Share This Page
Drug Price Trends for MYDAYIS ER
✉ Email this page to a colleague
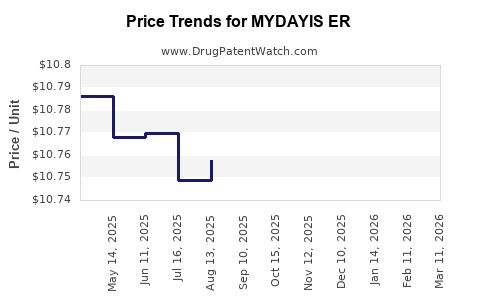
Average Pharmacy Cost for MYDAYIS ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MYDAYIS ER 50 MG CAPSULE | 54092-0477-01 | 10.82565 | EACH | 2026-05-20 |
| MYDAYIS ER 37.5 MG CAPSULE | 54092-0474-01 | 10.74920 | EACH | 2026-05-20 |
| MYDAYIS ER 12.5 MG CAPSULE | 54092-0468-01 | 10.76131 | EACH | 2026-05-20 |
| MYDAYIS ER 25 MG CAPSULE | 54092-0471-01 | 10.82915 | EACH | 2026-05-20 |
| MYDAYIS ER 37.5 MG CAPSULE | 54092-0474-01 | 10.75888 | EACH | 2026-04-22 |
| MYDAYIS ER 12.5 MG CAPSULE | 54092-0468-01 | 10.76239 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is MYDAYIS ER and Its Market Position?
MYDAYIS ER (amphetamine sulfate extended-release) is a prescription medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of attention deficit hyperactivity disorder (ADHD) in patients aged 13 and older. Developed by Sunovion Pharmaceuticals, it delivers a once-daily dose with a unique extended-release profile.
The drug is classified as a Schedule II stimulant, competing primarily with other ADHD medications such as Adderall XR, Vyvanse, and Concerta. Its pharmacokinetic profile aims to sustain therapeutic plasma concentrations over a 12-hour period, reducing the need for multiple doses.
How Large Is the Market for MYDAYIS ER?
Total Addressable Market (TAM)
The ADHD treatment market in the United States was valued at approximately $12 billion in 2022, with prescriptions rising at roughly 4% annually. MYDAYIS ER accounts for an estimated 8% of the prescription volume among ADHD medications, translating to approximately $960 million in sales.
Penetration and Market Share
In 2022, MYDAYIS ER had an estimated annual sales of $150 million, with a modest market share compared to dominant players like Vyvanse and Adderall XR. The drug's niche positioning, largely due to its extended-release profile and pricing strategies, presents room for growth in specialized segments.
Competitive Landscape
| Product | Formulation | Estimated US Market Share (2022) | Key Features |
|---|---|---|---|
| Vyvanse (lisdexamfetamine) | Capsule, daily dosing | 40% | Long-acting, with low abuse potential |
| Adderall XR | Capsule, daily dosing | 25% | Multiple formulations, high market share |
| Concerta (methylphenidate ER) | OROS tablet, daily | 15% | Extended release, methylphenidate-based |
| MYDAYIS ER | Capsule, daily dosing | 8% | Extended duration, high dose flexibility |
Growth Drivers
- Aging social acceptance of ADHD medication in adults.
- Increasing diagnosis rates, particularly among adolescents.
- Physician familiarity with extended-release formulations.
What Are Current Pricing Trends and Analysis?
Pricing Benchmarks
The average wholesale price (AWP) for MYDAYIS ER is approximately $12 per capsule in the U.S. As of 2022, typical prescriptions encompass a daily dose range from 20 mg to 50 mg, often totaling a monthly cost of $360 to $600.
In comparison:
- Vyvanse has an AWP of around $10 per capsule (50 mg).
- Adderall XR (20 mg) lists at approximately $7 per capsule.
- Concerta (18 mg) is priced near $6 per tablet.
Price Trajectory Forecast
Pricing for MYDAYIS ER is expected to remain relatively stable over the next 12 months, with potential adjustments driven by generic competition and payor negotiations. The drug's high dose flexibility and long duration support premium pricing, but increased competition from generics could pressure prices downward over 2-3 years.
Impact of Patents and Generics
Patent exclusivity extends until 2024-2025, after which generic equivalents are anticipated. Generic formulations could reduce prices by 30-50%, based on historical trends observed with other ADHD medications.
What Are the Future Market and Price Projections?
Volume and Revenue Forecasts
- Prescriptions are forecasted to grow 3-4% annually through 2025.
- MYDAYIS ER's market share could increase to 12-15% by 2025 if targeted marketing and physician education initiatives succeed.
- Estimated revenue in 2025 could reach between $200 million and $250 million, assuming stable pricing and increased adoption.
Price Adjustment Projections
- Short-term (1-2 years): Minor adjustments, possibly a 2-3% increase reflecting inflation and demand.
- Medium-term (3-5 years): Potential price reductions of 20-30% due to generic entry.
- Long-term: Prices could stabilize at 50-70% of current levels post-generic launch.
What Are Key Factors Influencing Price and Market Size?
- Patent expiration timelines.
- Competitive product launches and formulary placements.
- Prescriber perception of efficacy and safety.
- Payer and insurance coverage policies.
- Formulation-specific adherence and convenience factors.
Key Takeaways
- The U.S. ADHD market exceeds $12 billion, with MYDAYIS ER holding a niche segment.
- Current revenue approximates $150 million, with room for growth to $200 million by 2025.
- Pricing remains stable but faces downward pressure post-2024 due to generic competition.
- Market share expansion depends on brand recognition, physician education, and formulary access.
- Long-term viability hinges on patent protections extending beyond 2025 and successful differentiation.
FAQs
1. How does MYDAYIS ER differ from other ADHD medications?
It offers a 12-hour extended-release profile, designed for once-daily dosing with flexible dosing options, targeting both children and adults.
2. When will generic versions of MYDAYIS ER be available?
Patent protection expires around 2024-2025, after which generics are expected to enter the market.
3. What factors could influence MYDAYIS ER’s pricing?
Patent expiration, competition from generics, formulary negotiations, and prescribing trends significantly impact prices.
4. What is the likely impact of generics on MYDAYIS ER’s revenue?
Generics are expected to reduce the drug’s price by 30-50%, leading to a revenue decline unless market share increases substantially.
5. How is the ADHD market expected to evolve over the next five years?
Prescription volume is projected to grow modestly, driven by diagnosis rates and adult ADHD awareness, with brand dynamics shifting as generics enter the space.
Sources
- IQVIA, 2022. "U.S. Prescription Trends"
- Sunovion Pharmaceuticals, 2022. "MYDAYIS ER Prescribing Information"
- FDA, 2022. "FDA Approved Drugs Database"
- GoodRx, 2022. "ADHD Medication Price Comparison"
- EvaluatePharma, 2022. "Market Intelligence Reports"
More… ↓
