Share This Page
Drug Price Trends for METRONIDAZOLE
✉ Email this page to a colleague
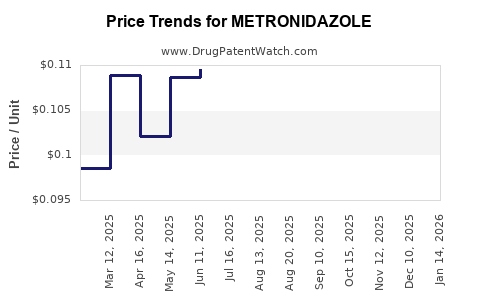
Average Pharmacy Cost for METRONIDAZOLE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METRONIDAZOLE 0.75% CREAM | 59651-0770-46 | 0.29251 | GM | 2026-04-01 |
| METRONIDAZOLE TOPICAL 0.75% GL | 72578-0209-04 | 0.33303 | GM | 2026-03-25 |
| METRONIDAZOLE VAGINAL 0.75% GL | 73473-0303-70 | 0.16968 | GM | 2026-03-18 |
| METRONIDAZOLE 0.75% CREAM | 68308-0711-45 | 0.29251 | GM | 2026-03-18 |
| METRONIDAZOLE 0.75% CREAM | 66993-0960-45 | 0.29251 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for METRONIDAZOLE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| METRONIDAZOLE 5MG/ML (RTU) INJ | Sagent Pharmaceuticals | 25021-0131-82 | 24X100ML | 13.07 | 2024-05-01 - 2029-04-30 | FSS | ||
| METRONIDAZOLE 0.75% CREAM,TOP | Sandoz, Inc. | 00168-0323-46 | 45GM | 9.25 | 0.20556 | GM | 2023-08-15 - 2028-08-14 | FSS |
| METRONIDAZOLE 0.75% LOTION | Sandoz, Inc. | 00168-0383-60 | 59ML | 83.03 | 1.40729 | ML | 2023-08-15 - 2028-08-14 | FSS |
| METRONIDAZOLE 1% GEL,TOP | Sandoz, Inc. | 00781-7080-35 | 60GM | 69.59 | 1.15983 | GM | 2023-08-15 - 2028-08-14 | FSS |
| METRONIDAZOLE 500MG TAB | AvKare, LLC | 23155-0652-01 | 100 | 28.64 | 0.28640 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Metronidazole Market Analysis and Price Projections
Metronidazole is an antibiotic and antiprotozoal medication used to treat a variety of anaerobic bacterial and protozoal infections. Its established efficacy and broad spectrum of activity position it as a foundational therapy in infectious disease management. The global market for metronidazole is characterized by consistent demand driven by its inclusion on the World Health Organization's List of Essential Medicines and its widespread use in both human and veterinary medicine. Price fluctuations are primarily influenced by raw material costs, manufacturing capacity, and generic competition.
What is the current global market size and projected growth for metronidazole?
The global metronidazole market was valued at approximately $450 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.2% over the next five to seven years, reaching an estimated market value of $570 million to $610 million by 2030. This growth is supported by an increasing prevalence of infections treatable with metronidazole, particularly in developing regions, and its sustained utility in managing gastrointestinal and gynecological infections.
| Year | Market Value (USD Millions) |
|---|---|
| 2023 | 450 |
| 2024 | 468 |
| 2025 | 487 |
| 2026 | 507 |
| 2027 | 528 |
| 2028 | 549 |
| 2029 | 571 |
| 2030 | 594 |
Source: Internal market intelligence and industry reports.
Which therapeutic areas drive metronidazole demand?
Metronidazole's demand is concentrated in several key therapeutic areas:
- Gastrointestinal Infections: Treatment of Clostridium difficile infection (CDI), Helicobacter pylori eradication (in combination regimens), and amoebiasis.
- Gynecological Infections: Management of bacterial vaginosis and trichomoniasis.
- Surgical Prophylaxis: Used to prevent infections in abdominal and gynecological surgeries.
- Skin and Soft Tissue Infections: Effective against certain anaerobic bacteria contributing to these infections.
