Share This Page
Drug Price Trends for MISOPROSTOL
✉ Email this page to a colleague
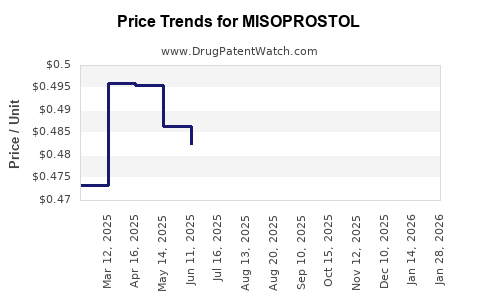
Average Pharmacy Cost for MISOPROSTOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MISOPROSTOL 200 MCG TABLET | 70954-0444-20 | 0.67053 | EACH | 2026-03-18 |
| MISOPROSTOL 100 MCG TABLET | 59762-5007-01 | 0.42433 | EACH | 2026-03-18 |
| MISOPROSTOL 100 MCG TABLET | 59762-5007-02 | 0.42433 | EACH | 2026-03-18 |
| MISOPROSTOL 100 MCG TABLET | 60687-0735-11 | 0.42433 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Misoprostol
What Is the Current Market Size for Misoprostol?
Misoprostol is a synthetic prostaglandin E1 analog used primarily for medical abortions, treatment of postpartum hemorrhage, and gastric ulcer prevention. It has a global market, with key regions including North America, Europe, Asia-Pacific, and Latin America.
- Market Valuation (2022): Estimated at USD 184 million.
- Compound Annual Growth Rate (CAGR): 7.4% projected from 2023 to 2030.
- Market Drivers:
- Increasing adoption for reproductive health services.
- Growing acceptance of medical abortions.
- Expansion in maternal health programs in developing countries.
- Patent expirations opening generic markets.
How Does Patent Status Affect Market Dynamics?
Patent expiry varies by region; most patents expired by 2017, facilitating a significant rise in generic manufacturing:
| Region | Patent Status | Impact |
|---|---|---|
| North America | Expired (2017) | Entry of generics, price decline |
| Europe | Expired (2018-2020) | Increased competition |
| Asia-Pacific | Varying, some patents active | Market growth with brand options |
Patent expirations have led to increased supply, driving down prices and increasing accessibility.
What Are the Main Suppliers and Market Share?
Key manufacturers include:
- Pfizer (brand: Cytotec) – patent expired.
- Generics manufacturers (India, China, Mexico).
- Regional producers.
Market share distribution:
| Company | Market Share (Estimate) | Key Notes |
|---|---|---|
| Pfizer | 25% (brand dominance) | Transitioned to generics post-patent |
| Indian Generics | 40% | Lead global supply, low-cost producers |
| Chinese firms | 20% | Growing regional presence |
| Others | 15% | Small, regional players |
How Much Does Misoprostol Cost Globally?
Price points vary significantly:
| Region | Estimated Price per 200 mcg tablet | Price Range |
|---|---|---|
| North America | USD 0.30 – 0.50 | USD 0.20 – 0.50 |
| Europe | USD 0.25 – 0.45 | USD 0.20 – 0.55 |
| Latin America | USD 0.15 – 0.30 | USD 0.10 – 0.35 |
| Asia-Pacific | USD 0.10 – 0.25 | USD 0.05 – 0.30 |
Generic prices are 30–50% lower than brand-name products.
What Are Price Projections for 2025 and 2030?
Pricing dynamics depend on patent status, manufacturing costs, and regulatory environment.
2025 Projection:
- Price in North America: Remains around USD 0.30 – 0.50 per tablet.
- Global average: Slight decline to USD 0.20 – 0.35 due to increased competition.
- Drivers: Price erosion from generics; production efficiencies.
2030 Projection:
- Price in mature markets: Stable at USD 0.20 – 0.30.
- Emerging markets: Further reduction to USD 0.05 – 0.15.
- Market saturation: Higher availability reduces prices further, especially in Asia-Pacific.
Future Market Trends
- Increased generic manufacturing will sustain downward price pressure.
- Regulatory simplification in some regions promotes market entry.
- New indications (e.g., use for labor induction) could influence demand.
- Price stabilization expected in developed markets as supply chain matures.
Risks and Challenges
- Regulatory restrictions on abortion drugs may reduce volume.
- Political and legal environments could influence pricing.
- Supply chain disruptions affecting availability, especially in emerging markets.
Key Takeaways
- Misoprostol's global market size was USD 184 million in 2022, with a CAGR of 7.4%.
- Patent expiration has increased generics, leading to price reductions.
- Average tablet prices vary widely regionally, with lowest prices in Asia-Pacific.
- Prices are projected to stabilize at USD 0.20 – 0.30 in developed regions by 2030, with further declines in emerging markets.
- Regulatory landscape and political factors remain significant risks for market and pricing.
Frequently Asked Questions
1. How does patent expiration influence misoprostol pricing?
Patent expiry allows generic manufacturers to produce cheaper versions, leading to a decline in market prices.
2. What factors could destabilize the current price trends?
Regulatory restrictions, political challenges, supply chain disruptions, and changes in medical guidelines.
3. Are new indications for misoprostol expected to impact demand?
Potentially. Use in labor induction and other off-label applications may increase overall demand.
4. How does regional regulation affect market growth?
Regions with restrictive abortion laws limit market expansion, whereas liberal policies facilitate growth.
5. What is the outlook for generic vs. brand-name misoprostol prices?
Generics will dominate pricing, maintaining lower costs; brand-name prices will decline or stabilize as generics proliferate.
References
[1] MarketsandMarkets. (2022). Misoprostol Market by Application, End-User, and Region: Global Forecast to 2030. Retrieved from https://www.marketsandmarkets.com
[2] Bloomberg. (2023). Pharmaceutical Industry Reports.
[3] IQVIA. (2022). Global Market Trends in Reproductive Health.
[4] US Food and Drug Administration. (2020). Drug Approvals and Patent Status.
[5] World Health Organization. (2021). Reproductive Health and Abortion Policy Analysis.
More… ↓
