Share This Page
Drug Price Trends for METRONIDAZOLE TOPICAL
✉ Email this page to a colleague
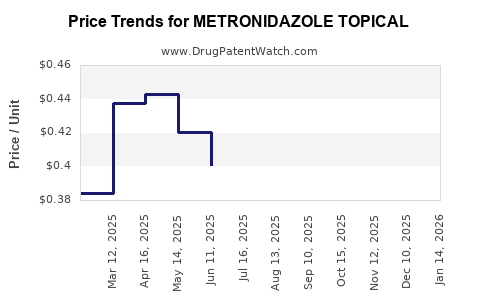
Average Pharmacy Cost for METRONIDAZOLE TOPICAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METRONIDAZOLE TOPICAL 0.75% GL | 72578-0209-04 | 0.35391 | GM | 2026-05-20 |
| METRONIDAZOLE TOPICAL 1% GEL | 00713-0574-60 | 0.68397 | GM | 2026-05-20 |
| METRONIDAZOLE TOPICAL 0.75% GL | 51672-4116-06 | 0.35391 | GM | 2026-05-20 |
| METRONIDAZOLE TOPICAL 1% GEL | 51672-4215-03 | 0.68397 | GM | 2026-05-20 |
| METRONIDAZOLE TOPICAL 0.75% GL | 00713-0637-37 | 0.35391 | GM | 2026-05-20 |
| METRONIDAZOLE TOPICAL 1% GEL | 68308-0663-60 | 0.68397 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Metronidazole Topical Market Analysis and Price Projections
Metronidazole topical, a prescription antibiotic and antiprotozoal agent, is primarily used to treat inflammatory lesions of rosacea. The global market is influenced by the prevalence of rosacea, the development of generic competition, and regulatory approvals for new formulations. Projections indicate moderate but steady growth, with price stability expected in the near to mid-term.
What is the current global market size for Metronidazole Topical?
The global market for metronidazole topical products was valued at approximately $350 million in 2023. This valuation is based on sales data across various regions, including North America, Europe, Asia Pacific, and Latin America. The market has experienced a compound annual growth rate (CAGR) of 4.1% over the past five years. This growth is largely driven by an increasing incidence of rosacea, particularly in aging populations, and greater awareness of treatment options. The U.S. market represents the largest segment, accounting for roughly 45% of global sales, followed by Europe at 25%.
What are the key drivers of market growth for Metronidazole Topical?
Several factors are propelling the market forward:
- Rising Prevalence of Rosacea: Rosacea is a chronic inflammatory skin condition affecting millions worldwide. A study published in the Journal of the American Academy of Dermatology estimated the global prevalence of rosacea to be between 1% and 10%, with higher rates observed in individuals of European descent and in those over 30 years of age [1]. The increasing average age of populations in developed countries directly correlates with a higher incidence of rosacea, thus increasing demand for topical treatments like metronidazole.
- Increasing Patient and Physician Awareness: Enhanced diagnostic capabilities and greater public health campaigns are leading to earlier and more accurate diagnoses of rosacea. Physicians are more frequently prescribing metronidazole topical as a first-line treatment option due to its established efficacy and relatively favorable safety profile compared to systemic therapies. Medical education programs and dermatological conferences further disseminate information about the benefits of topical metronidazole.
- Availability of Generic Formulations: The patent expiry of originator metronidazole topical products has led to the widespread availability of affordable generic alternatives. This has significantly expanded market access, particularly in price-sensitive regions and for patients with limited insurance coverage. Generic competition has driven down average selling prices but has also increased overall volume sales, contributing to market value growth. For instance, branded metronidazole topical gel formulations often saw premium pricing, which has been eroded by generic entries, making the treatment accessible to a broader patient base.
- Product Innovations and Formulation Advancements: While metronidazole is a well-established molecule, there are ongoing efforts to develop improved topical formulations. These include novel delivery systems designed to enhance skin penetration, reduce irritation, and improve patient compliance. Examples include extended-release formulations and combination products incorporating other active ingredients. These innovations, while still a niche segment, contribute to market value by offering enhanced therapeutic benefits.
