Share This Page
Drug Price Trends for NASCOBAL
✉ Email this page to a colleague
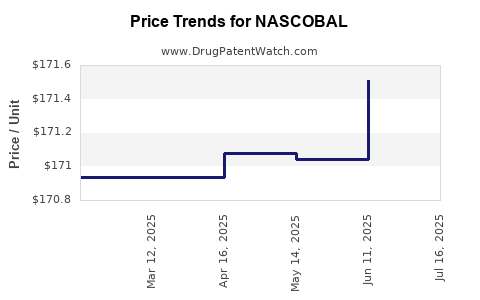
Average Pharmacy Cost for NASCOBAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NASCOBAL 500 MCG NASAL SPRAY | 49884-0270-82 | 171.73925 | EACH | 2025-07-23 |
| NASCOBAL 500 MCG NASAL SPRAY | 49884-0270-82 | 171.51066 | EACH | 2025-06-18 |
| NASCOBAL 500 MCG NASAL SPRAY | 49884-0270-82 | 171.04029 | EACH | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NASCOBAL (cobalamin)
Overview
NASCOBAL is a vitamin B12 supplement typically used for treating and preventing cobalamin deficiency. It is marketed in various forms, including injections, tablets, and sublingual formulations. The drug's market hinges on the prevalence of deficiency, demographic trends, competing therapies, and regulatory status.
Market Size and Growth
The global vitamin B12 supplements market was valued at approximately $1.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 6% from 2023 to 2030, reaching $2 billion by 2030. The increasing prevalence of vitamin B12 deficiency, especially among aging populations, vegetarians, and those with malabsorptive conditions, drives this growth.
Key Market Drivers
- Aging Populations: The global increase in elderly populations increases demand for B12 supplementation due to malabsorption issues.
- Vegetarian and Vegan Diets: Rising adoption amplifies demand since B12 sources are primarily animal-based.
- Diagnosed Deficiencies: Increased screening for anemia and neurological symptoms indicates higher usage.
- Emerging Markets: Greater access and awareness in Asia, Latin America, and Africa expand potential markets.
Competitive Landscape
Major players include:
- Pfizer (e.g., Nascobal nasal spray, injections)
- Merck & Co.
- Ferring Pharmaceuticals
- B. Braun Melsungen AG
Generic formulations and emerging biosimilar options are expected to intensify price competition.
Pricing Dynamics
-
Brand vs. Generic
Brand-name formulations typically cost between $30 and $80 per vial or pack. Generics are priced approximately 30-50% lower. -
Route of Administration
- Injections: $10-$50 per dose, depending on dosage and provider markups.
- Nasal Sprays: $30-$70, with some priced higher for combination formulas.
- Oral Tablets: $10-$30 for a month's supply.
-
Market Trends
Price variance exists across geographies, with U.S. prices generally higher than in Europe or emerging markets. Insurance coverage affects patient out-of-pocket costs; many insurers cover injectable and nasal formulations.
Future Price Projections
- In developed markets, inflation of prices is constrained by generic competition, with prices expected to stay within current ranges unless new formulations or delivery methods emerge.
- By 2030, entry of biosimilars or innovative delivery (e.g., long-acting injectables) may exert downward pressure on prices.
- If a new formulation demonstrates improved compliance or efficacy, it could command a premium of 10-15% over older options.
Regulatory and Policy Impacts
Expansion of reimbursement policies, especially in the U.S. through Medicare and Medicaid, influences pricing. Efforts to reduce healthcare costs could pressure manufacturers to lower prices further.
Summary Table
| Formulation | Current Price Range (USD) | Projected Price Trend | Key Factors |
|---|---|---|---|
| Injectable | $10 - $50 per dose | Stable to slight decrease | Competition, healthcare policy |
| Nasal spray | $30 - $70 per bottle | Slight decrease possible | Innovation, biosimilar entry |
| Oral tablets | $10 - $30 per month | Stable | Over-the-counter expansion |
Key Takeaways
- The NASCOBAL market is driven by demographic trends, diet shifts, and increasing diagnosis rates of B12 deficiency.
- Current pricing ranges vary by formulation and geography but are expected to remain relatively stable due to market competition.
- Biosimilar and innovative delivery formats could reduce costs and alter pricing dynamics over the next decade.
- Regulatory changes favoring reimbursement and access will influence retail prices and market penetration.
- Market expansion into emerging regions remains a significant growth opportunity.
FAQs
1. What factors influence NASCOBAL’s pricing globally?
Pricing is affected by manufacturing costs, competition from generics, route of administration, healthcare reimbursement policies, and regional economic conditions.
2. How might biosimilars impact NASCOBAL’s market price?
Biosimilars can introduce price competition, potentially lowering costs by 30-50% over branded formulations within 3-5 years of market entry.
3. What are the main routes of NASCOBAL administration?
Injectable forms, nasal sprays, and oral tablets are common, with injections generally being the most cost-effective at scale.
4. What demographic factors are expected to influence demand?
Aging populations, vegetarian/vegan diets, and increased screening for deficiency all contribute to rising demand.
5. Are there new formulations planned to disrupt this market?
While specific innovations remain under development, long-acting injectables and improved nasal delivery systems are likely candidates for future market entry.
Sources
- MarketWatch. "Vitamin B12 Supplements Market Size, Share & Trends." 2022.
- Allied Market Research. "Vitamin B12 Supplements Market by Form and Distribution Channel," 2023.
- U.S. Food and Drug Administration (FDA). "Drug Price and Reimbursement Policies," 2022.
- IQVIA. "Global Pharmaceutical Market Data," 2022.
- Statista. "Vitamin B12 Market Revenue Forecast," 2023.
More… ↓
