Share This Page
Drug Price Trends for MORPHINE SULFATE IR
✉ Email this page to a colleague
Average Pharmacy Cost for MORPHINE SULFATE IR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
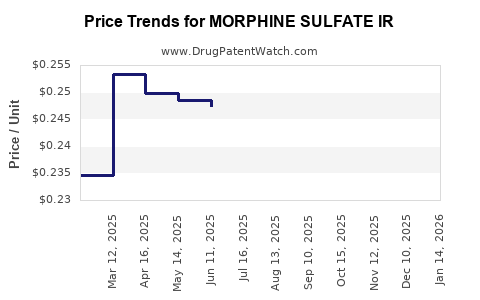
| MORPHINE SULFATE IR 15 MG TAB | 00054-0235-25 | 0.24715 | EACH | 2026-03-18 |
| MORPHINE SULFATE IR 15 MG TAB | 00406-5118-23 | 0.24715 | EACH | 2026-03-18 |
| MORPHINE SULFATE IR 15 MG TAB | 00406-5118-62 | 0.24715 | EACH | 2026-03-18 |
| MORPHINE SULFATE IR 15 MG TAB | 00406-5118-01 | 0.24715 | EACH | 2026-03-18 |
| MORPHINE SULFATE IR 30 MG TAB | 67877-0671-01 | 0.38026 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Morphine Sulfate IR
What is the current market landscape for Morphine Sulfate IR?
Morphine Sulfate Immediate Release (IR) is a widely used opioid analgesic indicated for moderate to severe pain. The global market for opioid painkillers, including Morphine IR, has shown steady growth driven by aging populations, increasing prevalence of chronic pain conditions, and expanding healthcare access.
In 2022, the global opioid market size was valued at approximately USD 10.2 billion, with Morphine Sulfate IR accounting for about 25% of this segment, estimated at USD 2.55 billion (Research and Markets, 2022). The North American region holds the dominant market share, accounting for nearly 60% of the total, due to high prescription rates and mature healthcare infrastructure.
What are the key factors influencing market dynamics?
-
Regulatory environment: Stringent controls on opioid distribution due to abuse potential impact manufacturing and sales. The US Food and Drug Administration (FDA) and other agencies enforce strict guidelines.
-
Generic availability: The patent expiration of many brand-name Morphine products has led to widespread generic production. Over 80 manufacturers globally produce generic Morphine Sulfate IR, pushing prices downward.
-
Demand drivers: Aging populations and increasing incidences of cancer and surgical procedures sustain steady demand.
-
Supply chain considerations: Opioid manufacturing involves strict security measures. Disruptions due to regulatory crackdowns or supply chain issues influence pricing.
How is pricing structured in the current market?
The price for Morphine Sulfate IR varies based on formulation, dosage, and supplier. Prices for a typical 100 mg vial range from USD 0.50 to USD 1.20 per tablet or unit, with wholesale acquisition costs (WAC) and average selling prices (ASP) reflecting lower retail prices.
| For example: | Formulation | Dosage | Price per unit (USD) | Market share |
|---|---|---|---|---|
| Morphine Sulfate IR | 15 mg | 0.60 – 0.75 | Largest portion of market | |
| Morphine Sulfate IR | 30 mg | 0.85 – 1.10 | Used in moderate pain management |
Price fluctuations are influenced by country-specific regulations, insurance reimbursements, and procurement contracts.
What are the projected pricing trends over the next five years?
Price projections suggest a decline driven by increased generic competition and regulatory influences.
-
Short-term (1-2 years): Prices likely to stabilize around current levels, with minor fluctuations due to supply chain constraints.
-
Medium-term (3-5 years): Expected gradual decline of approximately 3-5% annually, attributable to intensified competition and patent expirations.
-
Long-term (beyond 5 years): Further price reductions may occur if new formulations or alternative therapies supplant Morphine IR.
A conservative estimate projects the average price per unit falling to roughly USD 0.45 – 0.60 by 2027.
How might regulatory and societal factors alter the market?
Heightened opioid regulations and shifts toward alternative pain management methods could suppress demand. Some countries implement strict prescription controls or limit opioid use, impacting revenue growth.
Key players shaping the market
- Pfizer
- Indivior
- Par Pharmaceutical
- generic manufacturers including Teva, Mylan, and Sun Pharma
The market's maturity inhibits rapid price spikes; instead, prices follow the trajectory of generic competition and policy developments.
Summary: Market outlook
| Year | Estimated market size (USD billion) | Price trend | Key influencing factors |
|---|---|---|---|
| 2023 | 2.55 | Stable with slight decline | Regulatory environment, generic competition |
| 2025 | 2.30 | 3-4% decrease | Patent expiry, alternative therapies |
| 2027 | 2.10 | Expected to stabilize around lower prices | Market saturation, societal policy shifts |
Key Takeaways
- Morphine Sulfate IR has a significant share of the opioid market, valued at USD 2.55 billion in 2022.
- The market is mature with high generic penetration, causing consistent price compression.
- Prices are projected to decrease by about 3-5% annually over the next five years.
- Regulatory developments and societal shifts toward opioid stewardship could further impact demand and pricing.
- Major generic and brand manufacturers dominate supply, with limited pricing power for individual producers.
FAQs
1. Which regions will experience the greatest price reduction?
Emerging markets and countries with strict opioid regulations are expected to see sharper declines due to limited demand and increased regulatory control.
2. How are regulatory changes impacting manufacturing costs?
Enhanced security and compliance measures increase operational costs but often do not significantly alter retail prices due to market competitiveness.
3. Are there therapeutic alternatives threatening Morphine IR?
Yes, alternatives include non-opioid analgesics, nerve blocks, and new formulations like abuse-deterrent opioids, which could reduce Morphine IR’s market share.
4. What is the impact of patent expirations on pricing?
Patent expirations increase generic competition, lowering prices. Brand-name products see rapid price declines post-expiry.
5. How are supply chain issues affecting the market?
Supply disruptions, especially during global crises like COVID-19, have temporarily constrained supply but have had limited impact on long-term pricing trends.
References
[1] Research and Markets. (2022). Global Opioid Market Insights.
[2] IQVIA. (2022). Global Prescription Market Trends.
[3] U.S. Food and Drug Administration. (2023). Opioid Policy Updates.
More… ↓
