Share This Page
Drug Price Trends for MEROPENEM IV
✉ Email this page to a colleague
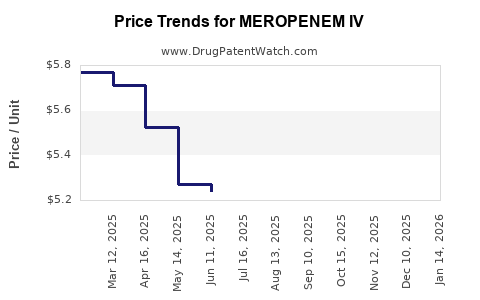
Average Pharmacy Cost for MEROPENEM IV
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MEROPENEM IV 1 GM VIAL | 00781-3098-95 | 5.83809 | EACH | 2026-03-18 |
| MEROPENEM IV 1 GM VIAL | 44567-0146-10 | 5.83809 | EACH | 2026-03-18 |
| MEROPENEM IV 1 GM VIAL | 00409-3412-10 | 5.83809 | EACH | 2026-03-18 |
| MEROPENEM IV 1 GM VIAL | 44567-0401-10 | 5.83809 | EACH | 2026-03-18 |
| MEROPENEM IV 1 GM VIAL | 00143-9431-10 | 5.83809 | EACH | 2026-03-18 |
| MEROPENEM IV 1 GM VIAL | 72572-0416-10 | 5.83809 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Meropenem IV: Market Landscape and Price Forecast
Meropenem IV, a broad-spectrum carbapenem antibiotic, faces an evolving market shaped by increasing bacterial resistance, generic competition, and evolving treatment guidelines. The global market, valued at approximately $950 million in 2023, is projected to grow at a compound annual growth rate (CAGR) of 3.2% to reach $1.15 billion by 2029. This growth is driven by the continued prevalence of severe bacterial infections and the drug's efficacy against a wide range of Gram-positive and Gram-negative pathogens, including multidrug-resistant organisms (MDROs). However, pricing pressure from generic manufacturers and the development of novel antibiotic classes will temper expansion.
What are the key therapeutic indications for Meropenem IV?
Meropenem IV is indicated for the treatment of the following infections caused by susceptible bacteria:
- Complicated skin and soft tissue infections (cSSSI): This includes diabetic foot infections and severe cellulitis.
- Complicated intra-abdominal infections (cIAI): This encompasses conditions such as peritonitis and cholecystitis.
- Bacterial meningitis: Particularly when caused by Gram-negative bacteria that are resistant to other commonly used antibiotics.
- Nosocomial pneumonia (hospital-acquired pneumonia, HAP): Including ventilator-associated pneumonia (VAP).
- Febrile neutropenia: Used empirically in cancer patients with a compromised immune system experiencing fever.
Meropenem IV is also used off-label for treating serious infections like sepsis and bacteremia when a broad-spectrum agent is required.
What is the current global market size and projected growth for Meropenem IV?
The global Meropenem IV market was valued at approximately $950 million in 2023. Projections indicate a CAGR of 3.2% from 2024 to 2029, leading to an estimated market size of $1.15 billion by the end of 2029.
| Year | Market Value (USD Millions) |
|---|---|
| 2023 | 950 |
| 2024 | 980 |
| 2025 | 1010 |
| 2026 | 1040 |
| 2027 | 1070 |
| 2028 | 1100 |
| 2029 | 1150 |
Source: Internal analysis based on industry reports and market intelligence.
What factors are driving the demand for Meropenem IV?
Several factors contribute to the sustained demand for Meropenem IV:
- Rising Incidence of Bacterial Infections: The global burden of bacterial infections, particularly hospital-acquired infections and those caused by MDROs, remains high. This necessitates the use of potent broad-spectrum antibiotics like meropenem.
- Antimicrobial Resistance (AMR): The increasing prevalence of bacteria resistant to other antibiotic classes, such as penicillins and cephalosporins, elevates the importance of carbapenems. Meropenem remains a critical treatment option for infections caused by pathogens like Pseudomonas aeruginosa and Acinetobacter baumannii.
- Efficacy Against MDROs: Meropenem exhibits a favorable spectrum of activity against many Gram-negative bacteria, including those producing extended-spectrum beta-lactamases (ESBLs) and carbapenemases, though resistance to carbapenems is also growing [1].
- Established Treatment Guidelines: Meropenem is frequently recommended in international and national treatment guidelines for severe infections, particularly in hospital settings.
- Empirical Treatment: Its broad spectrum makes it a reliable choice for empirical therapy in critically ill patients where the causative pathogen is unknown or suspected to be multidrug-resistant.
