Share This Page
Drug Price Trends for MESALAMINE ER
✉ Email this page to a colleague
Average Pharmacy Cost for MESALAMINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
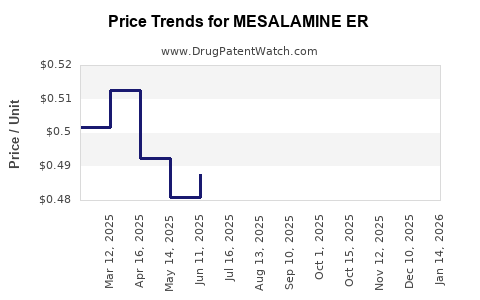
| MESALAMINE ER 0.375 GRAM CAP | 60687-0650-32 | 0.48846 | EACH | 2026-05-20 |
| MESALAMINE ER 0.375 GRAM CAP | 50268-0577-11 | 0.48846 | EACH | 2026-05-20 |
| MESALAMINE ER 0.375 GRAM CAP | 59651-0397-08 | 0.48846 | EACH | 2026-05-20 |
| MESALAMINE ER 0.375 GRAM CAP | 00093-9224-89 | 0.48846 | EACH | 2026-05-20 |
| MESALAMINE ER 0.375 GRAM CAP | 50268-0577-15 | 0.48846 | EACH | 2026-05-20 |
| MESALAMINE ER 0.375 GRAM CAP | 00378-1375-78 | 0.48846 | EACH | 2026-05-20 |
| MESALAMINE ER 0.375 GRAM CAP | 00832-6056-12 | 0.48846 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Mesalamine ER Market Analysis and Price Projections
Executive Summary
The mesalamine extended-release (ER) market, critical for treating inflammatory bowel disease (IBD) including ulcerative colitis (UC) and Crohn's disease, is projected to experience moderate growth driven by an increasing IBD prevalence and demand for convenient oral therapies. Patent expirations for key innovator products have led to generic competition, impacting pricing dynamics and creating opportunities for biosimil developers. The market is characterized by established treatment protocols and a focus on patient adherence.
What is Mesalamine ER and Its Therapeutic Significance?
Mesalamine, or 5-aminosalicylic acid (5-ASA), is the active metabolite of sulfasalazine and a cornerstone therapy for mild to moderate ulcerative colitis. Extended-release formulations of mesalamine are designed to deliver the active drug directly to the colon, the primary site of inflammation in UC. This targeted delivery minimizes systemic absorption, thereby reducing potential side effects while maximizing therapeutic efficacy. Mesalamine ER is indicated for the induction and maintenance of remission in patients with ulcerative colitis. Its therapeutic significance lies in its anti-inflammatory properties within the gastrointestinal tract, targeting inflammatory pathways and inhibiting the production of inflammatory mediators.
What is the Current Market Landscape for Mesalamine ER?
The mesalamine ER market is mature, with several branded and generic products available. Key market segments include various dosage forms and strengths designed for different patient needs and adherence preferences.
Leading Mesalamine ER Products
| Product Name | Manufacturer(s) | Active Ingredient | Dosage Form | Key Indication |
|---|---|---|---|---|
| Lialda | Takeda Pharmaceutical | Mesalamine | Delayed-release tablets | Ulcerative Colitis |
| Apriso | Actelion Pharmaceuticals (now part of Johnson & Johnson) | Mesalamine | Granules | Ulcerative Colitis |
| Delzicol | Tris Pharma | Mesalamine | Delayed-release capsules | Ulcerative Colitis |
| Various Generics | Multiple manufacturers | Mesalamine | Tablets, Capsules, Granules | Ulcerative Colitis |
The market has seen the impact of generic entry following patent expirations for innovator products such as Lialda. This has intensified price competition among manufacturers.
What are the Key Drivers of the Mesalamine ER Market?
Several factors are influencing the demand and growth trajectory of the mesalamine ER market.
Increasing Prevalence of Inflammatory Bowel Disease
The global incidence and prevalence of IBD, including ulcerative colitis and Crohn's disease, are on the rise. A study published in The Lancet in 2018 estimated that over 6.8 million people worldwide were living with IBD in 2015 [1]. This growing patient population directly translates to a larger addressable market for therapeutic agents like mesalamine ER. Environmental factors, genetic predispositions, and changes in gut microbiota are implicated in this trend.
