Share This Page
Drug Price Trends for MOMETASONE FUROATE
✉ Email this page to a colleague
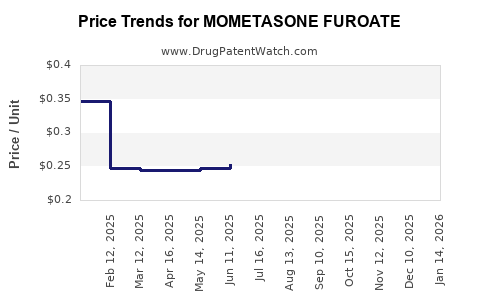
Average Pharmacy Cost for MOMETASONE FUROATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MOMETASONE FUROATE 0.1% CREAM | 00713-0634-37 | 0.27574 | GM | 2026-03-18 |
| MOMETASONE FUROATE 0.1% CREAM | 45802-0257-42 | 0.27574 | GM | 2026-03-18 |
| MOMETASONE FUROATE 0.1% OINT | 00713-0635-15 | 0.31622 | GM | 2026-03-18 |
| MOMETASONE FUROATE 0.1% CREAM | 45802-0257-35 | 0.40870 | GM | 2026-03-18 |
| MOMETASONE FUROATE 50 MCG SPRY | 65162-0891-29 | 1.63895 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for MOMETASONE FUROATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SINUVA SINUS IMPLANT | Intersect ENT, Inc. DBA Intersect | 10599-0003-01 | 1 | 1054.24 | 1054.24000 | EACH | 2023-09-01 - 2028-08-31 | Big4 |
| SINUVA SINUS IMPLANT | Intersect ENT, Inc. DBA Intersect | 10599-0003-01 | 1 | 1452.12 | 1452.12000 | EACH | 2023-09-01 - 2028-08-31 | FSS |
| SINUVA SINUS IMPLANT | Intersect ENT, Inc. DBA Intersect | 10599-0003-01 | 1 | 1103.57 | 1103.57000 | EACH | 2024-01-01 - 2028-08-31 | Big4 |
| SINUVA SINUS IMPLANT | Intersect ENT, Inc. DBA Intersect | 10599-0003-01 | 1 | 1452.12 | 1452.12000 | EACH | 2024-01-01 - 2028-08-31 | FSS |
| MOMETASONE FUROATE 0.1% LOTION,TOP | Golden State Medical Supply, Inc. | 51672-1305-04 | 60ML | 35.14 | 0.58567 | ML | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Mometasone Furoate: Market Dynamics and Price Forecast
Mometasone furoate, a potent corticosteroid, demonstrates significant market presence across respiratory and dermatological applications. The market is driven by increasing prevalence of allergic rhinitis and asthma, alongside a rising demand for topical treatments for inflammatory skin conditions. Patent expirations for originator products have opened avenues for generic competition, influencing pricing dynamics.
What are the Key Therapeutic Areas for Mometasone Furoate?
Mometasone furoate's primary therapeutic applications are in the management of inflammatory conditions. These are predominantly:
- Respiratory Diseases: Mometasone furoate is a cornerstone in treating allergic rhinitis and asthma. Intranasal formulations are widely prescribed for allergic rhinitis due to their efficacy in reducing nasal congestion, sneezing, and itching. Inhalation products are utilized as long-term control medications for persistent asthma, reducing airway inflammation and exacerbations.
- Dermatological Conditions: Topical mometasone furoate creams, ointments, and lotions are effective in treating a range of inflammatory skin disorders. This includes atopic dermatitis (eczema), psoriasis, contact dermatitis, and seborrheic dermatitis. Its anti-inflammatory, anti-pruritic, and vasoconstrictive properties alleviate symptoms such as redness, swelling, and itching.
What is the Current Market Landscape for Mometasone Furoate?
The global market for mometasone furoate is characterized by robust demand and a growing generic segment. The originator product, Nasonex (Schering-Plough, now Merck & Co.), for allergic rhinitis and asthma, and Elocon (Schering-Plough, now Merck & Co.) for dermatological conditions, have faced patent cliffs, leading to the introduction of numerous generic alternatives.
Key market drivers include:
- Rising Prevalence of Allergic Diseases: Global incidence of allergic rhinitis and asthma is increasing, particularly in developed and developing nations, due to urbanization, environmental factors, and lifestyle changes. The World Health Organization (WHO) estimates that allergic rhinitis affects 10-30% of the global population [1].
- Growing Demand for Topical Corticosteroids: The increasing incidence of chronic inflammatory skin conditions fuels the demand for effective topical treatments. Advancements in formulation technology, offering improved cosmetic acceptability and efficacy, further support market growth.
- Patent Expirations and Generic Entry: The expiry of key patents for branded mometasone furoate products has led to a significant influx of generic competitors. This has intensified market competition and driven down prices for both generic and, to some extent, branded products facing generic erosion.
- Healthcare Infrastructure Development: Expansion of healthcare access and infrastructure in emerging economies facilitates greater patient reach and prescription volumes for established treatments like mometasone furoate.
