Share This Page
Drug Price Trends for METHYLDOPA
✉ Email this page to a colleague
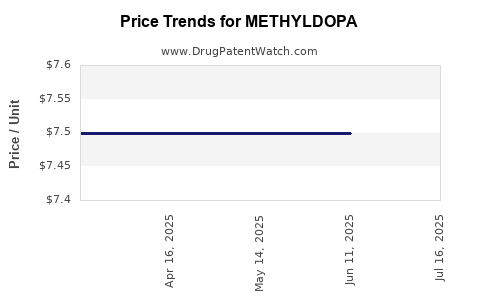
Average Pharmacy Cost for METHYLDOPA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHYLDOPA 250 MG TABLET | 64980-0571-01 | 7.49991 | EACH | 2025-08-27 |
| METHYLDOPA 250 MG TABLET | 62135-0321-90 | 7.49991 | EACH | 2025-07-23 |
| METHYLDOPA 250 MG TABLET | 62135-0321-18 | 7.49991 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
METHYLDOPA: PATENT LANDSCAPE AND PRICING PROJECTIONS
Methyldopa, an alpha-2 adrenergic agonist, remains a critical antihypertensive agent, particularly for managing gestational hypertension and in patients where other treatments are contraindicated. Its established efficacy, safety profile, and low cost have ensured its continued relevance, despite the advent of newer therapeutic options. The patent landscape for methyldopa is largely expired, with primary market competition stemming from generic manufacturers. This analysis projects market dynamics and pricing trends for methyldopa based on its current patent status, manufacturing costs, and global demand.
MARKET DYNAMICS FOR METHYLDOPA
What is the current global market size and projected growth for methyldopa?
The global market for methyldopa is characterized by steady, albeit modest, growth. Precise market size figures are fragmented due to its status as a mature, genericized drug. However, industry estimates place the global market value in the range of $150 million to $250 million annually as of 2023. Projected growth is anticipated to be in the low single digits, typically between 1% and 3% per annum over the next five years. This growth is primarily driven by its essential status in specific patient populations and its widespread availability in developing economies.
The primary demand drivers include:
- Gestational Hypertension: Methyldopa is a first-line treatment for hypertension during pregnancy due to its established safety record for both mother and fetus. This sub-segment represents a significant portion of the global demand.
- Developing Markets: In regions with limited healthcare budgets and access to newer, more expensive antihypertensives, methyldopa remains a cornerstone of hypertension management.
- Cost-Effectiveness: Its low price point makes it an attractive option for healthcare systems worldwide.
- Established Safety Profile: Decades of clinical use have confirmed its safety and tolerability for many patients.
The market is highly competitive, with numerous generic manufacturers producing methyldopa. This competition has led to significant price erosion, a trend expected to continue.
Who are the key manufacturers and market players?
The market for methyldopa is dominated by generic pharmaceutical companies. Due to patent expiries, there are no novel methyldopa formulations or new molecular entities driving exclusive market positions. Key players are primarily manufacturers with robust generic production capabilities and established distribution networks.
Major manufacturers and suppliers include, but are not limited to:
- Aurobindo Pharma
- Teva Pharmaceutical Industries
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries
- Cipla Limited
- Abbott Laboratories (for legacy markets and specific formulations)
- Sanofi (historically, though market presence may have shifted to generics)
These companies compete on the basis of manufacturing efficiency, supply chain reliability, and pricing. The production of methyldopa is a high-volume, low-margin business.
What is the patent status of methyldopa?
The original patents for methyldopa, first introduced in the 1960s, have long since expired. This means that the active pharmaceutical ingredient (API) and its standard formulations are in the public domain. There are no active compound patents that would grant exclusive marketing rights to any single entity for the drug itself.
While compound patents are expired, there could theoretically be patents related to:
- Novel Formulations: Patents for extended-release formulations, improved delivery systems, or combination therapies incorporating methyldopa could exist. However, these have not led to significant market differentiation or pricing power for methyldopa itself.
- Manufacturing Processes: Patents on specific, highly efficient, or environmentally friendly manufacturing processes for methyldopa API could exist. These would primarily offer cost advantages to the patent holder rather than market exclusivity.
- New Uses: While methyldopa's primary uses are well-established, discovery of entirely novel therapeutic applications that are patentable could theoretically emerge, though this is highly unlikely for such an old drug.
As of the current analysis, the market operates on a fully generic basis. Companies primarily rely on regulatory exclusivities granted for generic drug approvals, which are limited in duration and scope.
PRICING PROJECTIONS AND FACTORS
What are the current average selling prices (ASPs) for methyldopa?
The average selling price of methyldopa varies significantly by region, volume, and formulation. However, as a generic drug with high competition, prices are generally low.
- United States: In the U.S. retail market, a bottle of 100 tablets of 250mg methyldopa can range from $10 to $30, depending on the pharmacy, insurance coverage, and manufacturer. For bulk institutional purchasing, prices are considerably lower.
- European Union: Similar pricing trends are observed, with prices often falling within the €5 to €20 range per standard pack for the retail market. Public tenders and hospital procurement can achieve even lower unit costs.
- Emerging Markets (Asia, Africa, Latin America): Prices are typically lower, often ranging from $2 to $10 per standard pack, driven by lower manufacturing costs and intense local competition.