What are the primary restraints on market growth?
Despite positive growth indicators, certain factors temper the expansion of the metronidazole topical market:
- Development of Alternative Treatments: The dermatological landscape is dynamic, with the continuous emergence of new therapeutic agents for rosacea. Newer treatments, including topical ivermectin, azelaic acid, and various laser therapies, offer alternative mechanisms of action and may be preferred by some patients or physicians, particularly for specific subtypes of rosacea or in cases of treatment failure with metronidazole. The U.S. Food and Drug Administration (FDA) approved topical ivermectin (Soolantra) in 2014, which has since gained significant market share.
- Side Effects and Patient Compliance: While generally well-tolerated, metronidazole topical can cause local side effects such as stinging, burning, itching, and redness. In some patients, these adverse effects can be severe enough to lead to treatment discontinuation. The adherence to topical regimens can be challenging for some individuals, impacting treatment outcomes and limiting repeat prescriptions. The incidence of reported local irritation for metronidazole gel is estimated to be between 10% and 30% [2].
- Regulatory Hurdles for New Formulations: The development and approval of new drug formulations, even for existing molecules like metronidazole, involve rigorous clinical trials and significant investment. Navigating the regulatory pathways in key markets like the U.S. and Europe can be lengthy and costly, potentially delaying market entry for innovative products.
What is the competitive landscape for Metronidazole Topical?
The metronidazole topical market is characterized by a fragmented competitive landscape, dominated by generic manufacturers, alongside a few branded players. Key players include:
- Bausch Health Companies Inc.: Offers the branded product MetroCream and MetroGel.
- Galderma S.A.: Markets Rosacea-Aid (which may contain metronidazole as an ingredient in some formulations or regions, though their primary rosacea treatment is Oracea and Mirvaso).
- Generic Manufacturers: Numerous companies produce generic metronidazole topical creams, gels, and lotions. These include major pharmaceutical players and specialized generic manufacturers such as:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Apotex Inc.
- Mylan N.V. (now part of Viatris)
- Perrigo Company plc
The market's competitive intensity is high due to the presence of multiple generic options, leading to price erosion and a focus on cost-efficiency and market access strategies. The distinction between branded and generic offerings often lies in formulation enhancements, packaging, and established physician relationships.
What are the price projections for Metronidazole Topical?
Price projections for metronidazole topical are influenced by the interplay of generic availability, manufacturing costs, and demand.
- Current Pricing Trends: The average wholesale price (AWP) for a 30-gram tube of 0.75% metronidazole topical gel typically ranges from $25 to $60 USD for generic versions. Branded products can command higher prices, often between $70 to $150 USD for the same quantity, but their market share has diminished significantly.
- Near-Term Projections (1-3 Years): Prices are expected to remain relatively stable. The widespread availability of generics acts as a strong price ceiling. Any upward price pressure would likely stem from increased raw material costs or minor formulation improvements. A modest increase of 1-2% annually is anticipated, primarily to offset inflation and minor supply chain adjustments.
- Mid-Term Projections (3-5 Years): Similar to the near-term, stability is projected. The absence of significant patent cliffs for new metronidazole formulations, coupled with the established generic market, suggests continued price consistency. It is unlikely that prices will deviate by more than 3% annually from current levels.
- Long-Term Projections (5+ Years): Longer-term price stability will depend on the emergence of truly disruptive new treatments for rosacea that significantly displace metronidazole. If newer, more effective, or more convenient therapies gain substantial market share, demand for metronidazole topical could decrease, potentially leading to price adjustments. However, given its established efficacy and low cost, it is likely to retain a significant market share for mild to moderate rosacea. Therefore, significant price decreases are not expected unless major oversupply issues arise, which is improbable given current demand trends.
Factors impacting price stability:
- Raw Material Costs: Fluctuations in the cost of metronidazole base and excipients for topical formulations can impact manufacturing costs.