- Combination Therapies: The development and adoption of fixed-dose combination products, such as mometasone furoate with long-acting beta-agonists (LABAs) or long-acting muscarinic antagonists (LAMAs) for asthma, and with other topical agents for skin conditions, contribute to market value and complexity.
Market segmentation analysis reveals:
- By Formulation: Nasal sprays, inhalers, creams, ointments, lotions, and solutions represent distinct market segments, each catering to specific administration routes and indications.
- By Distribution Channel: Retail pharmacies, hospital pharmacies, and online pharmacies are key distribution channels. The growth of e-pharmacies is a notable trend.
- By Geography: North America and Europe currently dominate the market due to established healthcare systems and high disease prevalence. The Asia-Pacific region is projected to exhibit the fastest growth, driven by increasing healthcare expenditure and a large patient population.
What are the Key Patents and Expiry Dates Affecting Mometasone Furoate?
The patent landscape for mometasone furoate has been dynamic. While primary composition of matter patents have expired, secondary patents related to specific formulations, delivery devices, and manufacturing processes continue to influence market exclusivity and generic entry timelines.
- US Patent 5,037,830: This patent, covering the compound mometasone furoate, was a foundational patent for the originator products. It expired in the early 2010s.
- Formulation and Device Patents: Numerous patents have been filed and granted for specific aspects of mometasone furoate delivery. For example, patents related to nasal spray devices (e.g., to ensure consistent droplet size and delivery) or specific topical formulations (e.g., for enhanced skin penetration or stability) can provide extended protection for particular product iterations. The expiry dates for these secondary patents vary significantly, impacting the timing of generic launches for specific branded formulations.
- Orphan Drug Exclusivity: While mometasone furoate is not typically associated with orphan drug designation for its primary indications, any niche applications or specific pediatric formulations that may have received such designation would have their own exclusivity periods.
The general trend indicates that the core patents allowing for the basic production of mometasone furoate have expired in major markets like the US and Europe. This has paved the way for widespread generic competition. However, specific branded formulations with unique delivery systems or combination products may still benefit from residual patent protection or regulatory exclusivities.
What are the Price Trends and Projections for Mometasone Furoate?
Price trends for mometasone furoate are largely dictated by the balance between robust demand for its therapeutic benefits and intense price competition from generic manufacturers.
Current Price Trends:
- Branded Products: Prices for originator branded mometasone furoate products (e.g., Nasonex, Elocon) remain at a premium, though they have experienced price erosion since the advent of generics. This is often mitigated by market strategies, such as branded generics or lifecycle management.
- Generic Products: Generic mometasone furoate products are priced significantly lower than their branded counterparts. The average wholesale price (AWP) for a generic nasal spray bottle (e.g., 140 sprays) can range from $15 to $30 USD, while a generic topical cream (e.g., 15g tube) might range from $10 to $25 USD. These prices vary by manufacturer, strength, quantity, and geographic market.
- Price Competition: The generic market is highly competitive. With multiple manufacturers producing bioequivalent generics, prices are driven down to reflect manufacturing costs, market share objectives, and tender bids in institutional settings.
Price Projections:
- Continued Generic Dominance: The market will continue to be dominated by generic mometasone furoate. The average selling price (ASP) for generic products is expected to stabilize or experience marginal declines as manufacturers optimize production and face ongoing competition.
- Brand Price Moderation: Branded products will likely see continued, albeit slower, price moderation as they compete with generics. Strategies to maintain market share may include offering value-added services or focusing on specific patient populations.
- Impact of New Formulations/Combinations: The introduction of novel mometasone furoate formulations or fixed-dose combinations could command higher price points, at least initially, due to innovation and perceived added value. However, these too will eventually face generic challenges upon patent expiry.
- Geographic Price Variations: Price disparities between developed and emerging markets will persist. Developing markets are expected to see more significant price reductions as generic penetration increases and local manufacturing capabilities grow.
- Regulatory and Reimbursement Policies: Changes in healthcare policies, formulary decisions by insurance providers, and government pricing regulations will directly influence mometasone furoate pricing. Payers are likely to favor lower-cost generic options, reinforcing downward price pressure.
Overall, while the therapeutic utility of mometasone furoate remains high, its market pricing is characterized by a significant discount attributed to generic availability. Significant price increases are unlikely for widely established generic formulations.
What are the Key Manufacturers and Competitors in the Mometasone Furoate Market?
The mometasone furoate market is comprised of both originator companies and a large number of generic pharmaceutical manufacturers.
Originator Companies:
- Merck & Co. (formerly Schering-Plough): The primary originator of Nasonex and Elocon. While still a significant player with branded products, its market share is increasingly challenged by generics.
- AbbVie: Through its acquisition of Allergan, AbbVie has a presence in related therapeutic areas, though not directly with a primary mometasone furoate branded product.
Key Generic Manufacturers:
The generic landscape is highly fragmented and competitive, with numerous companies producing mometasone furoate APIs (Active Pharmaceutical Ingredients) and finished dosage forms. Some prominent generic players include:
- Teva Pharmaceutical Industries: A major global generic drug manufacturer with a broad portfolio.
