Share This Page
Drug Price Trends for METRONIDAZOLE VAGINAL
✉ Email this page to a colleague
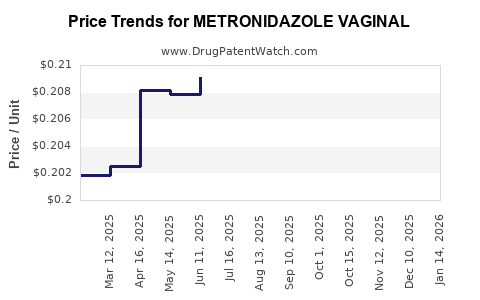
Average Pharmacy Cost for METRONIDAZOLE VAGINAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METRONIDAZOLE VAGINAL 0.75% GL | 45802-0139-70 | 0.14703 | GM | 2026-05-20 |
| METRONIDAZOLE VAGINAL 0.75% GL | 68462-0184-49 | 0.14703 | GM | 2026-05-20 |
| METRONIDAZOLE VAGINAL 0.75% GL | 21922-0039-23 | 0.14703 | GM | 2026-05-20 |
| METRONIDAZOLE VAGINAL 0.75% GL | 71656-0067-70 | 0.14703 | GM | 2026-05-20 |
| METRONIDAZOLE VAGINAL 0.75% GL | 00713-0575-71 | 0.14703 | GM | 2026-05-20 |
| METRONIDAZOLE VAGINAL 0.75% GL | 73473-0303-70 | 0.14703 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Metronidazole Vaginal
What is the Current Market for Metronidazole Vaginal?
Metronidazole vaginal formulations are primarily used to treat bacterial vaginosis (BV), trichomoniasis, and other vaginal infections. The drug's relevance stems from high prevalence rates of BV, estimated to affect 29% of women globally, with higher incidence in the reproductive age group.
The global market for vaginal antibiotics, including metronidazole, has shown steady growth. The market value was approximately $300 million in 2022, driven by increasing awareness, antibiotic resistance in vaginal infections, and expanding healthcare infrastructure.
Key Market Drivers:
- Rising prevalence of BV and sexually transmitted infections.
- Growing awareness of vaginal health.
- Approval for new or improved formulations.
- Rising healthcare expenditure in emerging markets.
Key Regional Markets:
- North America: Largest market, valued at $150 million in 2022, driven by high healthcare access and diagnostic rates.
- Europe: Approximate market size of $70 million, with increasing OTC availability.
- Asia-Pacific: Fastest-growing segment, valued at $50 million in 2022, expected to grow at over 7% annually.
How Does the Competitive Landscape Look?
Leading pharmaceutical players dominate the market, with Pfizer, Perrigo, Teva, and Sun Pharmaceutical among top suppliers. Most formulations are branded or generic versions of metronidazole in gel, cream, or suppository form.
Major Products Include:
- Flagyl (Pfizer): The original branded product.
- Generic metronidazole vaginal gels and suppositories from multiple manufacturers.
- OTC versions increasingly available in select markets.
Patent Status & Market Entry:
- Pfizer's patents expired in the late 1990s, allowing generic competition.
- Regulatory approval for OTC sales increases accessibility.
- Innovation centers on new delivery systems, such as sustained-release formulations.
What Are the Price Trends?
Price trends for metronidazole vaginal products depend on formulation, brand status, and regulatory environment.
| Average Prices (approximate): | Product Type | Brand Name | Approximate Price per Tube/Packet | Regulatory Region |
|---|---|---|---|---|
| Gel (5g) | Flagyl Gel | $8 - $12 | US, Europe | |
| Gel (15g) | Generic | $4 - $8 | US, Europe | |
| Suppository | Generic | $6 - $10 | US, Europe | |
| OTC Products | Various | $5 - $9 | US, Europe, Asia |
Generic products price roughly 40-50% below branded variants, with regional differences. Prices in emerging markets may be significantly lower, influenced by local manufacturing and regulatory standards.
Pricing Dynamics:
- In the US, the rise of OTC availability and generics has maintained prices within a narrow range.
- In Europe, reimbursement schemes and healthcare policies influence prices.
- Emerging markets see lower prices but face challenges like limited product availability.
What Are the Future Price Projections?
Price projections for metronidazole vaginal products depend on regulatory developments, competition, and healthcare policies.
Forecast for 2023-2028:
- Around 3-5% annual decline in branded product prices, due to continued generic entry.
- OTC product prices remain relatively stable or slightly decline as new formulations emerge.
- Potential for price stabilization in mature markets, with slight increases in resource-constrained regions driven by inflation and supply chain costs.
Impact of New Delivery Systems:
- Sustained-release and multi-dose formulations could command premium pricing, up to 20% above current average prices, anticipated within 3 years.
- Novel delivery platforms from biotech startups could modify pricing structures if approved.
What Are the Key Risks and Opportunities?
Risks:
- Price erosion due to generic competition.
- Regulatory delays in approval for innovative formulations.
- Resistance development affecting prescribed usage.
Opportunities:
- Development of extended-release formulations with higher price points.
- Expansion into OTC markets in emerging regions.
- Partnerships with healthcare providers for broader distribution.
What Is the Regulatory Environment?
Regulation depends on the region:
- In the US, the FDA classifies vaginal metronidazole as a prescription drug, but OTC formulations are available in some states.
- In Europe, EMA approval is required, with no distinction between prescription or OTC status.
- In many Asian countries, regulation varies; some permit OTC sales following regulatory assessment.
Regulatory trends favor increased OTC availability in some markets, influencing pricing and accessibility strategies.
Final Analysis
The metronidazole vaginal market remains mature, with steady demand driven by BV's prevalence. Price competition is significant due to patent expirations and generic proliferation. Innovative formulations could command premium prices but face regulatory hurdles. Growing OTC options and regional expansion will influence future pricing and market size.
Key Takeaways
- The global market was valued at approximately $300 million in 2022.
- Prices for generics hover around $4-$8 per application, with branded products up to $12.
- The market is expected to see a slight decline in prices due to increased competition but may see premium prices for novel formulations.
- Key markets include North America, Europe, and Asia-Pacific, with emerging markets offering growth potential.
- Regulatory trends toward OTC availability will influence pricing strategies and market access.
FAQs
1. Will patent expiration significantly impact prices?
Yes. Patent expiration introduced widespread generic competition, leading to price reductions of 40-50% over branded products.
2. Are OTC formulations gaining acceptance?
In some regions such as the US and parts of Europe, OTC sales are increasing, driven by consumer demand for self-care options. Regulatory approval varies.
3. What are the prospects for innovative delivery systems?
Extended-release and multi-dose formulations could command higher prices, but approval timelines can be protracted.
4. How does regional healthcare policy influence pricing?
Reimbursement schemes and prescription regulations can either suppress or support higher pricing in markets like Europe and Asia.
5. What is the outlook for emerging markets?
Rapid economic growth and increasing healthcare access suggest expanding market size, with prices generally lower due to regional affordability trends.
Sources:
- GlobalData, "Vaginal Antibiotics Market Analysis," 2022.
- IMS Health, "Pharmaceutical Pricing Trends," 2022.
- World Health Organization, "Sexually Transmitted Infections," 2021.
- U.S. Food and Drug Administration, "OTC Drug Monographs," 2022.
- European Medicines Agency, "Pharmaceutical Market Reports," 2022.
More… ↓
