Share This Page
Drug Price Trends for NASAL SPRAY
✉ Email this page to a colleague
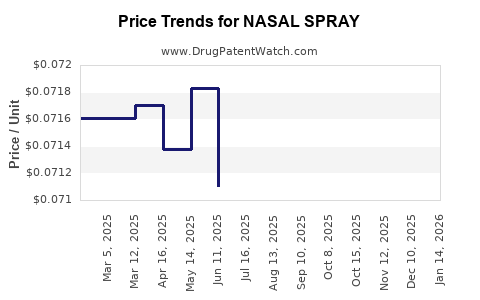
Average Pharmacy Cost for NASAL SPRAY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NASAL SPRAY 0.05% | 45802-0410-59 | 0.07371 | ML | 2026-03-18 |
| NASAL SPRAY ORIGINAL 0.05% | 46122-0165-10 | 0.07371 | ML | 2026-03-18 |
| NASAL SPRAY 0.05% | 11527-0140-55 | 0.07371 | ML | 2026-03-18 |
| NASAL SPRAY 0.05% | 45802-0410-59 | 0.07058 | ML | 2026-02-18 |
| NASAL SPRAY ORIGINAL 0.05% | 46122-0165-10 | 0.07058 | ML | 2026-02-18 |
| NASAL SPRAY 0.05% | 11527-0140-55 | 0.07058 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for NASAL SPRAY
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| NASAL SPRAY (OXYMETAZOLINE) | United Drug Supply, Inc. | 00113-0304-10 | 30ML | 2.56 | 0.08533 | ML | 2023-12-01 - 2028-11-30 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Nasal Spray Drug Market: Patent Landscape and Price Forecasts
This report analyzes the current patent landscape and projects price trends for nasal spray drugs. The market is characterized by a diverse range of therapeutic applications, with significant patent expirations anticipated in the coming years, potentially leading to price erosion for originator products. Generic competition is a key driver of price dynamics.
What are the Key Therapeutic Areas for Nasal Sprays?
Nasal spray drugs target a broad spectrum of conditions, leveraging the nasal route for localized or systemic delivery. Major therapeutic areas include:
- Allergy and Rhinitis: This segment is dominated by corticosteroids (e.g., fluticasone, mometasone) and antihistamines (e.g., azelastine, olopatadine). These formulations provide direct relief from nasal congestion, sneezing, and itching.
- Migraine: Calcitonin gene-related peptide (CGRP) receptor antagonists and ergotamine-based drugs are formulated as nasal sprays for rapid migraine symptom relief, offering an alternative to oral or injectable administration.
- Pain Management: Opioid analgesics and other pain relievers are available as nasal sprays for faster onset of action in managing moderate to severe pain.
- Hormone Replacement Therapy (HRT): Oxytocin and certain estrogen-based therapies are delivered via nasal spray for conditions such as lactation induction or menopausal symptom management.
- Respiratory Infections: Antiviral and antibiotic nasal sprays are used for the prevention or treatment of respiratory tract infections, including influenza and sinusitis.
- Neurological Disorders: Emerging applications include drugs for Parkinson's disease (e.g., apomorphine) and erectile dysfunction, utilizing the nasal route for improved bioavailability and faster therapeutic effect.
What is the Current Patent Expiration Landscape for Nasal Sprays?
Patent expiration is a critical factor influencing market competition and drug pricing. For many established nasal spray drugs, key patents have expired or are nearing expiration, paving the way for generic market entry.
Major Nasal Spray Drugs Approaching Patent Expiration
| Drug Name | Active Ingredient(s) | Primary Indication(s) | Original FDA Approval Date | Key Patent Expiration Year (Estimated) | Notes |
|---|---|---|---|---|---|
| Flonase | Fluticasone propionate | Allergic Rhinitis | 1996 | Expired | OTC availability since 2014. |
| Nasonex | Mometasone furoate | Allergic Rhinitis | 1997 | Expired | Generic versions available. |
| Rhinocort Aqua | Budesonide | Allergic Rhinitis | 2000 | Expired | OTC availability. |
| Narcan | Naloxone | Opioid Overdose | 1975 | Expired (for IV/IM); Nasal Spray patent extended | Nasal spray formulation extended exclusivity. |
| Migranal | Dihydroergotamine | Migraine | 1998 | Expired | Generic versions available. |
| Stiolto Respimat | Tiotropium/Olodaterol | COPD | 2015 | 2029 | Device patent is a key component. |
| Xhance | Fluticasone propionate | Nasal Polyps | 2019 | 2036 | Device and formulation patents provide extended protection. |
Source: FDA Orange Book, Patent databases, Pharmaceutical news.
