Share This Page
Drug Price Trends for MORPHINE SULFATE
✉ Email this page to a colleague
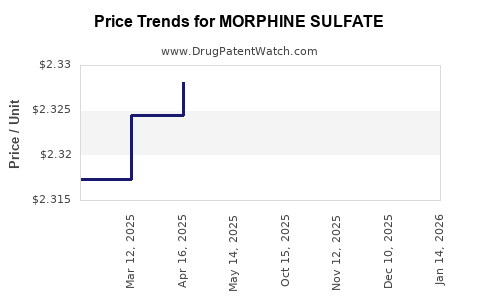
Average Pharmacy Cost for MORPHINE SULFATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MORPHINE SULFATE IR 30 MG TAB | 67877-0671-01 | 0.38026 | EACH | 2026-03-18 |
| MORPHINE SULFATE ER 10 MG CAP | 00832-0225-00 | 2.60312 | EACH | 2026-03-18 |
| MORPHINE SULFATE ER 20 MG CAP | 00832-0226-00 | 3.39600 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Morphine Sulfate Market Analysis and Price Projections
Morphine sulfate, a potent opioid analgesic, faces a complex market dynamic influenced by evolving regulatory landscapes, the ongoing opioid crisis, and advancements in pain management. While its therapeutic value remains significant, particularly in severe pain management and palliative care, market growth is constrained by concerns over addiction and diversion. Price projections for morphine sulfate are subject to fluctuations based on manufacturing costs, supply chain stability, and governmental pricing policies.
What is the Current Global Market Size and Projected Growth for Morphine Sulfate?
The global market for morphine sulfate is estimated to be valued at approximately USD 1.5 billion in 2023. Projections indicate a Compound Annual Growth Rate (CAGR) of 3.5% from 2024 to 2030. This growth is primarily driven by its continued use in oncology, post-operative care, and chronic pain management where alternative analgesics are insufficient.
The market is segmented by:
- Formulation:
- Injectable solutions (45% market share)
- Oral tablets and capsules (40% market share)
- Suppositories (10% market share)
- Others (e.g., extended-release formulations, transdermal patches) (5% market share)
- Application:
- Oncology pain
- Post-operative pain
- Chronic pain
- Palliative care
- Other
- Distribution Channel:
- Hospitals
- Retail pharmacies
- Online pharmacies
Geographically, North America currently holds the largest market share, accounting for approximately 35% of the global revenue, followed by Europe at 30%. The Asia-Pacific region is expected to exhibit the highest growth rate due to an increasing prevalence of chronic diseases and improving healthcare infrastructure.
What Factors Are Driving Demand for Morphine Sulfate?
Several key factors contribute to the sustained demand for morphine sulfate:
- Efficacy in Severe Pain: Morphine sulfate remains a gold standard for managing moderate to severe pain, particularly in settings like cancer treatment and intensive care units. Its efficacy is well-established and often unparalleled by other analgesics.
- Palliative Care Needs: The growing global elderly population and the increasing incidence of terminal illnesses necessitate robust pain management solutions, a role morphine sulfate effectively fills in palliative care.
- Post-Operative Pain Management: Following major surgical procedures, effective pain relief is critical for patient recovery. Morphine sulfate is frequently employed in hospital settings for this purpose.
- Availability and Cost-Effectiveness: Compared to newer, patented pain management drugs, generic morphine sulfate is generally more cost-effective, making it a vital option for healthcare systems with budget constraints.
What Challenges Impact the Morphine Sulfate Market?
The market for morphine sulfate is subject to significant challenges:
- Opioid Crisis and Regulatory Scrutiny: The widespread opioid crisis in many Western countries has led to intensified regulatory oversight, stricter prescribing guidelines, and increased monitoring for diversion and abuse. This impacts physician prescribing habits and patient access.
- Development of Alternative Analgesics: Research and development in pain management are yielding non-opioid alternatives, including NSAIDs, certain antidepressants, anticonvulsants, and novel non-addictive pain therapies. These alternatives, while not always as potent for severe pain, offer a safer profile.
- Addiction Potential and Side Effects: The inherent risk of addiction, dependence, respiratory depression, constipation, and nausea associated with morphine sulfate use necessitates careful patient selection, monitoring, and dose management.
- Supply Chain Vulnerabilities: Like many pharmaceutical products, morphine sulfate production is reliant on specific raw materials and manufacturing processes, making it susceptible to supply chain disruptions due to geopolitical events, natural disasters, or manufacturing issues.
