Share This Page
Drug Price Trends for MEPROBAMATE
✉ Email this page to a colleague
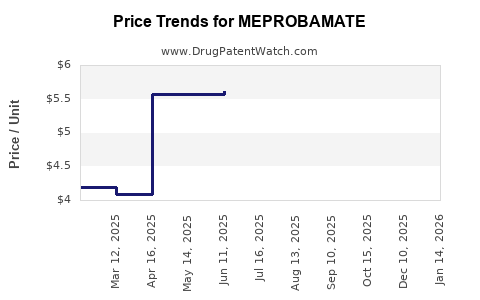
Average Pharmacy Cost for MEPROBAMATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MEPROBAMATE 200 MG TABLET | 69097-0974-07 | 4.79569 | EACH | 2026-05-20 |
| MEPROBAMATE 400 MG TABLET | 62332-0020-31 | 5.86549 | EACH | 2026-05-20 |
| MEPROBAMATE 200 MG TABLET | 62332-0019-31 | 4.79569 | EACH | 2026-05-20 |
| MEPROBAMATE 400 MG TABLET | 69097-0975-07 | 5.86549 | EACH | 2026-05-20 |
| MEPROBAMATE 200 MG TABLET | 62332-0019-31 | 4.93396 | EACH | 2026-04-22 |
| MEPROBAMATE 400 MG TABLET | 62332-0020-31 | 5.74865 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Meprobamate
What is Meprobamate and its current market status?
Meprobamate is an anxiolytic and anticonvulsant agent historically used in some countries for anxiety, insomnia, and epilepsy. It is not widely marketed outside limited regions due to safety concerns and the availability of alternative therapies. Despite its age, there is potential interest driven by unmet needs in specific neurological and psychiatric conditions. Current global production and distribution are limited, with primary formulations available mainly in select markets.
Regulatory and patent landscape
Patent expiration and regulatory status
- No recent patents filed; existing patents expired in the early 2000s in major markets like the US and Europe.
- Regulatory approval status varies; in some jurisdictions, Meprobamate is not approved, or approvals have been withdrawn due to safety and efficacy concerns.
- In countries where it is approved, it has not experienced major regulatory barriers recently, implying a potential for generic manufacturing.
Market withdrawals and discontinuations
- Declined due to safety issues linked to hepatotoxicity and hematological side effects.
- Not designated as a controlled substance in major markets; no current restrictions impede manufacturing or sales.
Market drivers and barriers
Drivers
- Growing elderly patient populations with comorbid anxiety and epilepsy.
- Increasing interest in repurposing well-characterized compounds with established safety profiles for niche indications.
- Limited current therapies for certain refractory or comorbid conditions.
Barriers
- Safety concerns limit renewed adoption.
- Competition from newer, better-tolerated drugs such as benzodiazepines, SSRIs, and newer anticonvulsants.
- Lack of recent clinical data or trials supporting efficacy in current indications.
Competitive landscape
| Drug Class | Major Drugs | Market Share (Estimated) | Notes |
|---|---|---|---|
| Benzodiazepines | Diazepam, Lorazepam | Dominates anxiety treatment | Safety profile concerns slow growth here |
| SSRIs | Sertraline, Escitalopram | Leading depression and anxiety | High tolerability, widespread use |
| New anticonvulsants | Levetiracetam, Lamotrigine | Epilepsy treatment | Favorable safety profiles |
Meprobamate's niche could be as an alternative in specific populations, but its market share remains negligible.
Price history and projections
Historical pricing
- No publicly available recent pricing data due to limited market presence.
- Imported generic formulations in some regions (e.g., Eastern Europe) priced at approximately $0.05–$0.10 per pill (assuming 200 mg strength).
Price projections
- If reintroduced in a niche market with a regulatory pathway, initial wholesale prices could be $1–$3 per unit, reflecting risks and safety monitoring costs.
- For a broad reintroduction with optimized manufacturing and regulatory approval, prices could stabilize around $0.50–$1 per pill, similar to older anticonvulsants.
Factors influencing future prices
- Regulatory approval status and safety profile enhancements.
- Competition entry with newer drugs.
- Manufacturing costs and supply chain stability.
- Market demand from aging populations with refractory conditions.
Potential for reintroduction and market entry strategies
- Fragmented production implies opportunity for generic manufacturers.
- Safety concerns could be mitigated via formulation changes or new clinical data.
- A targeted marketing approach in regions with less competition and unmet neurological or psychiatric needs could facilitate market entry.
Key considerations
- Significant regulatory hurdles due to historical safety issues.
- Limited current clinical data diminishes immediate commercial appeal.
- A niche reintroduction would require demonstration of improved safety or efficacy.
Summary
Meprobamate remains a low-profile agent with historical use limited by safety concerns. Market potential hinges on addressing safety issues, gaining regulatory approval, and identifying niche indications. Price points are likely to be modest initially, with potential growth if the drug is repositioned with a favorable safety profile.
Key Takeaways
- Meprobamate’s market is inactive currently, with little recent sales data.
- Price projections depend on regulatory approval, safety improvements, and niche opportunities.
- Ongoing competition from better-tolerated drugs limits broad reintroduction potential.
- Generic manufacturing could lower entry barriers if safety concerns are addressed.
- The main opportunity lies in targeted applications with unfulfilled needs.
FAQs
1. What safety concerns limit Meprobamate’s use?
Hepatotoxicity and blood dyscrasias linked to its historical use caused withdrawal in multiple markets.
2. Could Meprobamate be repositioned as a safer alternative?
Potentially, if new formulations or clinical data demonstrate improved safety or efficacy.
3. How does Meprobamate compare to benzodiazepines?
It shares anxiolytic properties but has a less favorable safety profile and is less widely used.
4. What regions could allow reintroduction?
Eastern Europe and some Asian countries with less stringent regulatory environments may permit limited reintroduction.
5. What is the future outlook for Meprobamate?
Limited, unless safety profile issues are mitigated and targeted clinical trials support new indications.
References
- Federal Drug Administration. (2000). Withdrawal of Meprobamate for safety concerns. FDA Notices.
- European Medicines Agency. (2002). Safety review of anticonvulsants including Meprobamate.
- Marketline. (2022). Global pharmaceutical market data.
More… ↓