The expiration of patents for widely prescribed corticosteroids like fluticasone propionate and mometasone furoate has already led to significant price decreases due to generic availability. For newer entrants or those with extended patent protection via formulation or device patents, pricing power may be sustained for longer.
How Do Formulation and Device Patents Impact Nasal Spray Pricing?
Formulation and device patents play a crucial role in extending market exclusivity for nasal spray products, thereby influencing pricing strategies.
- Device Innovation: Novel delivery devices that improve spray consistency, droplet size, ease of use, or patient compliance can be independently patented. For example, metered-dose inhalers or devices with advanced nozzle technology may receive separate patent protection, even if the active pharmaceutical ingredient (API) patent has expired. This can prevent generic manufacturers from easily replicating the product if they cannot independently develop a comparable device. Examples include breath-actuated or single-dose devices designed for specific therapeutic needs.
- Formulation Enhancements: Patents can be granted for new formulations that improve API stability, bioavailability, absorption rate, or reduce irritation. This includes novel excipients, drug layering within particles, or specialized suspension technologies. For instance, a formulation patent might cover a specific particle size distribution that enhances nasal deposition or a mucoadhesive system that prolongs drug residence time.
- Combination Products: Patents covering the combination of two or more active ingredients in a single nasal spray formulation can also extend market exclusivity. These products offer convenience and potentially synergistic therapeutic effects.
- Manufacturing Processes: Innovative manufacturing processes that are more efficient or yield a higher quality product can also be patented, indirectly protecting the commercial viability of the drug.
These extended exclusivity periods allow originator companies to maintain higher price points by limiting direct competition from generics that would typically enter upon API patent expiration.
What are the Price Drivers for Nasal Spray Drugs?
Multiple factors influence the pricing of nasal spray drugs, ranging from development costs to market dynamics.
- Research and Development Costs: High R&D expenditure for drug discovery, formulation development, clinical trials, and regulatory approval is a primary driver of initial drug pricing. For novel nasal spray products, especially those targeting complex diseases or employing advanced delivery systems, these costs can be substantial.
- Market Exclusivity and Patent Protection: As detailed previously, patent status is a dominant pricing determinant. Products with strong patent protection and extended exclusivity periods command higher prices. Conversely, drugs facing imminent patent expiration or significant generic competition will experience downward pricing pressure.
- Therapeutic Value and Unmet Needs: Drugs addressing significant unmet medical needs or offering superior efficacy, safety, or convenience compared to existing treatments can justify premium pricing. This is particularly relevant for indications like severe migraines or rare respiratory conditions.
- Competition: The presence and intensity of competition from other drugs within the same therapeutic class, regardless of delivery method, influence pricing. A market with multiple effective treatments will typically see more competitive pricing.
- Manufacturing Complexity and Cost of Goods Sold (COGS): The complexity of manufacturing the API, the specialized delivery device, and formulation processes all contribute to COGS. Nasal spray devices, especially those with intricate mechanisms, can have higher manufacturing costs than simple oral dosage forms.
- Reimbursement Policies and Payer Landscape: Payer negotiations and formulary placement significantly impact net prices. Health insurance providers and government payers influence drug accessibility and pricing through coverage decisions, co-pays, and rebate agreements.
- Market Size and Demand: The overall market size and patient demand for a particular nasal spray indication affect pricing power. Larger, more prevalent conditions may attract more competition but also offer greater sales volume.
- Brand Reputation and Marketing: Established brands with strong clinical track records and effective marketing strategies can often command higher prices than newer or less-known alternatives.
What are the Price Projections for Nasal Spray Drugs?
Price projections for nasal spray drugs vary significantly based on their patent status, therapeutic indication, and competitive landscape.
Price Projection Scenarios
- Patented, Novel Therapies: For nasal spray drugs with strong, ongoing patent protection targeting significant unmet needs (e.g., certain neurological or rare disease applications), prices are projected to remain high or experience modest annual increases, potentially in the range of +4% to +8% annually, driven by inflation, continued R&D investment, and market penetration.