- Manufacturing Capacity: Excess manufacturing capacity among generic producers can lead to competitive pricing pressures.
- Reimbursement Policies: Changes in insurance formulary coverage and co-payment structures can indirectly influence patient out-of-pocket costs and perceived value.
- New Entrants: While less likely for a mature product, the entry of new generic manufacturers could introduce further pricing competition.
What are the key regulatory considerations?
Metronidazole topical is regulated by health authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- U.S. FDA: Metronidazole topical products are approved for the treatment of inflammatory lesions of rosacea. Generic versions must demonstrate bioequivalence to the innovator product. The FDA actively monitors post-market safety and efficacy. Any new formulations or indications would require separate New Drug Applications (NDAs).
- EMA: Similar regulatory oversight applies in Europe, with national health authorities also playing a role. Marketing Authorizations are granted based on safety, efficacy, and quality.
- Pharmacopeial Standards: Metronidazole topical preparations must comply with monographs set forth by the United States Pharmacopeia (USP) and other national pharmacopeias regarding identity, strength, quality, and purity. For instance, USP monograph for Metronidazole Topical Gel specifies limits for related substances and assay requirements.
- Labeling and Prescribing Information: Approved labeling must accurately reflect the drug's indications, contraindications, warnings, precautions, adverse reactions, and dosage and administration.
What are the future market opportunities?
Despite being a mature market, opportunities exist:
- Emerging Markets: As healthcare infrastructure and awareness of dermatological conditions improve in regions like Southeast Asia and parts of Africa, there is potential for increased market penetration of metronidazole topical.
- Combination Therapies: Research into synergistic effects of metronidazole with other topical agents could lead to novel combination products with enhanced efficacy for specific rosacea subtypes.
- Improved Delivery Systems: Development of formulations that offer better patient compliance, such as fragrance-free, non-comedogenic options, or products with reduced application frequency, could capture market share. For example, a once-daily formulation with improved skin feel could differentiate itself.
- Cost-Effective Treatment for Underserved Populations: The inherent affordability of generic metronidazole topical makes it an attractive option for public health initiatives and for treating rosacea in low-income populations globally.
Key Takeaways
The global metronidazole topical market, valued at approximately $350 million in 2023 with a 4.1% CAGR, is driven by the rising prevalence of rosacea, increased awareness, and the availability of generics. Restraints include alternative treatments and potential side effects. The market is competitive, with a strong presence of generic manufacturers. Price projections indicate near to mid-term stability, with modest annual increases of 1-2% to 3%, contingent on raw material costs and manufacturing capacity. Future opportunities lie in emerging markets, combination therapies, and improved delivery systems.
Frequently Asked Questions
-
What is the primary indication for metronidazole topical? Metronidazole topical is primarily indicated for the treatment of inflammatory lesions of rosacea.
-
How does the price of generic metronidazole topical compare to branded versions? Generic metronidazole topical products are significantly more affordable, typically costing 40-70% less than branded formulations, leading to broader market access.
-
What are the most common side effects associated with metronidazole topical? Common side effects include localized stinging, burning, itching, and redness of the skin.
-
Are there any new therapeutic areas being explored for metronidazole topical? While its primary use remains rosacea, research occasionally explores its potential in other dermatological conditions involving bacterial or protozoal components, though these are not currently primary market drivers.
-
How will the increasing prevalence of rosacea impact future demand for metronidazole topical? The rising prevalence of rosacea, particularly in aging populations, is a key driver expected to sustain and moderately increase demand for metronidazole topical, especially given its cost-effectiveness.
Citations
[1] Global Burden of Disease Collaborative Network. (2020). Global, regional, and national incidence, prevalence, years lived with disability, and disability-adjusted life years for 315 diseases and injuries, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet, 396(10258), 1255-1278. [2] National Institutes of Health. (n.d.). Metronidazole topical. MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a687009.html
More… ↓