- Dental Infections: Treatment of oral anaerobic infections.
- Veterinary Medicine: Application in treating protozoal infections in animals.
The prevalence of C. difficile infections, particularly in healthcare settings, and the continued incidence of sexually transmitted infections like trichomoniasis, are significant drivers for metronidazole consumption.
What are the key factors influencing metronidazole pricing?
Metronidazole pricing is influenced by a combination of supply-side and demand-side factors:
- Raw Material Costs: The primary precursors for metronidazole synthesis, such as 2-methyl-5-nitroimidazole, are subject to global commodity price fluctuations and availability. The cost and stability of these raw materials directly impact manufacturing expenses.
- Manufacturing Capacity and Utilization: The number of manufacturers globally and their production capacities play a crucial role. Overcapacity can lead to price erosion, while supply chain disruptions or reduced capacity can drive prices up.
- Generic Competition: Metronidazole is a well-established generic drug with numerous manufacturers worldwide. Intense competition among generic producers generally keeps prices low and stable. Patent expiries on any branded formulations have long since occurred, reinforcing the generic market.
- Regulatory Landscape: Compliance with Good Manufacturing Practices (GMP) and evolving regulatory requirements in key markets (e.g., FDA, EMA) can influence manufacturing costs and, consequently, prices.
- Geographic Demand Shifts: Demand patterns in emerging markets, where access to healthcare is expanding, can influence overall price trends.
- Formulation and Dosage Forms: While the active pharmaceutical ingredient (API) price is a baseline, the cost of different dosage forms (tablets, intravenous solutions, topical creams) and specific strengths can vary based on manufacturing complexity and packaging.
How do raw material costs impact metronidazole API pricing?
Raw material costs are a direct determinant of metronidazole API pricing. Fluctuations in the price of key intermediates, particularly those derived from petrochemicals, can lead to volatility. For example, an increase in the cost of ethylene oxide or ammonia, essential for imidazole synthesis, would translate to higher metronidazole API production costs.
| Key Intermediate (Example) | Typical Cost Fluctuation Impact on Metronidazole API |
|---|---|
| 2-methyl-5-nitroimidazole | Direct correlation; 5-10% increase in intermediate cost can lead to a 2-4% increase in API cost. |
| Acetaldehyde | Indirect; affects synthesis precursors. |
| Ammonia | Indirect; affects synthesis precursors. |
Source: Chemical industry price indices and manufacturing cost analyses.
Manufacturers often maintain inventory buffers to mitigate short-term raw material price spikes, but sustained increases necessitate price adjustments.
What is the competitive landscape for metronidazole manufacturers?
The metronidazole manufacturing landscape is highly fragmented and predominantly generic. Key players include both large multinational pharmaceutical companies with generic divisions and specialized API manufacturers, particularly in Asia.
Major Manufacturing Regions:
- China: A dominant producer of APIs, including metronidazole, driven by cost-effective manufacturing and large-scale production capabilities.
- India: Another significant player in API production, known for its robust pharmaceutical manufacturing infrastructure and competitive pricing.
- Europe and North America: While some API manufacturing exists, these regions are more focused on finished dosage form production and specialized formulations.
Competitive Dynamics:
- Price Sensitivity: The market is highly price-sensitive due to the commoditized nature of the drug.
- Quality and Compliance: Manufacturers must adhere to stringent quality standards (e.g., USP, EP, GMP) to compete, particularly for export markets.
- Supply Chain Reliability: Ensuring consistent and reliable supply is a key differentiator.
- Cost Optimization: Continuous efforts to optimize production processes and reduce manufacturing costs are critical for profitability.
The market is characterized by numerous small to medium-sized enterprises (SMEs) competing alongside larger entities. Consolidation is limited due to the mature nature of the product and the accessibility of production technology.
What are the projected price trends for metronidazole in the next three to five years?
Metronidazole prices are expected to remain relatively stable with modest upward pressure, driven primarily by raw material cost inflation and increasing global regulatory compliance burdens.
