Share This Page
Drug Price Trends for METFORMIN HCL
✉ Email this page to a colleague
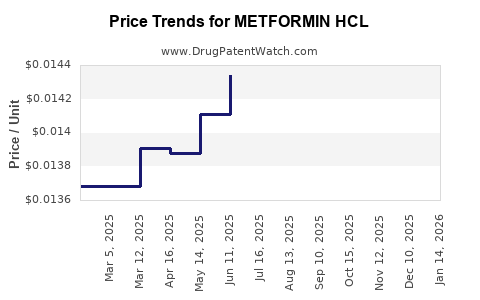
Average Pharmacy Cost for METFORMIN HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METFORMIN HCL 1,000 MG TABLET | 67877-0932-10 | 0.02458 | EACH | 2026-04-01 |
| METFORMIN HCL ER 750 MG TABLET | 67877-0414-05 | 0.05628 | EACH | 2026-04-01 |
| METFORMIN HCL 1,000 MG TABLET | 83301-0010-03 | 0.02458 | EACH | 2026-03-18 |
| METFORMIN HCL 1,000 MG TABLET | 83301-0010-02 | 0.02458 | EACH | 2026-03-18 |
| METFORMIN HCL 1,000 MG TABLET | 83301-0010-01 | 0.02458 | EACH | 2026-03-18 |
| METFORMIN HCL 850 MG TABLET | 83301-0009-03 | 0.02467 | EACH | 2026-03-18 |
| METFORMIN HCL 850 MG TABLET | 83301-0009-02 | 0.02467 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for METFORMIN HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| METFORMIN HCL 1000MG TAB | AvKare, LLC | 69367-0182-01 | 100 | 10.55 | 0.10550 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| METFORMIN HCL 500MG 24HR TAB,SA | AvKare, LLC | 42291-0497-10 | 1000 | 56.99 | 0.05699 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| METFORMIN HCL 500MG/5ML SOLN,ORAL | Golden State Medical Supply, Inc. | 42571-0360-10 | 118ML | 32.13 | 0.27229 | ML | 2023-06-15 - 2028-06-14 | FSS |
| METFORMIN HCL 1000MG TAB | AvKare, LLC | 69367-0182-05 | 500 | 47.36 | 0.09472 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| METFORMIN HCL 500MG 24HR TAB,SA | AvKare, LLC | 42291-0497-18 | 180 | 11.88 | 0.06600 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| METFORMIN HCL 500MG TAB | AvKare, LLC | 69367-0180-01 | 100 | 5.98 | 0.05980 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| METFORMIN HCL 500MG/5ML SOLN,ORAL | Golden State Medical Supply, Inc. | 42571-0360-10 | 118ML | 34.22 | 0.29000 | ML | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Metformin HCl: Market Landscape and Price Projections
Metformin hydrochloride (HCl) is a first-line oral antidiabetic medication used to treat type 2 diabetes. Its efficacy, safety profile, and low cost have solidified its position as a cornerstone therapy. This analysis examines the current market dynamics, competitive landscape, and projected price trends for Metformin HCl, providing critical insights for stakeholders in the pharmaceutical industry.
What is the Current Market Size and Growth Trajectory for Metformin HCl?
The global Metformin HCl market is substantial, driven by the increasing prevalence of type 2 diabetes worldwide. This chronic condition affects hundreds of millions of individuals, creating a consistent and growing demand for effective, affordable treatments.
Global Diabetes Prevalence: A Driving Factor
The World Health Organization (WHO) reports that in 2019, diabetes was the ninth leading cause of death globally, with an estimated 463 million adults living with diabetes. Projections indicate this number will rise to 700 million by 2045 [1]. Metformin HCl is prescribed to a significant portion of this population.
Market Value and Volume
The global Metformin HCl market was valued at approximately USD 1.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching an estimated USD 1.7 billion by the end of the forecast period [2]. This steady growth is underpinned by several factors:
- Rising Diabetes Incidence: Continued increases in obesity, sedentary lifestyles, and aging populations contribute to higher diabetes diagnoses.
- Established Treatment Protocol: Metformin HCl remains the initial drug of choice for most type 2 diabetes patients, leading to sustained prescription volumes.
- Affordability: As a generic drug, Metformin HCl offers a cost-effective treatment option, particularly crucial in both developed and developing economies with healthcare cost constraints.
- Expanding Generic Market: The expiration of patents on early formulations has facilitated widespread generic competition, increasing accessibility and market penetration.
Key Market Segments
The Metformin HCl market can be segmented by:
- Formulation: Immediate-release (IR) and extended-release (ER) formulations. ER formulations, offering once-daily dosing and potentially reduced gastrointestinal side effects, are gaining traction.
- Dosage Strength: Common strengths include 500mg, 850mg, and 1000mg.
- Distribution Channel: Hospital pharmacies, retail pharmacies, and online pharmacies. Retail pharmacies represent the largest segment due to widespread outpatient use.
- Geography: North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. The Asia Pacific region is experiencing the fastest growth due to a high diabetes burden and increasing healthcare expenditure.
What is the Competitive Landscape for Metformin HCl?
