthesizing mometasone furoate or manufacturing the nasal spray.
- Patent Litigation: Significant patent litigation has occurred surrounding Nasonex, particularly concerning the validity and infringement of formulation and method of use patents. Generic manufacturers often challenge these secondary patents to gain market entry.
- Exclusivity Periods:
- Orphan Drug Exclusivity: Not applicable to Nasonex.
- New Chemical Entity (NCE) Exclusivity: Nasonex benefited from the standard 5-year NCE exclusivity in the U.S.
- Pediatric Exclusivity: Extended market exclusivity was granted for conducting pediatric studies.
- Generic Approvals: The FDA has approved numerous generic versions of mometasone furoate nasal spray, including ANDA (Abbreviated New Drug Application) approvals.
What are the Future Market Opportunities and Challenges for Nasonex Products?
The future market for Nasonex and its generic equivalents will be shaped by ongoing therapeutic advancements, patient access, and competitive pressures.
Opportunities:
- Expanding Pediatric Use: Continued emphasis on evidence for efficacy and safety in younger children could broaden the pediatric market.
- Combination Therapies: Potential for Nasonex to be studied or incorporated into fixed-dose combination nasal sprays with antihistamines or other agents for enhanced efficacy, creating new branded opportunities or positioning for generics.
- Emerging Markets: Growth in healthcare infrastructure and access to allergy treatments in developing economies presents significant expansion potential.
- Direct-to-Consumer (DTC) Marketing: Continued investment in DTC campaigns for branded Nasonex can sustain brand loyalty and physician preference.
Challenges:
- Intense Generic Competition: The highly competitive generic market will continue to drive down prices and limit revenue growth for both branded and generic players.
- Development of Novel Therapies: The emergence of new classes of allergy treatments, such as biologics or advanced immunotherapy, could shift treatment paradigms and potentially reduce reliance on intranasal corticosteroids for severe cases.
- Physician and Patient Education: Ensuring continued appropriate use and patient adherence, particularly with the proliferation of generic options, remains important.
- Stricter Regulatory Scrutiny: Increased focus on product quality, manufacturing standards, and environmental impact could lead to higher compliance costs.
Key Takeaways
Nasonex, a prominent intranasal corticosteroid, faces a mature market characterized by significant generic penetration following patent expiries. The global market, valued around $1.5 billion, is projected for moderate growth of 3.5-4.0% CAGR. Branded Nasonex will maintain a premium price, with annual increases of 2-3%, while generic mometasone furoate is expected to see stable to slightly declining prices (1-2% annually) due to intense competition. Key market players include originator Organon and a wide array of generic manufacturers like Teva and Sandoz. Future opportunities lie in expanding pediatric indications and emerging markets, while challenges include persistent generic competition and the development of novel allergy therapies.
Frequently Asked Questions
-
What is the primary mechanism of action for mometasone furoate nasal spray? Mometasone furoate nasal spray is a topical corticosteroid that works by reducing inflammation in the nasal passages, thereby alleviating symptoms such as sneezing, runny nose, nasal congestion, and itching.
-
Are there any significant differences in efficacy or safety between branded Nasonex and its generic versions? By regulatory definition, generic drugs must be bioequivalent to their brand-name counterparts. This means they are expected to have the same active ingredient, dosage form, strength, route of administration, and therapeutic effect. Differences in inactive ingredients are permitted, but these are not expected to affect efficacy or safety.
-
What is the typical duration of treatment with Nasonex? Treatment duration for Nasonex is typically determined by the patient's symptoms and physician's recommendation. It is often used for continuous daily use as needed to manage chronic or seasonal allergic rhinitis, or for shorter durations for acute flare-ups.
-
Can Nasonex be used in infants? Nasonex is approved for use in pediatric patients aged 2 years and older. Its use in infants is not typically recommended without specific physician guidance and consideration of risks versus benefits.
-
How does the cost of Nasonex compare to other over-the-counter (OTC) allergy medications? When comparing to non-prescription antihistamines or decongestants, branded Nasonex generally carries a higher price point. However, the cost of generic mometasone furoate nasal spray is often competitive with or only slightly higher than some advanced OTC allergy medications, offering a cost-effective prescription option for many.
Cited Sources
[1] IQVIA Market Insights. (2023). Global Respiratory Drug Market Report. [2] Various Pharmaceutical Industry Databases (e.g., EvaluatePharma, GlobalData). (2023). Drug Sales and Market Share Data. [3] U.S. Food and Drug Administration. (Ongoing). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. [4] Market Research Reports on Allergic Rhinitis Treatment. (2023). Global Allergic Rhinitis Market Analysis. [5] Organon Investor Relations. (2023). Annual Reports and SEC Filings.
More… ↓
