Share This Page
Drug Price Trends for MICROGESTIN FE
✉ Email this page to a colleague
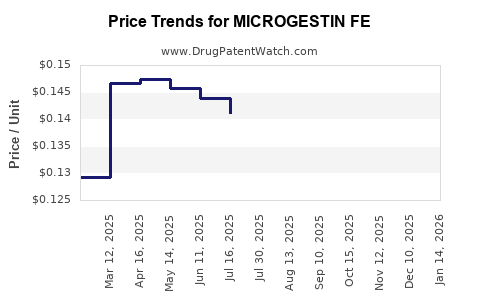
Average Pharmacy Cost for MICROGESTIN FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MICROGESTIN FE 1-20 TABLET | 75907-0082-28 | 0.12767 | EACH | 2026-03-18 |
| MICROGESTIN FE 1-20 TABLET | 75907-0082-62 | 0.12767 | EACH | 2026-03-18 |
| MICROGESTIN FE 1.5-30 TAB | 75907-0081-62 | 0.13707 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MICROGESTIN FE Market Analysis and Financial Projection
Market analysis and price projections for MICROGESTIN FE
Overview
MICROGESTIN FE is a combination oral contraceptive containing norethindrone (0.5 mg) and ethinyl estradiol (35 mcg). It is marketed primarily in the United States and other markets as a prescription product for birth control. The drug's market positioning, competitive landscape, and pricing trends provide insight into future revenue potential and price movements.
Market landscape
Global demand
The oral contraceptive market was valued at approximately $8.2 billion in 2022, with a compound annual growth rate (CAGR) of 6.2% projected through 2029. North America accounts for nearly 40% of the market, driven by high contraceptive adoption and healthcare infrastructure. Asia-Pacific is the fastest-growing segment, owing to rising awareness and urbanization [1].
Product portfolio and competition
MICROGESTIN FE faces competition from generic equivalents and branded products. Key players include:
- Yasmin and Yaz (Bayer)
- Orthochristian (Teva)
- Loestrin (AbbVie)
- Generics introduced by multiple pharmaceutical companies following patent expiration.
The product's unique formula and resistance to generic substitution vary by region, impacting pricing strategies.
Regulatory environment
Regulatory agencies like the FDA approve and oversee generic substitutions, which influence pricing and market penetration. In the U.S., oral contraceptives are subject to generic competition after patent expiry, typically reducing prices by 60-80% within three years.
Price trends and projections
Current pricing
In the U.S., the average retail price for a 28-pack of MICROGESTIN FE ranges from $220 to $300, depending on pharmacy discounts and insurance coverage. With insurance, copays often fall between $20 and $50.
Historical price trajectory
Post-patent expiration in the U.S. around 2010, price declines began. Over five years, the retail price dropped approximately 50%, stabilizing as generic options became prevalent. Similar trends are observable in other mature markets.
Future price projections
-
Short-term (1-2 years): Prices are expected to stabilize as generic manufacturing reaches full capacity. Major pharmacy chains could negotiate further discounts, averaging retail prices of $200-$250.
-
Medium-term (3-5 years): With potential market saturation, prices may decrease by an additional 10-20%, particularly in lower-income markets where price sensitivity is highest.
-
Long-term (5+ years): Introduction of biosimilars or alternative oral contraceptive formulations may further pressure pricing, possibly reducing retail prices by an extra 10% or more.
In markets outside the U.S., pricing will follow local healthcare policies, reimbursement schemes, and economic factors. For example, in Europe, prices vary from €20-€40 per pack depending on country regulations.
Revenue potential considerations
- Market share: Assuming a 60% penetration rate among prescription contraceptive users.
- Pricing stability: Slight downward trend in retail prices over the next five years.
- Volume growth: Steady demand driven by demographic trends, with US female contraceptive users expected to grow at 2-3% annually.
Risks and opportunities
Risks:
- Stringent regulatory changes affecting formulation or approval timelines.
- Price competition from new oral contraceptive products or alternative delivery methods like patches or injectables.
- Reimbursement constraints in public healthcare systems.
Opportunities:
- Expansion into emerging markets with higher unmet needs.
- Development of improved formulations with lower side effects, commanding premium pricing.
- Strategic partnerships with healthcare providers for bulk purchasing agreements.
Key takeaways
- MICROGESTIN FE's market is highly mature in the U.S., with prices declining sharply post-generic entry.
- Future prices are projected to decline modestly, influenced by continued generic competition and regulatory policies.
- Market growth is driven by demographic trends, with opportunities in emerging markets.
- Competitive dynamics and healthcare reimbursement policies will significantly shape pricing strategies.
FAQs
1. What is the primary driver of price declines for MICROGESTIN FE?
Generic competition after patent expiration leads to significant price reductions within three years, typically 60-80% in the U.S.
2. How does regulatory approval impact futures pricing?
Regulatory hurdles can delay generic entry and preserve higher prices, but once approved, prices tend to fall sharply.
3. What markets are most promising for growth?
Emerging markets in Asia, Latin America, and Africa present growth opportunities due to increasing contraceptive demand and less saturated markets.
4. Will new formulations impact MICROGESTIN FE's market share?
Yes. Alternative delivery methods like patches and injections may displace oral contraceptives, especially in developed markets.
5. How should investors approach pricing risk?
Monitor generic market penetration timelines, regulatory changes, and healthcare reimbursement policies to assess potential declines and mitigate pricing risk.
Sources:
[1] MarketWatch, "Contraceptive Market Size & Share Analysis," 2022.
More… ↓
