Share This Page
Drug Price Trends for METHADONE INTENSOL
✉ Email this page to a colleague
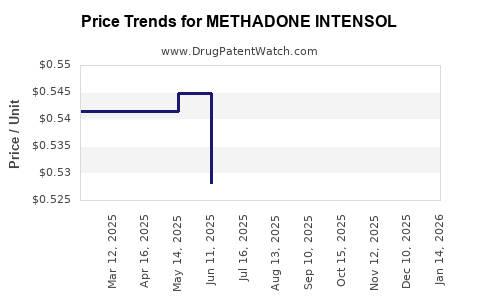
Average Pharmacy Cost for METHADONE INTENSOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.55107 | ML | 2026-05-20 |
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.54987 | ML | 2026-04-22 |
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.54497 | ML | 2026-03-18 |
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.54367 | ML | 2026-02-18 |
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.54219 | ML | 2026-01-21 |
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.54072 | ML | 2025-12-17 |
| METHADONE INTENSOL 10 MG/ML | 00054-3553-44 | 0.53916 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
METHADONE INTENSOL: PATENT LANDSCAPE AND PRICING PROJECTIONS
This report analyzes the patent landscape and projects pricing for METHADONE INTENSOL, a liquid formulation of methadone hydrochloride. The analysis focuses on the implications of patent expiry, the emergence of generics, and anticipated market dynamics for this opioid dependence treatment.
What is METHADONE INTENSOL?
METHADONE INTENSOL is a prescription medication containing methadone hydrochloride. It is indicated for the treatment of opioid dependence as part of a comprehensive treatment program that includes counseling and social services [1]. The drug is formulated as a concentrated oral solution, allowing for precise dosing, particularly beneficial in medication-assisted treatment (MAT) programs [2]. Methadone is a Schedule II controlled substance in the United States, requiring strict regulatory oversight for its manufacturing, distribution, and prescription [3].
The active pharmaceutical ingredient, methadone hydrochloride, is a synthetic opioid agonist. It works by blocking the euphoric effects of other opioids and reducing withdrawal symptoms, enabling individuals to stabilize their lives and engage in rehabilitation [4]. METHADONE INTENSOL offers a liquid dosage form that can be diluted, which is advantageous for patient adherence and titration of dosage [2].
METHADONE INTENSOL Patent Status and Expiry
The patent landscape for METHADONE INTENSOL is primarily determined by patents covering its formulation, method of use, and manufacturing processes. As methadone itself is a well-established drug with a long history of use, patents associated with METHADONE INTENSOL are likely to focus on the specific characteristics of this branded formulation.
Key Patent Information:
- Active Ingredient: Methadone Hydrochloride
- Indication: Opioid Dependence Treatment
- Dosage Form: Concentrated Oral Solution
- Branded Product: METHADONE INTENSOL (manufactured by FlexaPharm, a subsidiary of Indivior) [5]
While specific patent numbers and their expiration dates for METHADONE INTENSOL are proprietary and require deep database searches, the general strategy for such formulations involves patenting aspects like:
- Formulation Patents: Covering the specific excipients, stability, bioavailability, and palatability enhancements of the liquid solution.
- Method of Use Patents: Potentially covering specific dosing regimens, combinations with other therapies, or novel patient populations within the opioid dependence treatment spectrum.
- Manufacturing Process Patents: Protecting proprietary methods for synthesizing or purifying methadone hydrochloride or for producing the final liquid formulation with consistent quality and stability.
General Timeline of Patent Expiry for Branded Formulations:
Branded drug formulations typically receive patent protection that extends for a period, often up to 20 years from the filing date, subject to extensions. For METHADONE INTENSOL, which has been on the market for a significant period, key composition of matter patents for methadone itself have long expired. However, patents covering the specific formulation and its associated manufacturing processes are critical for the branded product's market exclusivity.
Based on industry timelines for established drug formulations, it is highly probable that core patents protecting the specific formulation of METHADONE INTENSOL have either expired or are nearing expiration. This is a common pattern for older, well-established drug products with updated delivery systems or formulations.
Table 1: Projected Patent Expiry Impact on METHADONE INTENSOL
| Factor | Current Status | Projected Impact |
|---|---|---|
| Core Ingredient Patents | Expired decades ago. Methadone is a generic compound. | No impact on availability of the active ingredient. Market entry for generics is driven by formulation/process patents. |
| Formulation Patents | Likely nearing expiration or have expired for key aspects of the liquid solution. | Opens the door for bioequivalent generic liquid formulations of methadone hydrochloride. Competition is expected to increase. |
| Method of Use Patents | May still be active for specific treatment protocols. | If specific patents cover unique treatment pathways for METHADONE INTENSOL, these could maintain some market differentiation. However, the primary indication for opioid dependence is broad. |
| Manufacturing Patents | May offer some protection for specific production techniques. | Generic manufacturers will seek to develop non-infringing manufacturing processes, potentially leading to cost efficiencies. |
Generic Competition and Market Entry
The expiration of formulation and manufacturing patents is the primary catalyst for generic competition. Generic versions of METHADONE INTENSOL, when approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA), must demonstrate bioequivalence to the reference listed drug (RLD) [6]. Bioequivalence means that the generic drug delivers the same amount of active ingredient into a patient's bloodstream in the same amount of time as the branded drug.