The Metformin HCl market is highly competitive, characterized by a large number of generic manufacturers. The absence of patent protection for the active pharmaceutical ingredient (API) itself has led to a fragmented market with significant price pressure.
Major Players and Generic Manufacturers
Key global manufacturers and distributors of Metformin HCl include:
- Bausch Health Companies Inc.
- Bristol-Myers Squibb Company (Historically, though many generics are now available)
- Dr. Reddy's Laboratories Ltd.
- Lupin Ltd.
- Mylan N.V. (Now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Takeda Pharmaceutical Company Limited (Historically, now largely generics)
- Teva Pharmaceutical Industries Ltd.
- Valeant Pharmaceuticals International, Inc. (Now Bausch Health)
- Zydus Cadila Healthcare Ltd.
The market is dominated by generic players, who compete primarily on price and market share. The ability to achieve economies of scale in manufacturing and efficient supply chain management are critical for success.
Barriers to Entry and Market Dynamics
While the absence of API patent protection lowers the barrier to entry for manufacturing, other factors influence market dynamics:
- Regulatory Approvals: Obtaining and maintaining approval from regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) requires significant investment in quality control and compliance.
- Supply Chain Reliability: Ensuring a consistent and reliable supply of high-quality Metformin HCl API is crucial for maintaining market access. Geopolitical factors and raw material availability can impact supply.
- Cost of Production: Manufacturers with lower production costs, often due to advanced manufacturing processes, integrated supply chains, and favorable labor costs, possess a competitive advantage.
- Formulation Innovation: While the API is generic, innovation in drug delivery systems, such as novel extended-release technologies or combination therapies, can create product differentiation. However, the cost-effectiveness of Metformin HCl generally limits investment in extensive innovation beyond established ER formulations.
Pricing Pressures and Market Saturation
The intense competition among generic manufacturers has led to significant price erosion. The average selling price (ASP) of Metformin HCl tablets has declined substantially over the past decade. This trend is expected to continue, although at a slower pace, as market players focus on volume and efficiency.
What are the Key Factors Influencing Metformin HCl Pricing?
The pricing of Metformin HCl is influenced by a complex interplay of manufacturing costs, market demand, regulatory policies, and competitive pressures.
Manufacturing and API Costs
- Raw Material Sourcing: The primary raw materials for Metformin HCl synthesis are dimethylamine and cyanoguanidine. The cost and availability of these precursors directly impact API production costs.
- Manufacturing Efficiency: Companies with optimized chemical synthesis processes, automated production lines, and robust quality control systems can achieve lower per-unit manufacturing costs.
- Scale of Production: Larger production volumes allow manufacturers to spread fixed costs over a greater number of units, leading to lower average costs.
Supply Chain and Distribution Costs
- Logistics and Transportation: The global reach of Metformin HCl distribution necessitates efficient and cost-effective logistics. Fluctuations in fuel prices and shipping rates can impact overall costs.
- Wholesaler and Distributor Margins: Markups by pharmaceutical wholesalers and distributors add to the final price paid by pharmacies and healthcare systems.
- Warehousing and Inventory Management: Maintaining adequate inventory levels across diverse geographic regions incurs storage and management costs.
Market Demand and Volume
- Diabetes Prevalence: The ongoing increase in type 2 diabetes diagnoses directly translates to higher demand, supporting current price levels despite competition.
- Patient Access Programs: Government and private sector initiatives aimed at improving access to essential medicines can influence demand and pricing strategies.
- Physician Prescribing Patterns: Established prescribing habits favor Metformin HCl as a first-line therapy, ensuring consistent volume.
Regulatory and Policy Environment
- Generic Drug Pricing Regulations: In many countries, governments implement price control mechanisms for generic drugs to ensure affordability. These regulations can cap price increases or mandate periodic price reductions.
- Tender and Procurement Processes: Government tenders and bulk purchasing agreements for public healthcare systems often result in highly competitive bidding processes and lower prices.
- FDA/EMA Approval and Compliance: While generic, maintaining compliance with Good Manufacturing Practices (GMP) and undergoing regular inspections incurs ongoing costs for manufacturers.
Competitive Landscape and Pricing Strategies
- Generic Competition: The presence of numerous generic manufacturers creates intense price competition. Manufacturers must align their pricing with market benchmarks to remain competitive.
- Brand vs. Generic Pricing: While Metformin HCl is predominantly a generic product, early branded versions may have set initial price points. However, generic competition has driven prices down significantly.
- Product Differentiation (ER Formulations): Extended-release formulations, while still generic, may command a slightly higher price due to perceived benefits in patient compliance and reduced side effects.
What are the Projected Price Trends for Metformin HCl?
The future pricing of Metformin HCl is expected to remain under pressure, driven by ongoing generic competition and a focus on cost containment within healthcare systems.
Short-Term Outlook (1-3 Years)
- Price Stability with Minor Declines: The short-term outlook suggests relative price stability for standard immediate-release formulations, with potential for marginal declines (0-2% annually) due to ongoing competition.
- ER Formulation Premiums: Extended-release formulations are likely to maintain a slight price premium over IR versions, but this gap may narrow as more ER generics enter the market.