- Branded Generics / Extended Exclusivity: Products with expired API patents but protected by strong device or formulation patents, or those marketed as branded generics with enhanced patient support, may see prices stabilize or increase at a lower rate, perhaps +2% to +5% annually. These prices will be sensitive to any challenges to their extended exclusivity.
- Generic and Biosimilar Competition: For nasal sprays where the API patent has expired and multiple generic manufacturers have entered the market, prices are projected to decline sharply post-entry. The initial price of a generic nasal spray can be 40% to 60% lower than the originator's list price. Continued generic competition can lead to further price erosion, with potential for declines of 10% to 20% per year in the initial years of generic availability until market stabilization.
- Over-the-Counter (OTC) Nasal Sprays: Established OTC nasal corticosteroid sprays, like fluticasone propionate and budesonide, are already subject to intense price competition among numerous brands and store brands. Prices are expected to remain relatively stable with minor fluctuations driven by promotional activities and retailer pricing strategies, likely seeing minimal price changes year-over-year (<2%).
Example of Price Erosion: Consider a branded nasal spray with an original WAC (Wholesale Acquisition Cost) of $300 per unit. Upon generic entry, the WAC for a generic version might drop to $150. Within 1-2 years, further competition could reduce the generic WAC to $100-$120.
The overall trend for the nasal spray market indicates a bifurcated pricing future: high prices for innovative, patented products and significant price declines for off-patent drugs facing generic competition.
Key Takeaways
- Nasal sprays are utilized across diverse therapeutic areas, with allergy, migraine, and pain management being prominent.
- Patent expiration is a primary driver of price reduction; many older, widely used nasal sprays are now off-patent and available generically.
- Device and formulation patents are critical for extending market exclusivity and sustaining higher prices for originator products.
- Pricing is influenced by R&D costs, patent protection, therapeutic value, competition, manufacturing costs, and payer policies.
- Projections show continued high pricing for novel, patented nasal sprays, while generic competition will drive significant price erosion for off-patent drugs.
Frequently Asked Questions
1. How quickly do prices fall after a nasal spray drug loses patent protection?
Price erosion typically begins immediately upon the entry of the first generic competitor. The magnitude of the initial price drop can range from 40% to 60% of the originator's wholesale acquisition cost. Subsequent price reductions can occur as more generic manufacturers enter the market, potentially leading to further declines of 10% to 20% annually in the initial years of generic availability.
2. Are all nasal spray drugs subject to generic competition?
No, not all nasal spray drugs are equally susceptible to generic competition. Drugs with expired active pharmaceutical ingredient (API) patents are prime targets. However, novel delivery devices, unique formulations, or combination products may have their own patents that extend market exclusivity beyond the API patent expiration. Additionally, some nasal sprays may be for rare diseases with limited market size, making generic development less economically attractive.
3. What impact do over-the-counter (OTC) nasal sprays have on the prescription market pricing?
OTC nasal sprays, particularly corticosteroid-based allergy relief products, have established a competitive market that influences pricing expectations for similar prescription products. The widespread availability and lower price points of OTC options can create pressure on prescription versions, especially when prescription alternatives offer only marginal clinical advantages for less severe conditions.
4. How do drug shortages affect nasal spray pricing?
Drug shortages can temporarily disrupt normal pricing dynamics. During a shortage, the limited supply may allow for price increases, particularly for critical medications where demand significantly outstrips availability. However, these price increases are often temporary and subject to regulatory scrutiny. Shortages can also incentivize manufacturers to ramp up production or new generics to enter the market more quickly.
5. What role do biosimil regulations play in the nasal spray market?
Biosimilar regulations, as they are currently defined, primarily apply to biologic drugs. Most nasal sprays are small molecule drugs. Therefore, biosimilar regulations do not directly impact the pricing of traditional small molecule nasal spray drugs. The equivalent for small molecules is generic drug regulation, which governs the entry and pricing of chemically identical copies.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA website]
[2] Pharmaceutical News Archives. (Various dates). Industry analyses and drug patent expiration reports.
[3] Patent Databases (e.g., USPTO, Espacenet). (Ongoing). Patent filing and grant information for pharmaceutical products and delivery devices.
More… ↓