- Mylan N.V. (now Viatris): Another large generic pharmaceutical company with a significant presence in respiratory and dermatological generics.
- Sun Pharmaceutical Industries: A leading Indian pharmaceutical company with extensive global reach in generics.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company known for its generic offerings.
- Lupin Ltd.: An Indian pharmaceutical company with a strong focus on generics.
- Aurobindo Pharma: An Indian vertically integrated pharmaceutical company with a significant API and finished dosage form business.
- Sandoz (Novartis): A major global player in generic and biosimilar medicines.
- Hikma Pharmaceuticals: A multinational pharmaceutical company focused on generic injectables and other dosage forms.
- Generic Manufacturers in China: Numerous Chinese API manufacturers supply mometasone furoate globally, contributing to price competitiveness.
The competitive intensity is high, with companies vying for market share through aggressive pricing, product differentiation (e.g., improved delivery devices, unique formulations), and strategic partnerships.
What are the Regulatory Considerations for Mometasone Furoate Products?
Regulatory pathways for mometasone furoate products are critical for market entry and ongoing compliance. These considerations are standard for corticosteroids and apply across different jurisdictions.
- Approval Pathways:
- New Drug Application (NDA) / Marketing Authorisation Application (MAA): For originator products, extensive clinical trial data demonstrating safety and efficacy is required.
- Abbreviated New Drug Application (ANDA) / Generic Marketing Authorisation Application: For generic products, applicants must demonstrate bioequivalence to the reference listed drug (RLD) and meet manufacturing quality standards. This significantly reduces the time and cost of development compared to originator drugs.
- Quality Standards: All manufacturing facilities must adhere to Good Manufacturing Practices (GMP) as mandated by regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others. This ensures the consistent quality, purity, and potency of the drug product.
- Labeling Requirements: Product labeling must accurately reflect the approved indications, dosage, contraindications, warnings, precautions, adverse reactions, and storage conditions. Specific warnings related to corticosteroid use, such as potential for systemic effects with prolonged high-dose use or skin atrophy with topical applications, are mandatory.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are required for all approved drug products. Manufacturers must have robust systems in place to monitor product safety and report any significant findings to regulatory authorities.
- Environmental Regulations: Manufacturing processes must comply with environmental regulations regarding waste disposal and emissions.
Specific regulatory hurdles can arise from:
- Complex Formulations: Novel or complex formulations (e.g., specific inhaler devices with unique engineering) may require additional data to demonstrate performance and reliability.
- API Sourcing: Ensuring a consistent and high-quality supply of mometasone furoate API from approved suppliers is a critical regulatory consideration.
- Bioequivalence Studies: Designing and executing bioequivalence studies that meet regulatory requirements can be challenging, particularly for inhaled or topical products where demonstration of equivalent systemic exposure or local effect is complex.
Key Takeaways
- Mometasone furoate is a well-established corticosteroid with significant markets in respiratory and dermatological indications, driven by high disease prevalence.
- Patent expirations for originator products have led to a highly competitive generic market, significantly influencing pricing.
- The market is projected to remain dominated by generics, with stable to declining price trends for these products.
- Originator pricing will likely moderate, while novel formulations or combinations may command initial premiums.
- Regulatory compliance, particularly regarding bioequivalence for generics and GMP standards, is paramount for market access.
FAQs
-
What is the primary reason for the significant price difference between branded and generic mometasone furoate? The primary reason is patent expiration. Once the patents protecting the originator drug expire, other pharmaceutical companies can manufacture and sell bioequivalent versions, leading to increased competition and lower prices.
-
Are there any mometasone furoate products currently protected by active patents that prevent generic competition? While core composition of matter patents have expired, secondary patents related to specific delivery devices, manufacturing processes, or novel formulations might still be in effect for certain branded products. These can delay or limit generic entry for those specific product variations.
-
How is the efficacy of generic mometasone furoate products typically assessed? Generic mometasone furoate products are assessed for bioequivalence to the reference listed drug. This means demonstrating that they are absorbed into the body at the same rate and to the same extent, or for inhaled/topical products, that they achieve equivalent local effects.
-
What is the projected impact of increasing healthcare expenditure in emerging markets on mometasone furoate pricing? Increased healthcare expenditure in emerging markets is likely to drive higher volumes of generic mometasone furoate. This, coupled with the expansion of local manufacturing capabilities, is expected to further contribute to competitive pricing and potentially lower average selling prices in these regions.
-
Beyond price, what factors differentiate mometasone furoate products in the current market? Differentiation can occur through various means, including the design and ease of use of delivery devices (e.g., nasal spray actuation, inhaler mechanism), formulation characteristics (e.g., scent, texture, absorption profile for topical products), and the availability of combination therapies that offer broader treatment benefits.
Citations
[1] World Health Organization. (n.d.). Allergic rhinitis. Retrieved from https://www.who.int/news-room/fact-sheets/detail/allergic-rhinitis
More… ↓