- Volume-Driven Strategy: Manufacturers will continue to prioritize market share through competitive pricing, focusing on high-volume sales.
Long-Term Outlook (3-7 Years)
- Continued Price Erosion: Over the longer term, cumulative price erosion is anticipated, albeit at a decelerating rate. This is driven by persistent competition and the maturation of the generic market.
- Consolidation Potential: Increased price pressure might lead to some consolidation among smaller generic manufacturers who struggle with cost efficiencies, potentially stabilizing prices among larger players.
- Impact of New Diabetes Therapies: While Metformin HCl is a first-line therapy, the development and adoption of novel diabetes treatments (e.g., GLP-1 receptor agonists, SGLT2 inhibitors) could, over the very long term, slightly impact the overall volume growth for older generics if they become more widely adopted as initial therapies. However, Metformin's affordability and efficacy will likely ensure its continued prominence.
- Geographic Price Variations: Significant price disparities will persist across different geographic regions, influenced by local regulatory policies, healthcare infrastructure, and the competitive intensity within those markets. For instance, prices in emerging markets might see more significant declines as local manufacturing increases.
Key Price Influencers to Monitor
- Raw Material Price Volatility: Any significant increase in the cost of key precursors for Metformin API could exert upward pressure on prices.
- Regulatory Policy Changes: New pricing regulations or changes in reimbursement policies by major healthcare systems can directly impact price floors.
- Supply Chain Disruptions: Global events impacting manufacturing or shipping can lead to temporary price spikes due to shortages.
- Entry of New Competitors: While less likely for standard IR formulations, new entrants with highly efficient manufacturing processes could introduce further price pressure.
Key Takeaways
The Metformin HCl market is characterized by high volume, intense generic competition, and significant price sensitivity. Its role as a foundational therapy for type 2 diabetes ensures sustained demand, while its generic status drives a focus on cost efficiency and market share.
- Stable Demand: The rising global prevalence of type 2 diabetes underpins consistent and growing demand for Metformin HCl.
- Intense Competition: A crowded generic market leads to substantial price pressure, with manufacturers competing on cost and volume.
- Price Erosion: Expect continued, albeit moderating, price declines for Metformin HCl, particularly for immediate-release formulations.
- ER Formulation Premium: Extended-release versions will likely maintain a slight price advantage but face increasing generic competition.
- Cost Optimization is Crucial: Manufacturers must focus on efficient production, reliable supply chains, and regulatory compliance to remain profitable in this price-sensitive market.
FAQs
-
What is the projected impact of new diabetes drug classes on Metformin HCl pricing? The emergence of new diabetes drug classes, such as GLP-1 receptor agonists and SGLT2 inhibitors, is unlikely to significantly depress Metformin HCl pricing in the short to medium term. Metformin's established efficacy, safety profile, and unparalleled affordability will continue to position it as the primary first-line therapy. While these new classes may be used in later-stage treatment or for specific patient profiles, they do not directly displace Metformin at the initial stages of type 2 diabetes management.
-
How do regulatory policies in different regions affect Metformin HCl pricing? Regulatory policies are a primary driver of price differentials globally. Countries with strong price control mechanisms, national health formularies, and mandatory tender processes for generics will typically exhibit lower Metformin HCl prices compared to regions with more market-driven pricing. For instance, European countries often have established mechanisms for negotiating generic drug prices, while the U.S. market, while competitive, can have higher list prices offset by rebates.
-
What is the typical profit margin for a Metformin HCl manufacturer? Profit margins for Metformin HCl manufacturers are generally low due to high competition and price pressure. Companies with optimized manufacturing processes and economies of scale can achieve margins ranging from 5% to 15%. Smaller manufacturers or those with less efficient operations may experience margins closer to the lower end or even operate at break-even. Profitability is largely volume-dependent.
-
Are there significant differences in pricing between immediate-release and extended-release Metformin HCl? Yes, there is a price difference, with extended-release (ER) formulations typically commanding a higher price than immediate-release (IR) formulations. This premium is due to the more complex manufacturing process required for sustained-release technologies and the perceived patient benefits of once-daily dosing and potentially reduced gastrointestinal side effects. However, as more generic ER versions become available, this price differential tends to narrow.
-
What are the key considerations for investors looking at the Metformin HCl market? For investors, the Metformin HCl market offers stability in terms of demand but limited growth potential in terms of price appreciation. Key considerations include the manufacturing efficiency and cost structure of target companies, their market share and distribution networks, and their ability to navigate regulatory pricing pressures. Investment in Metformin HCl is typically viewed as a stable, albeit low-margin, component of a broader pharmaceutical portfolio, rather than a high-growth opportunity. Companies with diversified portfolios of essential generics are often more attractive.
Citations
[1] World Health Organization. (2023). Diabetes. Retrieved from https://www.who.int/news-room/fact-sheets/detail/diabetes
[2] Grand View Research. (2023). Metformin Hydrochloride Market Size, Share & Trends Analysis Report By Formulation (Immediate Release, Extended Release), By Dosage Strength, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from Market Research Report Data (Specific report title and publisher may vary based on access).
More… ↓