- Stigma Associated with Opioid Use: The negative societal perception of opioid use can deter some patients and healthcare providers from considering morphine sulfate, even when medically indicated.
What Are the Key Trends in Morphine Sulfate Manufacturing and Supply?
Manufacturing and supply chain trends for morphine sulfate are evolving to address current market pressures:
- Focus on Extended-Release Formulations: To mitigate the risks of rapid dose escalation and improve patient compliance, there is a continued focus on developing and producing extended-release (ER) formulations of morphine sulfate. These formulations provide a more consistent level of analgesia over longer periods.
- Vertical Integration by Manufacturers: Some larger pharmaceutical companies are pursuing vertical integration, controlling the supply chain from raw material sourcing (e.g., poppy straw cultivation) to finished product manufacturing. This strategy aims to ensure supply security and cost control.
- Geographic Diversification of Production: To reduce reliance on single manufacturing locations and mitigate risks associated with regional supply chain disruptions, there is a trend towards diversifying production facilities across different geographical regions.
- Technological Advancements in Synthesis: While morphine sulfate is a well-established drug, ongoing research explores more efficient and environmentally friendly synthesis methods.
- Stringent Quality Control and Traceability: Regulatory bodies worldwide are mandating increasingly rigorous quality control measures and robust traceability systems to combat counterfeiting and ensure product integrity throughout the supply chain.
What Are the Projected Price Trends for Morphine Sulfate?
Projected price trends for morphine sulfate are influenced by a confluence of factors:
- Raw Material Costs: The price of poppy straw, the primary source of opium alkaloids, is subject to agricultural yields, weather conditions, and governmental cultivation policies in producing countries like Australia, India, and Turkey. Fluctuations in these costs directly impact the price of morphine sulfate.
- Manufacturing Expenses: Energy costs, labor, regulatory compliance, and the complex purification processes contribute to manufacturing expenses. As global energy prices remain volatile, these costs are likely to exert upward pressure on drug prices.
- Regulatory Compliance Costs: The increasing demands for quality control, security, and reporting associated with opioid production add significant costs for manufacturers. These are often passed on to consumers.
- Generic Competition: While morphine sulfate is largely a generic drug, the pricing of generic formulations is influenced by the number of manufacturers, production volumes, and the competitive landscape.
- Governmental Pricing Policies and Reimbursement Rates: In many countries, governments play a significant role in setting drug prices or influencing reimbursement rates. Changes in these policies, driven by healthcare budget considerations or efforts to control opioid expenditure, can directly impact the price of morphine sulfate.
- Supply Chain Stability: Disruptions in the supply chain, as noted previously, can lead to temporary price spikes due to scarcity. Conversely, a stable and robust supply chain can help moderate price increases.
Estimated Price Range Projections (USD per unit/mg):
- Injectable Solutions (per mg): Currently ranging from $0.10 - $0.25. Projected to increase by 4-6% annually, reaching $0.13 - $0.33 by 2030.
- Oral Tablets/Capsules (per mg): Currently ranging from $0.08 - $0.20. Projected to increase by 3-5% annually, reaching $0.10 - $0.26 by 2030.
- Extended-Release Formulations (per mg): These formulations command a premium due to their complex manufacturing and therapeutic advantages. Currently ranging from $0.20 - $0.40. Projected to increase by 5-7% annually, reaching $0.28 - $0.54 by 2030.
These projections are subject to significant variability based on the specific market region, manufacturer, and prevailing regulatory and economic conditions.
What are the Competitive Landscape and Key Players in the Morphine Sulfate Market?
The morphine sulfate market is characterized by a mix of large pharmaceutical companies and specialized generic manufacturers. The competitive landscape is influenced by production capacity, regulatory compliance, and distribution networks. Key players include:
- Pfizer Inc.: A major global pharmaceutical company with a significant presence in various therapeutic areas, including pain management.
- Fresenius Kabi AG: Specializes in generic injectable drugs, including opioid analgesics.
- Bausch Health Companies Inc.: Offers a portfolio of generic medications, including opioid formulations.
- Mallinckrodt Pharmaceuticals: Historically a significant producer of opioid analgesics, though facing ongoing legal and financial challenges.
- Grünenthal GmbH: A research-based pharmaceutical company active in pain management.
- Actavis plc (now part of AbbVie): A significant generic drug manufacturer.
- Teva Pharmaceutical Industries Ltd.: A leading global generic pharmaceutical company.
The market for morphine sulfate is mature, with limited opportunities for new entrants unless they can offer unique manufacturing efficiencies, robust supply chain capabilities, or specialized formulations that meet unmet clinical needs. The primary competitive battleground lies in cost-efficiency, reliability of supply, and adherence to stringent regulatory standards.