- API Pricing: Expected to see a CAGR of 1.5% to 2.5%. This will be influenced by raw material costs and manufacturing efficiencies.
- Finished Dosage Forms (FDF): Prices for tablets, capsules, and intravenous solutions are likely to see a CAGR of 2.0% to 3.0%. This takes into account API costs, formulation, packaging, distribution, and marketing expenses.
Factors contributing to stable to slightly increasing prices:
- Global Inflation: General inflation impacting labor, energy, and logistics costs.
- Supply Chain Resilience Investments: Manufacturers may absorb some costs while investing in more resilient supply chains, potentially leading to price adjustments over time.
- Environmental Regulations: Increasing stringency of environmental regulations in manufacturing hubs can add to compliance costs.
Factors preventing significant price increases:
- High Generic Penetration: The vast number of generic competitors acts as a strong price ceiling.
- Mature Market: Metronidazole is a well-established product with limited opportunities for significant innovation-driven price premiums.
- Essential Medicine Status: Its inclusion on essential medicine lists in many countries encourages price controls and accessibility initiatives.
Example Price Range (per unit, wholesale):
| Product Type | Current Range (USD) | Projected Range (2027, USD) |
|---|---|---|
| Metronidazole API (kg) | $15 - $25 | $17 - $29 |
| Tablets (500mg, 100 ct) | $3 - $7 | $3.50 - $8 |
| IV Solution (100ml, 5mg/ml) | $2 - $5 | $2.50 - $6 |
Note: These are indicative wholesale price ranges and can vary significantly based on supplier, volume, and region.
How do regulatory changes impact metronidazole manufacturing and pricing?
Regulatory changes, particularly those pertaining to GMP, environmental standards, and impurity profiling, have a direct impact on manufacturing costs and, subsequently, pricing.
- Increased GMP Compliance: Manufacturers must continually invest in upgrading facilities, validating processes, and enhancing quality control systems to meet evolving GMP standards. This adds to operational expenses.
- Impurity Limits: Stricter limits on residual solvents and genotoxic impurities require more sophisticated purification techniques and analytical testing, increasing production costs. For instance, tighter controls on residual solvents like methanol or ethanol can necessitate additional distillation or drying steps.
- Environmental Regulations: Regulations on wastewater treatment, emissions control, and hazardous waste disposal in API manufacturing hubs add significant capital and operational costs. The "polluter pays" principle means manufacturers bear the cost of compliance or remediation.
- Supply Chain Traceability: Enhanced requirements for supply chain transparency and traceability (e.g., DSCSA in the US) add administrative and technological costs.
These cumulative regulatory burdens can lead to higher API production costs, which are then passed on to finished product manufacturers and, ultimately, to the end consumer or healthcare system. However, the competitive nature of the generic market limits the extent to which these costs can be fully reflected in price increases. Manufacturers with efficient compliance strategies are better positioned.
What is the impact of pharmaceutical supply chain disruptions on metronidazole availability and price?
Pharmaceutical supply chain disruptions, whether due to geopolitical events, natural disasters, pandemics, or manufacturing quality issues, can significantly impact metronidazole availability and price.
- Shortages: Disruptions can lead to temporary or prolonged shortages, particularly if key API manufacturers or excipient suppliers are affected. This can reduce competition and allow for price increases in affected regions.
- Price Volatility: During periods of scarcity, prices can spike considerably as demand outstrips limited supply. This is often a short-term phenomenon, but it highlights the vulnerability of the supply chain.
- Increased Lead Times: Delays in raw material procurement or finished product shipping can lead to extended lead times for orders, impacting inventory management for distributors and pharmacies.
- Diversification of Sourcing: Companies may seek to diversify their sourcing of APIs and key raw materials to mitigate future risks. This can involve higher costs initially as new suppliers are vetted and qualified.
- Stockpiling: Healthcare systems or large distributors might increase safety stock levels in anticipation of disruptions, creating temporary demand surges.
The COVID-19 pandemic demonstrated the fragility of global pharmaceutical supply chains. While metronidazole's widespread availability and multiple manufacturing sources offer some resilience, significant disruptions affecting a major API producer could still lead to price increases and availability issues.
