Drug Price Trends for MICARDIS
✉ Email this page to a colleague
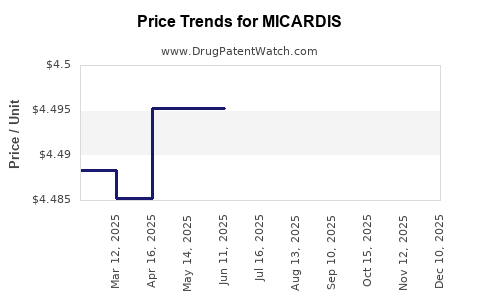
Average Pharmacy Cost for MICARDIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MICARDIS HCT 80-12.5 MG TABLET | 00597-0044-37 | 4.47615 | EACH | 2026-04-22 |
| MICARDIS HCT 80-12.5 MG TABLET | 00597-0044-37 | 4.47761 | EACH | 2026-03-18 |
| MICARDIS HCT 80-12.5 MG TABLET | 00597-0044-37 | 4.47361 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for MICARDIS
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| MICARDIS HCT 40/12.5MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0043-37 | 30 | 139.55 | 4.65167 | EACH | 2024-01-01 - 2027-09-14 | FSS |
| MICARDIS 40MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0040-37 | 30 | 94.58 | 3.15267 | EACH | 2024-01-01 - 2027-09-14 | Big4 |
| MICARDIS HCT 80/25MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0042-37 | 30 | 139.55 | 4.65167 | EACH | 2024-01-01 - 2027-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
