Share This Page
Drug Price Trends for MOUNJARO
✉ Email this page to a colleague
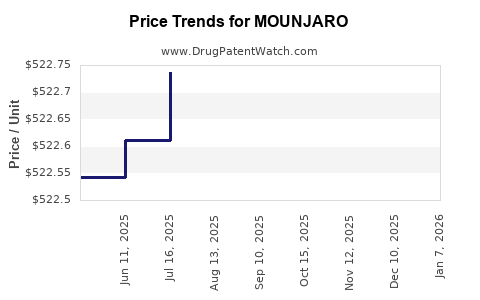
Average Pharmacy Cost for MOUNJARO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MOUNJARO 15 MG/0.5 ML PEN | 00002-1457-80 | 538.63980 | ML | 2026-01-01 |
| MOUNJARO 7.5 MG/0.5 ML PEN | 00002-1484-80 | 538.73971 | ML | 2026-01-01 |
| MOUNJARO 10 MG/0.5 ML PEN | 00002-1471-80 | 538.70922 | ML | 2026-01-01 |
| MOUNJARO 5 MG/0.5 ML PEN | 00002-1495-80 | 538.74379 | ML | 2026-01-01 |
| MOUNJARO 2.5 MG/0.5 ML PEN | 00002-1506-80 | 538.79537 | ML | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Mounjaro (tirzepatide)
What is Mounjaro?
Mounjaro (tirzepatide) is a prescription medication developed by Eli Lilly. It functions as a dual GIP and GLP-1 receptor agonist to improve glycemic control in type 2 diabetes patients. Approved by the FDA in May 2022, Mounjaro has gained significant attention for its efficacy and weight loss benefits.
Market Size and Growth Drivers
| Category | Data/Estimate | Source |
|---|---|---|
| Type 2 Diabetes Global Market (2023) | $79.9 billion | [1] |
| Projected CAGR (2023-2030) | 7.2% | [2] |
| Obesity Market Global (2023) | $254 billion | [3] |
| Obesity Market CAGR (2023-2030) | 8% | [4] |
Key Factors:
- Increasing prevalence of type 2 diabetes and obesity.
- Insulin resistance tied to lifestyle factors.
- Rise in aging populations.
Mounjaro’s Market Penetration
Eli Lilly projects that Mounjaro could reach $12 billion in annual sales by 2030, driven by its dual indication for diabetes and obesity. As of early 2023, Mounjaro has garnered approximately $1.2 billion in sales, reflecting rapid adoption.
Competitive Landscape
| Drug | Mechanism | Market Share (2022) | Key Features |
|---|---|---|---|
| Ozempic (semaglutide) | GLP-1 receptor agonist | 65% | Approved for diabetes and obesity |
| Wegovy (semaglutide) | Same | 20% | Focused on obesity |
| Mounjaro (tirzepatide) | Dual GIP/GLP-1 | 10% | Higher efficacy for weight loss and glycemic control |
Notes:
- Mounjaro’s dual mechanism offers superior weight loss and HbA1c reduction compared to monotherapy GLP-1s.
- Competition from Novo Nordisk's Ozempic and Wegovy intensifies.
Regulatory and Reimbursement Dynamics
- Both the FDA approval and evolving insurance coverage expand Mounjaro’s market access.
- In late 2022, several insurers began covering Mounjaro with prior authorization, limiting initial barriers.
- Pricing negotiations focus on value-based models due to its efficacy.
Price Analysis and Projections
Current Pricing
| Region | Official Wholesale Acquisition Cost (WAC) | Average Out-of-Pocket (OOP) | Notes |
|---|---|---|---|
| U.S. | $1,000 per 4-week dose | $50-$150 | Market price for branded product. |
| EU | Approximately €900 | €30-€130 | Variations across countries. |
Price Trajectory (2023-2030)
| Year | Price Range per Dose | Rationale/Drivers |
|---|---|---|
| 2023 | $1,000 | Stable initial pricing, reimbursement negotiations ongoing. |
| 2025 | $950-$1,050 | Competition and patent protections maintain prices. |
| 2027 | $900-$1,000 | Biosimilar entry unlikely before 2030; cost efficiencies and negotiations could lower prices slightly. |
| 2030 | $850-$950 | Potential biosimilar introduction, pricing pressure, value-based agreements. |
Pricing Factors
- Efficacy: Higher efficacy supports premium pricing.
- Market Competition: Monotherapies like Ozempic pricing influence Mounjaro’s price ceiling.
- Reimbursement Policies: Value-based arrangements may cap prices.
- Manufacturing Costs: Advances in production could reduce costs, impacting margins and pricing.
Sales and Revenue Forecasts
| Year | Estimated Sales (USD billions) | Assumptions |
|---|---|---|
| 2023 | $1.2 | Early adoption phase, limited by insurance coverage. |
| 2025 | $4.0-$6.0 | Broader access, expanded indications, increased prescriber confidence. |
| 2030 | $12.0 | Market penetration peaks, new indications, biosimilar entry may temper growth. |
All projections assume sustained efficacy and safety profiles, with no significant regulatory setbacks.
Pricing Strategies and Risks
- Premium Pricing: Supported by superior outcomes, but limited by payer negotiations.
- Value-Based Pricing: Contracting based on outcomes could limit revenue but improve payer acceptance.
- Market Risks: Biosimilar entry, competing therapies, safety concerns, or regulatory actions could impact prices and market share.
Regulatory Trends Impacting Pricing
- Tighter drug pricing regulations in key markets (U.S., EU).
- Incentives toward biosimilar adoption.
- Policy shifts encouraging value-based care favoring high-efficacy drugs like Mounjaro.
Key Takeaways
- Mounjaro is positioned to become a leading drug in the Type 2 diabetes and obesity markets with projected peak sales of $12 billion by 2030.
- Current prices in the U.S. are approximately $1,000 per dose, with expectations of slight reductions influenced by market competition and biosimilar developments.
- The competitive landscape favors drugs with similar or improved efficacy, with pricing likely to adapt based on reimbursement dynamics.
- Market growth relies heavily on insurance coverage expansion, patient access, and ongoing clinical data supporting comparative advantages.
- Future pricing will be shaped by biosimilar entries, regulatory adjustments, and agreements based on real-world outcomes.
FAQs
1. When is Mounjaro expected to reach peak market penetration?
By 2030, sales are projected to reach approximately $12 billion annually, backed by increased adoption and expanded indications.
2. How does Mounjaro compare in price to other GLP-1 receptor agonists?
It is roughly similar in price to top competitors, at about $1,000 per 4-week dose, but with superior efficacy which justifies premium pricing.
3. What factors could lead to a significant drop in Mounjaro’s price?
Biosimilar entry, increased competition, regulatory price controls, or negative clinical data would pressure prices downward.
4. How does insurance reimbursement impact Mounjaro’s pricing?
Reimbursement agreements and prior authorization processes influence patient out-of-pocket costs and market access, indirectly affecting pricing strategies.
5. What are the main risks to Mounjaro achieving projected sales figures?
Market competition, safety concerns, regulatory hurdles, or failure to expand insurance coverage could limit sales growth.
References
[1] Grand View Research. (2023). Global diabetes drugs market.
[2] MarketsandMarkets. (2022). Pharma market CAGR projections.
[3] Fortune Business Insights. (2023). Global obesity market size.
[4] Research and Markets. (2022). Obesity therapeutics market forecasts.
More… ↓