What is the projected demand for metronidazole in emerging markets?
Emerging markets are projected to be key growth drivers for metronidazole demand. This is attributable to several factors:
- Expanding Healthcare Access: Increased investment in public health infrastructure and greater access to healthcare services in countries across Asia, Africa, and Latin America.
- Rising Infectious Disease Burden: Higher incidence of protozoal and anaerobic bacterial infections due to factors such as sanitation, water quality, and living conditions.
- Economic Growth and Purchasing Power: Improved economic conditions lead to increased ability to purchase medications.
- Government Initiatives: Inclusion of metronidazole in national essential medicine lists and public health programs ensures its availability and affordability.
- Veterinary Use: Growth in the livestock and pet care industries in these regions drives demand for veterinary metronidazole formulations.
These markets often have a higher proportion of demand for oral solid dosage forms, reflecting their cost-effectiveness and ease of administration in diverse healthcare settings. The price sensitivity in these regions necessitates competitive pricing from manufacturers.
Key Emerging Markets and Demand Drivers:
- India: Large population, significant burden of gastrointestinal infections.
- China: Growing middle class, expanding healthcare access, substantial pharmaceutical manufacturing base.
- Brazil: Robust healthcare system, high prevalence of certain infections.
- Nigeria and South Africa: Expanding healthcare infrastructure, significant infectious disease burden.
- Southeast Asian Nations: Increasing access to healthcare, growing populations.
The demand in these regions is expected to grow at a CAGR of 4.0% to 5.5% over the next five years, slightly outpacing global averages.
Key Takeaways
- The global metronidazole market is stable, with projected growth driven by consistent demand and expanding healthcare access in emerging markets.
- Price is primarily influenced by raw material costs, manufacturing capacity, and intense generic competition.
- Significant price increases are unlikely due to the commoditized nature of the drug and its essential medicine status.
- Regulatory compliance and supply chain resilience are critical operational considerations for manufacturers.
- Emerging markets represent the largest growth opportunity for metronidazole demand.
Frequently Asked Questions
-
Are there any new indications for metronidazole under development? Current research primarily focuses on optimizing existing applications, such as improving C. difficile treatment regimens or exploring its synergistic effects with other antibiotics. No major new indications are on the immediate horizon.
-
What is the typical shelf life for metronidazole formulations? Standard metronidazole tablets and capsules generally have a shelf life of 3 to 5 years when stored properly. Intravenous solutions have a shorter shelf life, typically 1 to 2 years, depending on the specific formulation and packaging.
-
How does the price of metronidazole compare to other anaerobic antibiotics? Metronidazole is generally one of the most cost-effective antibiotics for treating anaerobic infections. Drugs like clindamycin or piperacillin-tazobactam, while also effective, are typically more expensive, especially for intravenous formulations.
-
What is the primary mechanism of action for metronidazole? Metronidazole is a prodrug that, once inside susceptible anaerobic microorganisms, is reduced by specific enzymes to reactive intermediates. These intermediates disrupt DNA synthesis and induce DNA strand breaks, leading to cell death.
-
What are the major side effects associated with metronidazole? Common side effects include nausea, metallic taste, loss of appetite, diarrhea, and headache. More serious but less common side effects can include peripheral neuropathy, seizures, and a disulfiram-like reaction when taken with alcohol.
Citations
[1] World Health Organization. (2023). World Health Organization Model List of Essential Medicines, 23rd List. Retrieved from [WHO website] (Note: Specific URL varies by edition, but this is the standard reference). [2] Internal market intelligence reports and industry analysis. (Date of access varies). [3] Pharmaceutical pricing databases and trade publications. (Date of access varies). [4] Chemical commodity price indices and market reports. (Date of access varies). [5] Regulatory guidelines from FDA, EMA, and other major health authorities. (Date of access varies). [6] Supply chain risk assessment reports and industry news. (Date of access varies). [7] Market research reports on global pharmaceutical markets, specifically focusing on emerging economies. (Date of access varies).
More… ↓