Preference for Oral Extended-Release Formulations
Patients often prefer oral medications due to convenience and ease of administration compared to alternative delivery methods such as suppositories or enemas. Extended-release formulations offer the benefit of once-daily dosing, which can significantly improve patient adherence. Poor adherence is a known challenge in chronic disease management, and once-daily regimens are associated with better outcomes in ulcerative colitis [2].
Development of Novel Formulations and Delivery Systems
While the core active ingredient remains mesalamine, ongoing research and development efforts are focused on optimizing delivery systems. This includes developing formulations with improved bioavailability, enhanced targeting to specific colonic segments, and potentially combination therapies. These advancements can lead to differentiated products and capture market share.
Generic Competition and Market Accessibility
The expiration of patents for innovator mesalamine ER products has opened the door for generic manufacturers. The introduction of generics has led to a significant decrease in prices for mesalamine ER, making the treatment more accessible to a broader patient population and reducing healthcare costs. This competition also drives innovation among generic companies to optimize manufacturing processes and supply chain efficiency.
What are the Restraints Affecting the Mesalamine ER Market?
Despite positive growth drivers, the mesalamine ER market faces certain limitations.
Emergence of Biologics and Advanced Therapies
For moderate to severe IBD cases, biologics (e.g., infliximab, adalimumab) and small molecule inhibitors (e.g., tofacitinib) have become standard treatment options. These advanced therapies offer different mechanisms of action and can be more effective for patients refractory to conventional treatments like mesalamine ER. The established efficacy and expanding use of these novel agents can limit the market expansion of mesalamine ER, particularly in advanced disease stages.
Side Effects and Patient Tolerance
While generally well-tolerated, mesalamine ER can cause side effects, including headache, nausea, and abdominal pain. In some individuals, nephrotoxicity (kidney damage) is a potential concern, necessitating regular monitoring of renal function. Patient intolerance or the development of adverse events can lead to treatment discontinuation or switching to alternative therapies, thereby limiting market penetration.
Pricing Pressures from Payers and Healthcare Systems
With the availability of multiple generic options, payers and healthcare systems are exerting significant downward pressure on mesalamine ER prices. This creates a challenging pricing environment for manufacturers, requiring them to focus on cost-effective production and efficient distribution networks to maintain profitability.
What are the Pricing Dynamics and Projections for Mesalamine ER?
The pricing of mesalamine ER is heavily influenced by the interplay of patent status, generic competition, and market demand.
Historical Pricing Trends
Innovator mesalamine ER products, such as Lialda, initially commanded premium pricing upon their launch. The average wholesale price (AWP) for a 30-day supply could range from $400 to $600 or more. Following patent expirations and the subsequent entry of generic alternatives, prices have declined significantly. Generic mesalamine ER 30-day supplies are now typically available at prices ranging from $50 to $150, depending on the manufacturer, dosage strength, and pharmacy.
Factors Influencing Future Pricing
- Generic Competition: The continued presence and potential increase in the number of generic manufacturers will maintain downward pressure on prices.
- Payer Negotiations: Increasingly sophisticated payer negotiations and formulary management strategies will aim to secure the lowest possible prices for mesalamine ER.
- Supply Chain Efficiencies: Manufacturers who can optimize their supply chains and reduce manufacturing costs will be better positioned to offer competitive pricing.
- Product Differentiation: While challenging in a genericized market, manufacturers might attempt to differentiate through novel delivery systems or extended release profiles, potentially allowing for slight pricing premiums if clinical benefits are demonstrated.
- Regulatory Landscape: Changes in regulatory requirements for drug approval and manufacturing could indirectly influence production costs and, consequently, pricing.
Price Projections
The mesalamine ER market is expected to see continued price erosion in the near to medium term due to robust generic competition.
- Short-term (1-3 years): Average prices for generic mesalamine ER are projected to remain stable or experience a slight decline of 2-5% annually as new generic entrants may emerge or existing ones adjust their strategies. The AWP for a 30-day supply is likely to stay within the $50-$120 range.
- Medium-term (3-7 years): Price stabilization is anticipated as the market matures with a defined set of generic competitors. Minor fluctuations might occur based on supply/demand dynamics and manufacturing innovations. Projections suggest prices will hover between $50-$110.
- Long-term (7+ years): In the long term, pricing will largely be dictated by manufacturing costs and the overall demand for mesalamine ER in the context of evolving IBD treatment paradigms. Significant price increases are unlikely unless substantial unmet needs are identified that mesalamine ER uniquely addresses.