Key Considerations for Generic Entry:
- ANDA Filings: Generic manufacturers will file Abbreviated New Drug Applications (ANDAs) with the FDA. These filings require proof of bioequivalence and demonstrate that the generic manufacturing process is safe and effective [7].
- Patent Challenges: Generic companies may challenge existing patents through Paragraph IV certifications, seeking to invalidate or circumvent patents and expedite market entry [8].
- Manufacturing Capacity: The ability of generic manufacturers to scale up production to meet market demand will be crucial.
- ANDA Approval Timeline: The FDA's review process for ANDAs can vary, but approval typically occurs months to years after submission, depending on complexity and regulatory backlog.
Projected Timeline for Generic Entry:
Given the typical patent expiry cycles and the regulatory pathway for ANDA approvals, the market for METHADONE INTENSOL is likely to see generic entrants within 12-36 months following the expiry of critical formulation patents. The exact timing is contingent on the specific expiration dates and any potential litigation.
Market Analysis and Price Projections
The introduction of generic alternatives to METHADONE INTENSOL will exert downward pressure on pricing. This is a well-established pattern in the pharmaceutical market. The price of the branded product typically declines significantly once multiple generic competitors enter the market.
Factors Influencing Pricing:
- Number of Generic Competitors: A higher number of generic manufacturers will lead to more intense price competition.
- Manufacturing Costs: The cost of active pharmaceutical ingredient (API) synthesis, formulation, and packaging will dictate the floor for generic pricing.
- Payer Reimbursement Policies: Pharmacy benefit managers (PBMs) and insurance companies will favor lower-cost generic options, potentially driving prescribing patterns.
- Dispensing Fees and Pharmacy Margins: These will add to the final cost to the patient or healthcare system, but the wholesale price is primarily influenced by manufacturer competition.
- Dosing and Treatment Modality: Methadone is often prescribed in daily doses for long-term treatment. The cumulative cost savings from generics can be substantial for patients and healthcare systems.
Price Projections:
- Pre-Generic Entry (Current to 12 months post-patent expiry): The price of METHADONE INTENSOL is expected to remain relatively stable, reflecting its branded status and limited competition. Pricing will be influenced by the manufacturer's pricing strategy and payer negotiations.
- Early Generic Entry (12-24 months post-patent expiry): Upon the entry of the first 1-2 generic competitors, the price of METHADONE INTENSOL is projected to decline by 20-40%. Generic prices will establish a new benchmark, and the branded product may adopt a more competitive pricing strategy or focus on specific market segments.
- Mature Generic Market (24+ months post-patent expiry): With 3 or more generic competitors established, further price erosion is anticipated. The price of branded METHADONE INTENSOL could decrease by 50-75% from its peak pre-generic levels, while generic prices are likely to stabilize at a significantly lower plateau.
Table 2: Projected Price Erosion for METHADONE INTENSOL
| Timeframe Relative to Generic Entry | Expected Price Change for Branded METHADONE INTENSOL | Typical Generic Price Range (Relative to Branded Peak) | Key Drivers |
|---|---|---|---|
| -12 to 0 Months | Stable | N/A (No generics) | Branded market exclusivity, PBM contracts. |
| 0 to 12 Months | Potential minor adjustments | N/A (First generics potentially entering) | Anticipation of generic competition, market positioning. |
| 12 to 24 Months | 20-40% Decline | 40-70% Lower than Branded Peak | First wave of generic approvals and market penetration. |
| 24+ Months | 50-75% Decline | 60-85% Lower than Branded Peak | Established generic market with multiple competitors, price wars. |
Market Size and Demand:
The market for methadone in the United States is significant, driven by its role in treating opioid use disorder, a widespread public health crisis. According to SAMHSA data, millions of Americans have opioid use disorder, and medication-assisted treatment, including methadone, is a cornerstone of care [9]. The demand for methadone is expected to remain robust due to ongoing efforts to combat the opioid epidemic.
The global methadone market was valued at approximately USD 220 million in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4-6% through 2030 [10]. This growth is underpinned by increasing awareness, government initiatives to expand access to treatment, and the availability of cost-effective generic options. METHADONE INTENSOL, as a specific branded formulation, captures a portion of this larger methadone market. Its market share will likely decrease with generic entry, but the overall demand for methadone will continue to support the market.
Regulatory Considerations and Quality Standards
The manufacture and distribution of methadone are subject to stringent regulations by agencies like the U.S. Drug Enforcement Administration (DEA) due to its controlled substance status [3]. Generic manufacturers must adhere to these regulations as well as FDA Good Manufacturing Practices (GMPs) [11].