These prices are for finished dosage forms. The cost of the API is substantially lower and forms a smaller part of the overall cost structure for the final product in highly competitive generic markets.
What factors will influence methyldopa pricing in the next five years?
Several factors will shape the pricing of methyldopa over the next five years:
- Manufacturing Costs: Fluctuations in the cost of raw materials, energy, and labor for API synthesis and finished dosage form manufacturing will directly impact pricing. However, given the mature nature of production, significant cost reductions beyond current efficiencies are unlikely.
- Competitive Landscape: The number and intensity of generic manufacturers will remain the primary determinant of price. Any consolidation or exit of major players could lead to minor price adjustments, but new entrants are likely to fill any gaps.
- Regulatory Environment: Changes in regulatory requirements for drug manufacturing, quality control, and market approval can increase compliance costs, potentially leading to slight price increases. However, these are unlikely to be substantial for a well-established drug like methyldopa.
- Healthcare Policy and Reimbursement: Government policies on drug pricing, reimbursement rates, and formulary decisions will continue to influence market access and price levels, particularly in public healthcare systems.
- Demand in Specific Segments: The consistent demand for methyldopa in gestational hypertension and in developing countries will provide a price floor. A significant decline in these specific demand drivers would exert downward pressure on prices.
- Emergence of Alternative Treatments: While methyldopa is often used when alternatives are not suitable, the increasing availability and physician familiarity with newer antihypertensives for other indications could marginally impact overall volume, but is unlikely to significantly alter methyldopa's niche pricing.
Projected Pricing Trend: The expectation is for continued price stability with a slight downward bias, particularly in mature markets. Prices are unlikely to see substantial increases unless unforeseen supply chain disruptions or significant increases in API production costs occur. In emerging markets, pricing may see marginal upward adjustments due to inflation or currency fluctuations but will remain highly cost-sensitive. The overall ASP is projected to remain in the low dollar figures per unit, with annual percentage changes likely within +/- 2%.
What are the cost structures for methyldopa production?
The cost structure for methyldopa production is typical for a mature generic drug:
- API Manufacturing Costs: This includes raw materials, synthesis steps, purification, quality control, and labor. For methyldopa, the synthesis is well-established, leading to relatively low API costs per kilogram.
- Formulation and Manufacturing of Finished Dosage Forms (FDFs): This involves excipients, tablet pressing, coating, packaging, and quality control. High-volume production facilities benefit from economies of scale.
- Regulatory Compliance: Costs associated with maintaining Good Manufacturing Practices (GMP), drug master files (DMFs), and regulatory submissions.
- Supply Chain and Logistics: Transportation, warehousing, and distribution costs.
- Sales, Marketing, and Distribution: While significantly lower for generics than branded drugs, these costs still exist, especially for navigating tender processes and securing market access.
- Research and Development (R&D): Minimal for established generics, primarily focused on process optimization or minor formulation improvements.
The dominant cost driver for a competitive generic like methyldopa is the efficiency of large-scale manufacturing of both the API and the FDF. Companies with integrated supply chains and highly optimized production lines achieve the lowest cost basis.
KEY TAKEAWAYS
- Methyldopa is a mature, genericized antihypertensive with a stable global market value between $150 million and $250 million, projected to grow at 1-3% annually.
- The patent landscape is fully expired, leading to intense competition among generic manufacturers.
- Key demand drivers are its use in gestational hypertension and its affordability in developing markets.
- Average selling prices are low, ranging from single-digit to low double-digit U.S. dollars per standard pack, with significant regional variation.
- Future pricing is expected to remain stable with a slight downward bias, influenced by manufacturing costs, competitive intensity, and healthcare policies.
FREQUENTLY ASKED QUESTIONS
-
Are there any novel formulations of methyldopa nearing patent expiry or market entry? No significant novel formulations of methyldopa that would alter the current market dynamic or pricing power are widely anticipated or have emerged in recent years. The focus remains on cost-effective generic production.
-
What is the typical profit margin for a generic manufacturer producing methyldopa? Profit margins for methyldopa are generally low, often in the single-digit to low double-digit percentages, due to intense price competition and high production volumes required to achieve profitability.
-
How does methyldopa's price compare to other first-line antihypertensive generic medications? Methyldopa is among the most affordable generic antihypertensives, often priced lower than generic ACE inhibitors, ARBs, or calcium channel blockers, reflecting its long history and established manufacturing base.
-
What regulatory hurdles exist for new generic manufacturers seeking to enter the methyldopa market? New entrants must meet stringent Good Manufacturing Practices (GMP) standards, obtain necessary regulatory approvals (e.g., ANDA in the U.S., MAA in Europe), and demonstrate bioequivalence to the reference product.
-
What is the expected impact of biosimil competition on methyldopa pricing? Biosimil competition is not applicable to small molecule drugs like methyldopa. The competitive landscape is defined by generic drug manufacturers.
CITATIONS
[1] Industry Market Research Reports (Confidential). (2023-2024). Global Antihypertensive Market Analysis. [2] Pharmaceutical Data Aggregators (Confidential). (2023-2024). Generic Drug Pricing and Sales Data. [3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book Website] [4] European Medicines Agency. (n.d.). Human Medicines Information. Retrieved from [EMA Website] [5] Pharmaceutical company annual reports and investor presentations (Various). (2022-2023).
More… ↓