What are the Regulatory Considerations Impacting Morphine Sulfate?
Regulatory considerations are paramount and have a profound impact on the morphine sulfate market:
- Controlled Substance Regulations: Morphine sulfate is a Schedule II controlled substance in the United States and is similarly classified in many other countries. This classification imposes strict controls on manufacturing, distribution, prescribing, and dispensing.
- DEA Regulations (United States): The U.S. Drug Enforcement Administration (DEA) sets quotas for opioid production, monitors inventory, and enforces regulations to prevent diversion and abuse. Quota allocations can directly influence market supply.
- FDA Oversight (United States): The Food and Drug Administration (FDA) approves manufacturing processes, inspects facilities, and monitors product safety and efficacy.
- International Narcotics Control Board (INCB): The INCB monitors governments' implementation of international drug control treaties, including those related to opium alkaloids.
- Prescription Drug Monitoring Programs (PDMPs): Many jurisdictions mandate the use of PDMPs to track opioid prescriptions, aiming to identify patients at high risk for addiction or diversion.
- European Medicines Agency (EMA) and National Regulatory Bodies: In Europe, the EMA and individual national competent authorities regulate the marketing and sale of morphine sulfate.
- Impact of Opioid Settlement Agreements: Pharmaceutical companies involved in the opioid crisis have faced significant litigation and settlement agreements, which can affect their financial stability and focus on opioid production.
These regulatory frameworks are dynamic and can change based on public health concerns, leading to shifts in market access and operational requirements for manufacturers.
Key Takeaways
- The global morphine sulfate market is projected to grow at a CAGR of 3.5% through 2030, driven by its established efficacy in severe pain management, palliative care, and post-operative settings.
- Demand is sustained by its cost-effectiveness compared to newer analgesics, particularly in healthcare systems with budgetary constraints.
- Significant challenges include intensified regulatory scrutiny due to the opioid crisis, the development of non-opioid alternatives, and the inherent risks of addiction and side effects.
- Market growth is constrained by these regulatory pressures and the potential for increased adoption of alternative pain management strategies.
- Price projections indicate a modest but steady increase, influenced by raw material costs, manufacturing expenses, stringent regulatory compliance, and potential supply chain vulnerabilities.
- The competitive landscape is dominated by established generic manufacturers and large pharmaceutical companies, with a strong emphasis on supply chain security and regulatory adherence.
- Navigating the complex and evolving regulatory environment, including controlled substance regulations and international monitoring, is critical for all market participants.
Frequently Asked Questions
1. What is the primary raw material for morphine sulfate production?
The primary raw material for morphine sulfate production is opium poppy straw, which is processed to extract alkaloids including morphine.
2. How does the opioid crisis specifically impact morphine sulfate pricing?
The opioid crisis has led to increased regulatory burden and scrutiny on all opioid production, including morphine sulfate. Costs associated with enhanced security, tracking, and compliance with stricter prescribing guidelines can contribute to higher manufacturing and distribution expenses, indirectly impacting pricing.
3. Are there any significant new therapeutic applications emerging for morphine sulfate?
Currently, there are no major new therapeutic applications emerging for morphine sulfate. Its primary utility remains firmly established in the management of severe acute and chronic pain, and in palliative care.
4. What is the expected impact of price controls or governmental negotiations on morphine sulfate prices?
Governmental price controls or negotiations, particularly in major markets like the US and Europe, can exert downward pressure on morphine sulfate prices by limiting price increases or establishing reimbursement ceilings. However, increased manufacturing and regulatory compliance costs may counteract these pressures to some extent.
5. How does the availability of generic versus branded morphine sulfate formulations affect market dynamics?
Morphine sulfate is predominantly a generic product. The availability of multiple generic manufacturers leads to competitive pricing and generally keeps prices lower than they would be for a patented, branded product. Competition among generic producers focuses on production efficiency, supply reliability, and compliance with regulatory standards.
Citations
[1] Market Research Future. (2023). Morphine Sulfate Market Research Report Global Forecast till 2030. [2] Grand View Research. (2023). Morphine Market Size, Share & Trends Analysis Report By Formulation, By Application, By Region, And Segment Forecasts, 2024 - 2030. [3] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. [4] Food and Drug Administration. (n.d.). Opioid Medications. [5] International Narcotics Control Board. (n.d.). Annual Reports. [6] Various Pharmaceutical Industry Reports and Financial Statements (2023-2024).
More… ↓