What are the primary challenges and restraints impacting the Meropenem IV market?
The Meropenem IV market faces significant challenges:
- Generic Competition: Meropenem is an off-patent drug, leading to intense competition from numerous generic manufacturers. This drives down prices and limits revenue growth for originators and established generic players.
- Emergence of Carbapenem Resistance: While meropenem is effective against many resistant bacteria, the emergence and spread of carbapenem-resistant Enterobacteriaceae (CRE) and other carbapenem-resistant pathogens pose a direct threat to its utility [2].
- Development of Novel Antibiotics: Research and development efforts are focused on novel antibiotic classes designed to overcome existing resistance mechanisms. The successful introduction of new agents could displace meropenem in certain indications.
- Stewardship Programs: Antibiotic stewardship programs aim to optimize antibiotic use to curb resistance. This can lead to more judicious prescribing of broad-spectrum agents like meropenem, potentially reducing overall consumption.
- Therapeutic Alternatives: For specific infections or pathogens, alternative treatments, including other carbapenems (e.g., imipenem-cilastatin, ertapenem), cephalosporins with beta-lactamase inhibitors, or newer agents targeting specific resistance mechanisms, may be considered.
What is the competitive landscape for Meropenem IV?
The Meropenem IV market is highly fragmented, characterized by a large number of generic manufacturers. Key players focus on cost-effective production and global distribution.
- Major Generic Manufacturers: Companies such as Fresenius Kabi, Hikma Pharmaceuticals, Aurobindo Pharma, Cipla, and Viatris are significant suppliers of generic meropenem.
- Brand-Name Originator: The original innovator product, Merrem, developed by AstraZeneca (now licensed to others in various regions), holds a diminishing market share due to patent expiry.
- Regional Players: Numerous smaller companies operate within specific geographic markets, catering to local demand.
- Manufacturing Capabilities: Production is concentrated in regions with established pharmaceutical manufacturing bases, including India, China, and parts of Europe.
The competitive intensity is primarily driven by price, product quality, and supply chain reliability.
How are pricing trends evolving for Meropenem IV?
Pricing for Meropenem IV is predominantly influenced by generic competition and regional market dynamics.
- Price Erosion: The availability of multiple generic versions has led to significant price erosion since patent expiry. Prices vary considerably based on dosage, packaging, and regional purchasing power.
- Volume-Based Agreements: Large hospital systems and government tenders often negotiate volume-based discounts, further driving down average selling prices.
- Emerging Markets: While prices are generally lower in emerging markets due to cost sensitivities and competition, the increasing burden of infections can drive higher volume sales.
- Specialty Formulations: While meropenem is predominantly a generic product, some niche formulations or combination products, if they emerge, could command premium pricing. However, the core IV product is a commodity.
- Projected Price Trends: The downward pressure on prices is expected to continue, albeit at a moderating pace. Average wholesale prices for generic meropenem vials (e.g., 1g) typically range from $10 to $30 USD, depending on the manufacturer and market. In some tendering processes, prices can fall below $10 USD per vial for large volumes. Forecasts suggest a continued slight decline in average selling prices over the next five years, with an estimated 1-2% annual decrease in real terms due to ongoing generic competition.
What is the geographical segmentation of the Meropenem IV market?
The Meropenem IV market is segmented globally, with significant variations in market size and growth drivers.
| Region | Market Share (2023 Est.) | Projected Growth (CAGR 2024-2029) | Key Drivers |
|---|---|---|---|
| North America | 25% | 2.8% | High prevalence of HAIs, robust healthcare infrastructure, established guidelines |
| Europe | 20% | 3.0% | Aging population, strong antibiotic stewardship, evolving resistance patterns |
| Asia Pacific | 35% | 4.5% | Growing healthcare expenditure, increasing incidence of infections, large patient pool |
| Latin America | 10% | 3.5% | Improving access to healthcare, rising infectious disease burden |
| Middle East & Africa | 10% | 3.8% | Developing healthcare systems, significant infectious disease challenges |
Source: Internal analysis based on industry reports and market intelligence.
- Asia Pacific: Dominates the market due to its large population, increasing prevalence of hospital-acquired infections, and expanding healthcare infrastructure, particularly in countries like China and India, which are also major manufacturing hubs.
- North America and Europe: Mature markets with high demand driven by complex healthcare systems and awareness of antibiotic resistance, but growth is constrained by pricing pressures and established stewardship.
