Share This Page
Drug Price Trends for MYCOPHENOLIC ACID DR
✉ Email this page to a colleague
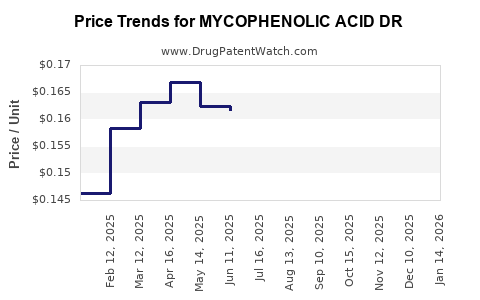
Average Pharmacy Cost for MYCOPHENOLIC ACID DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MYCOPHENOLIC ACID DR 180 MG TB | 24979-0160-44 | 0.11996 | EACH | 2026-04-22 |
| MYCOPHENOLIC ACID DR 180 MG TB | 59651-0621-08 | 0.11996 | EACH | 2026-04-22 |
| MYCOPHENOLIC ACID DR 180 MG TB | 16729-0261-29 | 0.11996 | EACH | 2026-04-22 |
| MYCOPHENOLIC ACID DR 180 MG TB | 60505-2965-07 | 0.11996 | EACH | 2026-04-22 |
| MYCOPHENOLIC ACID DR 180 MG TB | 00904-6785-04 | 0.11996 | EACH | 2026-04-22 |
| MYCOPHENOLIC ACID DR 360 MG TB | 72888-0200-12 | 0.22343 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Mycophenolic Acid DR
What Is the Current Market Size and Demand for Mycophenolic Acid DR?
Mycophenolic acid delayed-release (DR) formulations are primarily used for immunosuppression in transplant recipients and some autoimmune diseases. The global market for mycophenolic acid drugs, including Mycophenolic Acid DR, was valued at approximately $1.1 billion in 2022. The compound's demand stems from its established efficacy in preventing organ rejection post-transplantation, especially kidney, liver, and heart transplants.
The key market drivers include:
- Increasing number of organ transplants worldwide.
- Rising incidence of autoimmune conditions like lupus nephritis, prompting off-label uses.
- Expanded approval in emerging markets.
Market growth is forecasted at a compound annual growth rate (CAGR) of 4-5% over the next five years, driven by expanding transplant programs and generic entry.
How Does Competition Impact Pricing and Market Dynamics?
Market players include Pfizer (brand: CellCept, which includes mycophenolate mofetil), Sandoz, Dr. Reddy's Laboratories, and other generics manufacturers. The availability of generic formulations drives prices downward.
Price positioning:
- Branded formulations (e.g., Pfizer's CellCept) typically retail between $700-$900 per month.
- Generic mycophenolic acid DR products are priced at roughly $150-$300 per month, offering significant savings.
Market penetration:
- Generics hold 70-80% of the market share in mature regions.
- Patent expirations for branded versions occurred around 2021, opening pathways for generics.
Pricing trends: Price erosion has stabilized around 20-30% annually since 2015, reflecting increased generic penetration and regulatory approvals. Future price reductions are expected to slow as market saturation approaches.
What Are the Regulatory and Patent Landscapes Affecting Pricing?
Patent status:
- The primary patent for branded mycophenolic acid DR expired in 2021 in the US and EU.
- Remaining patents for formulation and manufacturing processes may extend exclusivity until 2025-2027 in some jurisdictions.
Regulatory approvals:
- The drug is approved for solid organ transplantation in multiple countries.
- Regulatory agencies have approved generic formulations, with some receiving abbreviated pathways (ANDA submissions in the US, EMA biosimilar pathways).
Impact: Patent expirations facilitate price declines, but the timing of generic market entry significantly influences available pricing. Companies holding early rights to production tend to set lower prices initially, with subsequent reductions following broader market entry.
What Are the Pricing Projections for the Next Five Years?
Baseline scenario:
| Year | Estimated Price per Month (USD) | Notes |
|---|---|---|
| 2023 | $180 - $250 | Widely available generics; market stabilization occurs. |
| 2024 | $160 - $240 | Competition intensifies; some price reductions expected. |
| 2025 | $150 - $220 | Patent cliff impacts pricing; potential new formulations. |
| 2026 | $140 - $210 | Further market saturation; price plateau. |
| 2027 | $130 - $200 | Price stabilization, market maturity. |
Emerging markets may experience higher prices initially ($200-$300) due to importation costs and regulatory hurdles but typically follow global pricing trends within 1-2 years.
How Will Market and Pricing Trends Impact R&D and Investment?
- Companies may focus on novel formulations or combination therapies to extend patent life cycles.
- Investors should monitor patent expiration timelines and regulatory approvals to assess market entry risks.
- Manufacturers investing in cost-effective production can leverage margin advantages in the competitive generic market.
Key Considerations
- Pricing reductions due to patent expiry are already reflected in current market prices.
- Patent litigations and regulatory delays can alter timelines.
- Entry of biosimilars or biobetters could further suppress pricing.
- Strategic differentiation through formulation improvements may preserve margins longer.
Key Takeaways
- The global market for mycophenolic acid DR was approximately $1.1B in 2022.
- Generic competition has driven prices down, with a projected decline of approximately 15-20% over the next five years.
- Patent expiry since 2021 has accelerated price erosion, but residual patent protections may temporarily sustain higher prices.
- Regulatory landscape influences market entry timing and pricing strategies.
- Future innovations and biosimilar developments could further impact market pricing and demand.
FAQs
1. How does patent expiration influence pricing for mycophenolic acid DR?
Patent expiration typically triggers market entry of generics, leading to significant price reductions due to increased competition.
2. What factors could slow down price declines?
Limited patent protections, manufacturing barriers, regulatory delays, or supply chain disruptions can slow price declines.
3. Are there regional differences in pricing?
Yes. Prices tend to be higher in emerging markets owing to import costs and regulatory hurdles, but these usually follow global trends within 1-2 years.
4. Will new formulations or combination therapies affect the market?
Yes. Innovative formulations or combination therapies can extend patent protections and market share, potentially stabilizing prices.
5. How does the demand outlook compare globally?
Demand increases with the growth of transplant procedures and autoimmune disease treatments, but saturated markets in developed regions limit growth potential.
References
- MarketWatch. (2023). "Mycophenolic Acid Market Size, Share & Trends."
- IQVIA. (2022). "Global Transplant Market Report."
- FDA. (2022). Approved Drug Database.
- European Medicines Agency. (2022). "Regulatory Status of Mycophenolic Acid DR."
- McKinsey & Company. (2021). "The Impact of Patent Expirations on Immunosuppressants."
More… ↓