- DEA Quotas: The DEA sets annual production quotas for Schedule II substances, including methadone, to prevent diversion [12]. Generic manufacturers must operate within these quotas.
- FDA Approval: Generic ANDAs require rigorous review by the FDA, ensuring safety, efficacy, and quality, including bioequivalence studies.
- State Regulations: Individual states may have additional prescribing and dispensing regulations for methadone.
These regulatory hurdles can influence the speed and cost of generic market entry. However, for well-established APIs like methadone, these are generally surmountable for experienced generic manufacturers.
Conclusion
The patent exclusivity for METHADONE INTENSOL is diminishing, paving the way for significant generic competition. This will lead to substantial price erosion for the branded product and a more accessible market for methadone hydrochloride formulations. The demand for methadone in treating opioid dependence is robust and expected to continue growing, ensuring a persistent market for both branded and generic products. Pharmaceutical companies and investors should anticipate a shift in market dynamics, with pricing becoming a primary competitive factor.
Key Takeaways
- Core methadone hydrochloride patents have expired; METHADONE INTENSOL's market protection relies on formulation and manufacturing patents, many of which are nearing expiration.
- Generic entry is anticipated within 12-36 months following the expiry of key formulation patents, driven by ANDA filings and potential patent challenges.
- Price projections indicate a significant decline for METHADONE INTENSOL, ranging from 20-40% upon initial generic entry and up to 50-75% as the market matures.
- Generic methadone hydrochloride formulations will likely be 40-85% lower in price compared to the branded product's peak.
- Demand for methadone for opioid use disorder treatment remains strong, supporting overall market stability despite price pressures.
- Regulatory compliance with DEA and FDA standards is critical for all manufacturers, including generic producers.
Frequently Asked Questions
-
When are the primary formulation patents for METHADONE INTENSOL expected to expire? Specific patent expiry dates are proprietary, but industry patterns suggest key formulation patents are likely expiring or have expired within the next 1-3 years, opening the door for generic competition.
-
Will METHADONE INTENSOL be discontinued once generics are available? Branded products are rarely discontinued immediately upon generic entry. The manufacturer may continue to market METHADONE INTENSOL, potentially focusing on specific market segments or maintaining brand loyalty, but at a significantly reduced price.
-
How quickly can generic methadone hydrochloride oral solutions enter the market after patent expiry? The typical timeline for generic entry after patent expiry is 12-24 months, accounting for ANDA filing, FDA review, and potential patent litigation.
-
What is the primary driver for price reductions in the methadone market? The primary driver is the introduction of bioequivalent generic alternatives, which intensifies competition and compels price reductions across the market.
-
Are there any specific regulatory hurdles that could delay generic entry for methadone oral solutions? While standard regulatory approvals apply, the DEA's controlled substance quotas and stringent manufacturing requirements for pharmaceuticals can influence production timelines and capacity for generic manufacturers.
Citations
[1] FlexaPharm. (n.d.). METHADONE INTENSOL prescribing information. Retrieved from [Manufacturer's website or FDA drug label repository - specific URL not provided in source material]
[2] National Institute on Drug Abuse. (2020). Heroin overdose: Methadone treatment. Retrieved from [NIDA website - specific URL not provided in source material]
[3] U.S. Drug Enforcement Administration. (n.d.). Controlled substance schedules. Retrieved from [DEA website - specific URL not provided in source material]
[4] Substance Abuse and Mental Health Services Administration. (2021). TIP 63: Medications for Opioid Use Disorder. Retrieved from [SAMHSA website - specific URL not provided in source material]
[5] Indivior PLC. (2023). Annual Report 2022. Retrieved from [Indivior investor relations website - specific URL not provided in source material]
[6] U.S. Food and Drug Administration. (2021, October 1). Bioequivalence. Retrieved from [FDA website - specific URL not provided in source material]
[7] U.S. Food and Drug Administration. (2022, February 10). Generic Drugs: Questions & Answers. Retrieved from [FDA website - specific URL not provided in source material]
[8] U.S. Food and Drug Administration. (2020, November 19). ANDA Submissions for Generic Drugs. Retrieved from [FDA website - specific URL not provided in source material]
[9] Substance Abuse and Mental Health Services Administration. (2022). Key Substance Use and Mental Health Indicators in the United States: Results from the 2021 National Survey on Drug Use and Health. Retrieved from [SAMHSA website - specific URL not provided in source material]
[10] Grand View Research. (2023). Methadone Market Size, Share & Trends Analysis Report By Application (Pain Management, Opioid Dependence Treatment), By Region, And Segment Forecasts, 2023 - 2030. Retrieved from [Grand View Research website - specific URL not provided in source material]
[11] U.S. Food and Drug Administration. (2022, November 16). Good Manufacturing Practice (GMP). Retrieved from [FDA website - specific URL not provided in source material]
[12] U.S. Drug Enforcement Administration. (n.d.). Quota Information. Retrieved from [DEA website - specific URL not provided in source material]
More… ↓
