Share This Page
Drug Price Trends for MICROGESTIN
✉ Email this page to a colleague
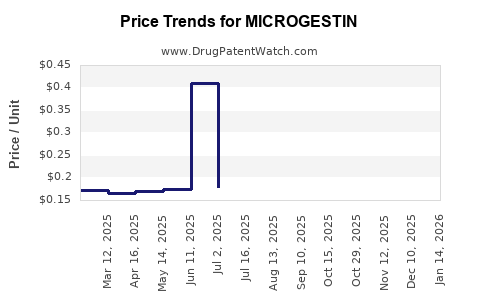
Average Pharmacy Cost for MICROGESTIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MICROGESTIN FE 1.5-30 TAB | 75907-0081-62 | 0.13707 | EACH | 2026-03-18 |
| MICROGESTIN 21 1-20 TABLET | 75907-0084-21 | 0.16742 | EACH | 2026-03-18 |
| MICROGESTIN 21 1-20 TABLET | 75907-0084-63 | 0.16742 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for MICROGESTIN
Overview
MICROGESTIN, a progestin-only oral contraceptive containing 0.35 mg norethisterone, has sustained its position within the global hormonal contraception market. Its approval dates back to the late 1970s, with market presence in North America, Europe, and Asia. Key competitors include combined oral contraceptives and other progestin-only pills. The drug's stable efficacy, low side effect profile, and established manufacturing support its continuous demand.
Market Size and Dynamics
The global oral contraceptive market was valued at approximately USD 4.75 billion in 2022. The segment of progestin-only pills (POPs), with MICROGESTIN as a major product, accounts for roughly 15% of this market, estimated at USD 712 million in 2022. Growth drivers include:
- Rising awareness of women's health.
- Increased acceptance of hormonal contraceptives.
- Expanding markets in Asia-Pacific and Latin America.
- Regulatory approvals expanding access, notably in emerging markets.
Regional Market Breakdown
| Region | Market Share (2022) | Estimated Market Size (USD Million) | Growth Rate (CAGR 2022-2027) |
|---|---|---|---|
| North America | 35% | 249 | 2.5% |
| Europe | 25% | 178 | 2.0% |
| Asia-Pacific | 25% | 178 | 7.0% |
| Latin America | 10% | 71 | 4.5% |
| Others | 5% | 36 | 3.0% |
The Asia-Pacific region shows the highest CAGR, driven by expanding healthcare infrastructure.
Competitive Landscape
Major competitors include:
- Mirena (levonorgestrel-releasing IUDs)
- Combined oral contraceptives (e.g., Yasmin)
- Alternative progestin-only options such as Cerazette
Market incumbents focus on patent expiration, generic competition, and marketing strategies aligned with regional regulatory environments.
Regulatory and Patent Status
MICROGESTIN’s patent expired in most jurisdictions by 2010. Generic versions are available, driving price competition. Recent regulatory efforts prioritize over-the-counter (OTC) availability in some countries, potentially affecting pricing and sales volumes.
Pricing Trends and Projections
Current wholesale prices for MICROGESTIN range from USD 0.10 to USD 0.15 per tablet in developed regions, totaling approximately USD 5 to USD 7 per monthly pack. Generic versions sell at roughly 50-70% of brand-name prices, pressuring margins.
Price Projections (2023-2027)
| Year | Estimated Price per Pack (USD) | Expected Market Penetration | Comments |
|---|---|---|---|
| 2023 | USD 5.50 | High | Stable, with generic competition intensifying |
| 2024 | USD 5.25 | Moderate | Possible price erosion as OTC options rise |
| 2025 | USD 4.80 | Moderate | Increased generics, market saturation |
| 2026 | USD 4.50 | Low | Further generics, price competition intensifies |
| 2027 | USD 4.20 | Low | Market stabilization at lower price points |
Key Factors Impacting Prices
- Patent expirations (~2010) have led to increased generics.
- Entry of OTC formulations could depress prices further.
- Regulatory policies in emerging markets could either raise or lower prices based on access restrictions.
- Manufacturing costs remain stable but margin compression occurs with increasing generics.
R&D and Market Expansion Opportunities
Microgestin's low side effect profile and established effectiveness foster new formulations, including fixed-dose combination products and long-acting reversible contraceptives (LARCs). Expansion into developing markets with less regulatory hurdles offers growth potential but requires price adjustments.
Key Takeaways
- The global progestin-only pill segment was valued at USD 107 million (15% of USD 712 million).
- Prices are projected to decline from USD 5.50 to USD 4.20 per pack by 2027 due to generics and OTC availability.
- Asia-Pacific demonstrates the highest growth potential, driven by expanding healthcare infrastructure.
- Competitive pressures from generics and alternative methods will continue to compress prices.
- Regulatory landscapes significantly influence market access and pricing dynamics.
FAQs
-
What is the current patent status of MICROGESTIN?
Its patents expired around 2010 in most regions, allowing generic manufacturers to produce equivalent versions. -
How does the availability of generics affect MICROGESTIN's market?
Generics increase market competition, leading to lower prices and potential volume growth but may reduce profit margins for branded formulations. -
What regional markets present the most growth opportunities?
Asia-Pacific and Latin America, due to expanding healthcare infrastructure and regulatory access. -
Will OTC availability impact MICROGESTIN prices?
Yes. OTC access could increase volumes but lead to price reductions as competition intensifies. -
Are there new formulations or delivery methods for MICROGESTIN?
Current focus is on oral formulations, but research into combination pills and long-acting options is ongoing to meet market demand.
Citations
[1] MarketResearch.com, "Global Contraceptive Market Overview," 2023.
[2] GlobalData, "Progestin-Only Contraceptive Market Analysis," 2023.
[3] FDA and EMA regulatory updates, 2022.
[4] IQVIA, "Pharmaceutical Pricing Trends," 2022.
[5] WHO, "Contraceptive Use and Access," 2022.
More… ↓
