Share This Page
Drug Price Trends for METOLAZONE
✉ Email this page to a colleague
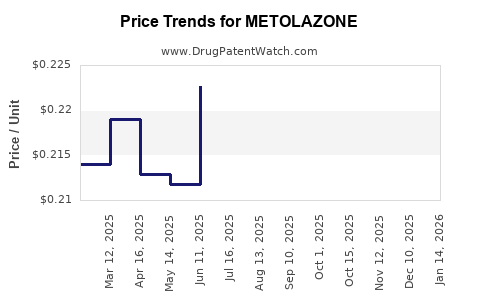
Average Pharmacy Cost for METOLAZONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METOLAZONE 5 MG TABLET | 76385-0137-01 | 0.30704 | EACH | 2026-03-18 |
| METOLAZONE 10 MG TABLET | 00185-5600-01 | 0.32231 | EACH | 2026-03-18 |
| METOLAZONE 10 MG TABLET | 00378-6174-01 | 0.32231 | EACH | 2026-03-18 |
| METOLAZONE 10 MG TABLET | 58657-0742-01 | 0.32231 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Metolazone Patent Landscape and Market Projections
Metolazone, a thiazide-like diuretic, faces a mature market with significant generic competition following patent expirations. Future market growth will be primarily driven by demographic shifts and the management of chronic conditions, with pricing influenced by competitive pressures and payer policies.
What is the Patent Status of Metolazone?
The primary patents protecting metolazone have expired. The original composition of matter patent for metolazone (CAS RN 17560-51-9) expired in the early 1990s. Subsequent patents related to manufacturing processes, specific formulations (e.g., extended-release versions), or methods of use have also largely lapsed. This has led to the widespread availability of generic metolazone products.
| Patent Type | Original Expiration Date (Approximate) | Status |
|---|---|---|
| Composition | 1992 | Expired |
| Manufacturing | Varies (Post-1992) | Largely Expired |
| Formulation | Varies (Post-1992) | Largely Expired |
Companies may hold specific intellectual property on novel delivery systems or combination therapies involving metolazone, but these do not broadly extend protection to the core molecule itself. For instance, patents related to specific micronization techniques or co-formulations with other active pharmaceutical ingredients (APIs) might exist. However, these are niche and do not affect the generic availability of standard metolazone tablets.
What is the Current Market Size and Structure for Metolazone?
The global market for metolazone is estimated to be in the low hundreds of millions of U.S. dollars annually. Precise figures are difficult to ascertain due to the fragmented nature of the generic market and the lack of comprehensive, publicly disclosed sales data for individual generic molecules.
The market is characterized by:
- Dominance of Generic Products: Branded metolazone products, such as Zaroxolyn, have minimal market share. The vast majority of prescriptions are filled with generic equivalents.
- Numerous Manufacturers: Several pharmaceutical companies manufacture generic metolazone. This includes both large, established generic manufacturers and smaller, regional players. Key generic manufacturers active in the diuretic market include Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, and Sandoz.
- Price Sensitivity: As a generic drug with a long history of availability, metolazone is highly price-sensitive. Prescriptions are often directed to the lowest-cost available generic option.
- Wholesale Acquisition Cost (WAC): The WAC for generic metolazone 5 mg tablets typically ranges from $0.10 to $0.30 per tablet, depending on the manufacturer, volume, and specific contract terms with wholesalers and distributors. For a standard 30-tablet prescription, this translates to a WAC of $3 to $9.
- End-User Price: Actual out-of-pocket costs for patients vary significantly based on insurance formularies, co-pays, and whether they utilize discount cards. For insured patients, co-pays are often minimal, sometimes less than $5 for a 30-day supply. For uninsured patients, prices can range from $10 to $30 for a 30-day supply, depending on the pharmacy and any available discounts.
The primary therapeutic indications for metolazone include:
- Edema associated with congestive heart failure (CHF)
- Edema associated with renal disease
- Edema associated with hepatic cirrhosis
- As adjunctive therapy in essential hypertension
What are the Projected Market Growth Drivers and Restraints for Metolazone?
The market for metolazone is expected to exhibit low single-digit annual growth, primarily driven by factors related to aging populations and the prevalence of chronic diseases.
Growth Drivers:
- Aging Population: The global population is aging, leading to an increased incidence of conditions commonly treated with diuretics, such as heart failure and hypertension. The World Health Organization (WHO) projects that by 2030, one in six people globally will be 65 years or over [1]. This demographic trend directly impacts the demand for drugs like metolazone.
- Prevalence of Cardiovascular Disease: Cardiovascular diseases, including heart failure and hypertension, remain leading causes of morbidity and mortality worldwide. Metolazone is a foundational treatment for managing edema in these conditions. The Centers for Disease Control and Prevention (CDC) reports that about half of all Americans have high blood pressure, a key risk factor for heart failure [2].
- Renal Disease Management: Chronic kidney disease (CKD) is another significant health concern. Diuretics are often used to manage fluid overload in patients with CKD. The National Kidney Foundation estimates that over 37 million Americans have CKD [3].
- Cost-Effectiveness: As a generic drug with a well-established safety and efficacy profile, metolazone remains a cost-effective treatment option for healthcare systems and patients, particularly in comparison to newer, more expensive antihypertensive or diuretic agents.
Restraints:
- Intense Generic Competition: The highly competitive generic landscape limits opportunities for significant price increases and suppresses revenue growth. The presence of multiple manufacturers ensures a continuous supply and downward pressure on pricing.