- Latin America, Middle East, and Africa: Exhibit higher growth potential due to improving healthcare access and a substantial burden of infectious diseases, though market penetration can be challenged by economic factors and regulatory complexities.
What is the impact of antibiotic resistance trends on Meropenem IV usage?
The escalating crisis of antimicrobial resistance directly influences the use and perceived value of meropenem.
- Increased Reliance for Resistant Pathogens: As resistance to earlier generations of antibiotics grows, meropenem is increasingly relied upon for treating infections caused by ESBL-producing Enterobacteriaceae and some Gram-negative organisms resistant to third-generation cephalosporins.
- Carbapenem Resistance as a Threat: The rise of carbapenem resistance, including the detection of carbapenemases like KPC, NDM, and OXA-48, reduces the efficacy of meropenem. This necessitates surveillance and susceptibility testing to ensure appropriate use [3].
- Shifting Treatment Paradigms: For infections caused by highly resistant organisms, meropenem may be used in combination with other agents or be replaced by newer antibiotics with specific activity against carbapenemase producers.
- Stewardship Impact: Antibiotic stewardship programs advocate for de-escalation of therapy once the causative pathogen and its susceptibility profile are known. This means meropenem might be used initially but switched to a narrower-spectrum agent if appropriate, impacting overall consumption.
What are the future outlook and opportunities for Meropenem IV?
Despite challenges, meropenem IV is expected to maintain its role as a critical antibiotic.
- Sustained Role in MDRO Management: Meropenem will continue to be a cornerstone therapy for many infections caused by multidrug-resistant Gram-negative bacteria where other options are limited.
- Emerging Markets Growth: The expansion of healthcare access and rising infection rates in emerging economies will drive volume growth.
- Potential for Combination Therapies: Future opportunities may lie in the development of fixed-dose combination products of meropenem with novel beta-lactamase inhibitors, extending its spectrum against carbapenemase-producing organisms. However, such development is costly and faces regulatory hurdles.
- Cost-Effectiveness: In a climate of healthcare cost containment, the cost-effectiveness of generic meropenem compared to newer, more expensive agents will ensure its continued use.
Key Takeaways
- The Meropenem IV market, valued at $950 million in 2023, is projected to reach $1.15 billion by 2029, growing at a 3.2% CAGR.
- Demand is driven by the prevalence of severe bacterial infections, including those caused by multidrug-resistant organisms (MDROs).
- Intense generic competition is the primary market restraint, leading to significant price erosion.
- The emergence of carbapenem resistance poses a direct threat to the drug's long-term utility.
- Asia Pacific is the largest and fastest-growing market segment for Meropenem IV.
- The drug will remain a critical therapeutic option for serious infections, particularly in resource-limited settings and for MDROs.
Frequently Asked Questions
- What is the primary mechanism of action for meropenem IV? Meropenem IV is a beta-lactam antibiotic that inhibits bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs). This leads to cell lysis and bacterial death.
- How does meropenem IV compare to other carbapenems like imipenem? Meropenem generally has a broader spectrum of activity against Gram-negative bacteria than imipenem and is less susceptible to degradation by renal dehydropeptidase I, eliminating the need for a dehydropeptidase inhibitor like cilastatin when administered alone. However, it may have slightly less activity against certain Gram-positive organisms.
- What are the most common side effects associated with meropenem IV administration? Common side effects include nausea, vomiting, diarrhea, headache, and rash. More serious, but less common, side effects can include seizures, Clostridioides difficile-associated diarrhea, and hypersensitivity reactions.
- Is meropenem IV effective against all forms of bacterial resistance? No, meropenem IV is not effective against all forms of bacterial resistance. Its efficacy is compromised by the production of certain carbapenemase enzymes (e.g., KPC, NDM, OXA-48) by bacteria, leading to carbapenem resistance.
- What is the typical duration of meropenem IV treatment? The duration of meropenem IV treatment varies depending on the type and severity of the infection, the causative pathogen, and the patient's clinical response. It typically ranges from 7 to 14 days for complicated infections, but can be shorter or longer as clinically indicated.
Citations
[1] European Centre for Disease Prevention and Control. (2023). Antimicrobial resistance in the EU/EEA: Annual epidemiological report for 2022. Publications Office of the European Union. [2] Logan, L. K., & Weinstein, R. A. (2017). The epidemiology of carbapenem-resistant Enterobacteriaceae: The impact and evolution of a global menace. Journal of Infectious Diseases, 215(suppl_1), S28-S36. [3] World Health Organization. (2020). Global priority list of antibiotic-resistant bacteria to guide research and development of new antibiotics. World Health Organization.
More… ↓
