Share This Page
Drug Price Trends for METHYLPHENIDATE ER
✉ Email this page to a colleague
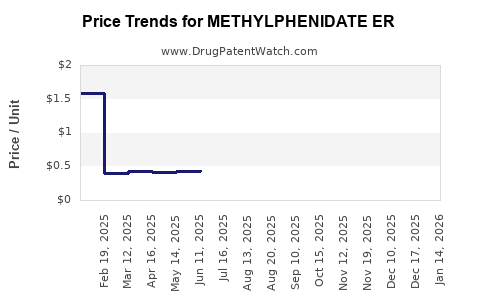
Average Pharmacy Cost for METHYLPHENIDATE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHYLPHENIDATE ER(LA) 60 MG CP | 75907-0053-30 | 10.57109 | EACH | 2026-03-18 |
| METHYLPHENIDATE ER(LA) 40 MG CP | 75907-0052-01 | 2.35720 | EACH | 2026-03-18 |
| METHYLPHENIDATE ER(LA) 30 MG CP | 75907-0051-01 | 2.10531 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
METHYLPHENIDATE ER Market Analysis and Financial Projection
What Is the Current Market Size and Demand for Methylphenidate ER?
Methylphenidate extended-release (ER) formulations are used primarily to treat Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy. Global sales for methylphenidate-based products reached approximately $4.2 billion in 2022, with steady growth observed over the past five years at an average annual rate of 8%. The U.S. dominates the market, accounting for roughly 65% of sales, driven by high diagnosis rates and prescribing patterns.
The demand for methylphenidate ER formulations is compounded by increased awareness and diagnosis of ADHD, especially in pediatric populations, and expanding adult ADHD treatment. The efficacy of ER formulations in improving adherence and compliance compared to immediate-release versions supports their sustained demand.
Who Are the Key Manufacturers and What Is Their Market Share?
Major players include Novartis (now part of Novartis Pharmaceuticals Corporation), Janssen (a subsidiary of Johnson & Johnson), and UCB. These companies hold significant market shares, with Janssen leading at approximately 40%, followed by Novartis at 25%, and UCB managing roughly 10%. Several generic manufacturers occupy the remaining 25%, offering cost-competitive options that have penetrated various markets.
In terms of pipeline products, several generic versions are in development, which could influence market dynamics and pricing strategies.
What Are Current Pricing Trends and Factors Influencing Price Levels?
Average wholesale prices (AWP) for methylphenidate ER formulations vary:
- Brand-name products: $400-$600 per month for a typical 30-day supply.
- Generics: $150-$300 per month, depending on formulations and dosing.
Pricing is influenced by manufacturing costs, patent statuses, the level of market competition, and regulatory policies:
- Patent expirations have resulted in increased generic availability, leading to price erosion.
- Managed care organizations negotiate discounts and rebates, lowering effective patient costs.
- Regulatory restrictions or healthcare policies in different countries influence pricing strategies.
In the U.S., pricing pressure from cost-containment programs has led to a decline of approximately 10-15% over the past three years for branded formulations.
How Might Future Price Trends Evolve?
Price projections suggest that generic competition will maintain downward pressure on prices through 2027, with an estimated compound annual decrease of 3-5% in average costs. However, if new proprietary ER formulations with improved delivery mechanisms (such as osmotic-release formulations) secure regulatory approval and patent protection, they might sustain higher prices for their extended patent terms.
Market penetration of biosimilar or biosimilar-like methylphenidate ER products remains limited but has the potential to influence pricing further, especially in markets outside the U.S.
What Are the Regulatory and Patent Landscape Implications?
The patent expiration dates for key methylphenidate ER products are as follows:
- Concerta (Janssen): patent expired in 2017 and 2020 in various jurisdictions.
- Ritalin LA (Novartis): patent expired in 2014, leading to increased generics.
Patent expiries have catalyzed generic entry, increasing market competition. New formulations with novel delivery systems—such as OROS (osmotic-controlled release oral delivery system)—are protected by additional patents, delaying generic competition.
Regulatory pathways for generic approval, such as FDA's ANDA process, have facilitated rapid market entry by generics once patents expire, contributing to the price decline trend.
What Are Industry Analysts’ Price Projections?
Analysts project that the global methylphenidate ER market will grow at a CAGR of 7-9% through 2030, driven by expanded indications and increased diagnosis rates. Price levels are expected to decline modestly, with average market prices dropping by 5-8% annually over the next five years due to intensifying generic competition.
Estimates from IQVIA predict that branded methylphenidate ER products will continue commanding premium prices in proprietary markets, whereas generics will sustain a downward pricing trajectory.
Key Takeaways
- Global methylphenidate ER sales reached around $4.2 billion in 2022, with the U.S. leading.
- Major companies include Janssen, Novartis, and UCB; generics account for roughly 25% of sales.
- Prices for brand-name products range from $400-$600/month; generics are priced between $150-$300/month.
- Patent expirations have spurred generic entry, leading to sustained price declines.
- Future pricing will depend on novel formulations, patent protections, and market penetration of biosimilars, with a projected 3-8% annual decrease in prices over the next five years.
Frequently Asked Questions
1. When do major patents for leading methylphenidate ER products expire?
Patents for key products like Concerta expired in 2017 and 2020 in various jurisdictions, opening the market for generics.
2. How do generic formulations impact pricing and availability?
Generics introduce significant price competition, leading to reductions of 10-15%, and increase access due to lower costs.
3. Are new delivery systems for methylphenidate ER in development?
Yes; innovations like osmotic delivery systems have received regulatory approval, extending patent protections and potentially sustaining higher prices.
4. Which regulatory factors influence methylphenidate ER market dynamics?
Patent protections, ANDA approvals, and healthcare reimbursement policies shape market entry and pricing strategies.
5. What are the main drivers behind future market growth?
Increased diagnosis of ADHD across age groups, expanding treatment indications, and new formulation approvals drive the growth, offset by pricing pressures.
Sources
- IQVIA. (2023). Pharmaceutical Market Data.
- FDA. (2022). ANDA Approvals and Patent Data.
- EvaluatePharma. (2023). Global Market Forecasts.
- MarketWatch. (2023). ADHD Drug Market Analysis.
- U.S. Patent and Trademark Office. (2022). Patent Expiry Data.
More… ↓