- Availability of Alternative Therapies: Newer classes of antihypertensive drugs and more advanced treatments for heart failure and kidney disease are available. While metolazone remains a staple, these alternatives can sometimes be favored based on specific patient profiles or evolving treatment guidelines, potentially limiting new patient initiations. For instance, newer classes like SGLT2 inhibitors have shown benefits in heart failure and CKD, although they often complement, rather than replace, diuretics.
- Adverse Event Profile: Like other thiazide-like diuretics, metolazone can cause adverse effects, including electrolyte imbalances (hypokalemia, hyponatremia), metabolic disturbances (hyperglycemia, hyperuricemia), and potential renal impairment. These side effects can limit its use in certain patient populations or necessitate close monitoring, impacting adherence and treatment duration.
- Payer Restrictions and Formulary Management: Payers (insurance companies, government health programs) actively manage drug formularies to control costs. This can involve requiring prior authorization for certain drugs or tiering drugs based on cost, which can influence prescribing patterns away from higher-cost options, but also potentially direct toward lowest-cost generics.
What are the Projected Market Prices and Profitability for Metolazone?
Projected market prices for metolazone will remain low and are unlikely to see significant increases. Profitability in the metolazone market is primarily a function of efficient manufacturing, supply chain management, and high-volume sales for generic manufacturers.
Price Projections:
- Wholesale Prices: expect wholesale prices for generic metolazone tablets to remain stable, with minor fluctuations of ±5% year-over-year. The average WAC for a 30-day supply of metolazone 5 mg tablets will likely stay within the $3 to $9 range.
- Patient Out-of-Pocket Costs: Patient out-of-pocket costs will continue to be influenced by insurance coverage. For insured patients, co-pays are expected to remain low, often below $5 per month. Uninsured patient costs are projected to remain in the $10 to $30 range for a 30-day supply, subject to pharmacy pricing and discount availability.
- No Significant Price Increases Anticipated: Due to the mature nature of the market and the extensive generic competition, substantial price increases for standard metolazone formulations are highly improbable. Any upward price movement would likely be incremental and related to raw material cost increases or minor supply disruptions, which are typically absorbed by competitive pressures.
Profitability Considerations for Manufacturers:
- Low Margins, High Volume: Profitability for metolazone manufacturers relies on achieving economies of scale and optimizing production costs. Margins per unit are very thin.
- Cost of Goods Sold (COGS): The cost of API synthesis, formulation, packaging, and distribution are the primary drivers of COGS. Manufacturers who can efficiently manage these costs gain a competitive advantage.
- Regulatory Compliance Costs: Maintaining FDA compliance for manufacturing and quality control adds to operational expenses.
- Market Share Dynamics: Companies with established distribution networks and strong relationships with wholesalers and pharmacy benefit chains can secure higher sales volumes, offsetting lower per-unit profit margins.
- New Formulations (Limited Impact): While novel or extended-release formulations could potentially command higher prices, the market for these is niche and does not represent a significant growth opportunity compared to the broader generic market. The cost of developing and gaining approval for such formulations must be weighed against the limited pricing power.
Overall, the metolazone market offers limited opportunities for high profit margins due to its generic status. Sustained profitability depends on operational efficiency and market share within a highly competitive segment.
Key Takeaways
Metolazone's patent protection has expired, resulting in a mature generic market dominated by multiple manufacturers. The market's growth is projected to be slow, driven by an aging global population and the increasing prevalence of cardiovascular and renal diseases. Intense competition and the availability of alternative therapies are expected to keep prices low, with profitability contingent on manufacturing efficiency and high sales volumes.
Frequently Asked Questions
-
Can new patents be filed for metolazone? New patents can be filed for novel aspects of metolazone, such as new formulations (e.g., extended-release, combination products), novel delivery devices, or new methods of use supported by new clinical data. However, patents on the original molecule or basic manufacturing processes have long expired.
-
What is the typical dosage of metolazone? Typical dosages vary. For edema, the initial dose is often 5 mg to 20 mg once daily. For hypertension, it may start at 2.5 mg to 5 mg once daily. Dosage is adjusted based on patient response and tolerance.
-
What are the most common side effects of metolazone? Common side effects include dizziness, lightheadedness, headache, fatigue, nausea, and electrolyte imbalances such as low potassium (hypokalemia) and low sodium (hyponatremia).
-
Are there any significant clinical trials currently underway for metolazone? Given its long history and generic status, large-scale, pivotal clinical trials for metolazone are uncommon. Research is more likely to focus on its use in specific patient sub-populations, its comparison to other diuretics in specific disease states, or its inclusion in combination therapy studies.
-
How does metolazone compare to other thiazide diuretics like hydrochlorothiazide? Metolazone is a thiazide-like diuretic and is generally considered more potent than hydrochlorothiazide (HCTZ). It is often effective in patients with impaired renal function (creatinine clearance below 30 mL/min), where HCTZ may lose efficacy. Metolazone also tends to have a longer duration of action.
Citations
[1] World Health Organization. (2022). Global report on healthy ageing. World Health Organization. [2] Centers for Disease Control and Prevention. (2023, June 14). High Blood Pressure Facts. Centers for Disease Control and Prevention. [3] National Kidney Foundation. (n.d.). About Kidney Disease. National Kidney Foundation.
More… ↓