It is important to note that these projections are based on current market conditions and do not account for unforeseen regulatory changes, significant clinical breakthroughs, or major shifts in IBD treatment guidelines.
What are the Key Opportunities in the Mesalamine ER Market?
The mesalamine ER market presents several avenues for strategic growth and investment.
Geographic Expansion
While established in North America and Europe, there is significant opportunity for mesalamine ER products in emerging markets in Asia, Latin America, and Africa where IBD prevalence is rising and access to advanced therapies may be limited. Tailoring market entry strategies to local regulatory environments and pricing sensitivities will be crucial.
Development of Improved Formulations
Research into novel mesalamine ER formulations that offer enhanced efficacy, better patient compliance, or reduced side effects could create a competitive advantage. Examples include formulations with improved colonic targeting, extended release profiles for even less frequent dosing, or combination products.
Strategic Partnerships and Acquisitions
Companies can pursue strategic partnerships or acquisitions to gain access to established distribution networks, expand their product portfolios, or acquire innovative technologies. This can accelerate market penetration and strengthen competitive positioning.
Focus on Patient Adherence Programs
Developing and implementing robust patient support and adherence programs can differentiate products and improve real-world outcomes. These programs can include educational materials, reminder systems, and nurse support, addressing a key challenge in chronic disease management.
What are the Threats to the Mesalamine ER Market?
Several factors pose potential risks to the mesalamine ER market.
Intensifying Generic Competition
The entry of additional generic manufacturers can further drive down prices, making it challenging for existing players to maintain profitability without significant cost efficiencies.
Shift Towards Advanced Therapies
As biologics and other novel treatments gain wider acceptance and are approved for earlier stages of IBD, the reliance on mesalamine ER for mild to moderate disease might diminish, particularly in regions with advanced healthcare infrastructure.
Regulatory Hurdles and Compliance Costs
Manufacturers must navigate complex and evolving regulatory landscapes, including stringent manufacturing standards and post-market surveillance requirements. Non-compliance can lead to product recalls, fines, and reputational damage, impacting market access and profitability.
Unforeseen Side Effect Profiles
While mesalamine is generally considered safe, any emerging evidence linking it to significant, previously uncharacterized adverse events could negatively impact its market perception and physician prescribing habits.
Key Takeaways
The mesalamine ER market is characterized by a growing patient base for IBD, driven by increasing disease prevalence and the preference for convenient oral therapies. Patent expirations have led to robust generic competition, resulting in significant price reductions and increased market accessibility. While biologics and advanced therapies pose a competitive threat for severe IBD cases, mesalamine ER remains a vital treatment option for mild to moderate ulcerative colitis. Future market growth will depend on factors such as the development of improved formulations, strategic geographic expansion, and effective patient adherence programs. Pricing will continue to be influenced by generic competition and payer negotiations, with an expectation of sustained price stability or marginal decline.
Frequently Asked Questions
What is the typical daily dosage of mesalamine ER for ulcerative colitis?
Typical daily dosages for mesalamine ER range from 2.4 grams to 4.8 grams, often administered once daily, depending on the specific product and the severity of the condition.
Are there any long-term safety concerns associated with mesalamine ER?
Long-term use of mesalamine ER is generally considered safe, but potential concerns include nephrotoxicity (kidney damage). Regular monitoring of renal function is recommended by healthcare professionals.
How does mesalamine ER compare to other 5-ASA formulations?
Mesalamine ER formulations are designed for targeted release in the colon, offering advantages in patient convenience and potentially reducing systemic side effects compared to older or non-extended-release formulations.
What is the patent expiration status for major mesalamine ER brands?
Key patents for major mesalamine ER brands, such as Lialda, have expired, allowing for the widespread availability of generic versions.
Can mesalamine ER be used to treat Crohn's disease?
While primarily indicated for ulcerative colitis, mesalamine ER is sometimes used off-label for mild cases of Crohn's disease affecting the colon, though its efficacy is more established for UC.
Citations
[1] Dean, N. I., et al. (2018). Global, regional, and national burden of inflammatory bowel disease, 1990–2018: a systematic analysis for the Global Burden of Disease Study 2018. The Lancet, 399(10329), 1055-1070.
[2] Afzali, S., et al. (2017). Adherence to mesalamine therapy in inflammatory bowel disease patients. Gastroenterology, 152(5), S231-S232.
More… ↓
