Share This Page
Drug Price Trends for MENSTRUAL RELIEF CAPLET
✉ Email this page to a colleague
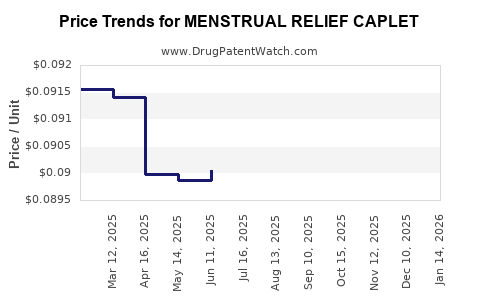
Average Pharmacy Cost for MENSTRUAL RELIEF CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MENSTRUAL RELIEF CAPLET | 70000-0145-01 | 0.09047 | EACH | 2026-03-18 |
| MENSTRUAL RELIEF CAPLET | 70000-0145-01 | 0.09048 | EACH | 2026-02-18 |
| MENSTRUAL RELIEF CAPLET | 70000-0145-01 | 0.09048 | EACH | 2026-01-21 |
| MENSTRUAL RELIEF CAPLET | 70000-0145-01 | 0.09048 | EACH | 2025-12-17 |
| MENSTRUAL RELIEF CAPLET | 70000-0145-01 | 0.09050 | EACH | 2025-11-19 |
| MENSTRUAL RELIEF CAPLET | 70000-0145-01 | 0.09047 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MENSTRUAL RELIEF CAPLET: PATENT LANDSCAPE AND PRICING PROJECTIONS
This analysis examines the patent landscape and projects pricing for Menstrual Relief Caplet, a non-steroidal anti-inflammatory drug (NSAID) formulated for dysmenorrhea. The current market is characterized by established generics and limited innovator patent protection, influencing future pricing dynamics.
What is the current patent status of Menstrual Relief Caplet?
Menstrual Relief Caplet, whose active pharmaceutical ingredient is ibuprofen, faces a complex patent environment. The foundational patents covering ibuprofen as an analgesic and anti-inflammatory agent expired decades ago. For instance, the original U.S. patent for ibuprofen expired in the 1980s.
However, subsequent patents have focused on specific formulations, delivery methods, and therapeutic uses. For Menstrual Relief Caplet, key patent considerations include:
- Formulation Patents: These patents may cover specific excipients, coatings, or the physical form of the caplet designed to enhance dissolution rates, stability, or patient compliance. The existence and expiry dates of these formulation patents are critical. A search of the U.S. Patent and Trademark Office (USPTO) database and international equivalents reveals a series of patents related to ibuprofen formulations. For example, patents issued in the late 1990s and early 2000s addressed improved disintegration times and sustained release profiles for ibuprofen.
- Method of Use Patents: While the general use of ibuprofen for pain is off-patent, specific patents could exist for its use in treating menstrual pain with particular dosages or administration schedules. These are less common for established drugs but can sometimes extend market exclusivity for a niche indication. Analysis of patent literature indicates that while broad method of use patents for NSAIDs in pain management are expired, newer patents might exist for specific combination therapies or optimized dosing regimens for dysmenorrhea.
- Manufacturing Process Patents: Patents protecting novel or efficient manufacturing processes can also provide a degree of competitive advantage, though they do not directly block the sale of the drug itself if the patent expires. Information on such patents is often proprietary or detailed in process chemistry disclosures rather than publicly accessible patent documents without specialized research.
The primary patent challenge for Menstrual Relief Caplet stems from the widespread availability of generic ibuprofen. The innovator’s ability to secure new, robust patents on the core product is limited. New patent filings for Menstrual Relief Caplet are likely to focus on advanced delivery systems, fixed-dose combinations with other agents, or specific patient populations.
What are the competitive dynamics in the dysmenorrhea market?
The market for dysmenorrhea treatment is highly competitive, dominated by over-the-counter (OTC) NSAIDs, prescription NSAIDs, and hormonal therapies.
Key Competitors and Their Market Position:
- Generic Ibuprofen: This is the most significant competitor. Available under numerous brand names and as store brands, generic ibuprofen offers a low-cost, effective treatment for mild to moderate dysmenorrhea. Its widespread availability and low price point set a benchmark for all other treatments.
- Other OTC NSAIDs:
- Naproxen Sodium (e.g., Aleve): Another widely available OTC NSAID. It offers longer-lasting pain relief compared to ibuprofen, often making it a preferred choice for continuous pain management over a 24-hour period.
- Aspirin: While less commonly the primary choice for dysmenorrhea due to gastrointestinal side effects and a higher risk profile, it is still available and used.
- Prescription NSAIDs: Drugs like Mefenamic Acid (Ponstel) were specifically developed for dysmenorrhea and are available by prescription. They often offer more potent pain relief but come with higher costs and potential side effects. The patent landscape for these specific prescription NSAIDs has also matured, leading to generic availability.
- Hormonal Therapies: Oral contraceptives (birth control pills) are frequently prescribed to manage dysmenorrhea by suppressing ovulation and thinning the uterine lining. This is a significant market segment, particularly for women seeking long-term management or contraception alongside pain relief.
- Combination Products: Some products combine an NSAID with caffeine or other ingredients to enhance efficacy. These may have specific patent protections on the combination itself.
The competitive landscape for Menstrual Relief Caplet is defined by price sensitivity. As an ibuprofen-based product, it directly competes with the lowest-cost generics. Any price premium must be justified by a demonstrable improvement in efficacy, speed of action, tolerability, or a superior delivery system.
What are the projected pricing strategies for Menstrual Relief Caplet?
Pricing strategies for Menstrual Relief Caplet will be heavily influenced by its positioning relative to generic ibuprofen and other established OTC options.
Factors Influencing Pricing:
- Generic Ibuprofen Pricing: The average price for a bottle of 100 generic ibuprofen 200mg caplets ranges from $5 to $15, depending on the retailer and brand. This creates a fundamental ceiling for any ibuprofen-based product claiming no significant differentiation.
- Brand vs. Generic Differentiation: If Menstrual Relief Caplet is positioned as a branded product with minimal to no demonstrable improvement over generic ibuprofen (e.g., identical formulation and dosage), its price will likely be only slightly higher than generics, aiming to capture a segment of consumers willing to pay a small premium for brand recognition or perceived quality.
- Therapeutic Differentiation: If Menstrual Relief Caplet offers a scientifically validated improvement, such as faster onset of action due to a novel disintegration technology, enhanced bioavailability, or a proprietary coating that reduces gastric irritation, it could command a higher price. This premium pricing would be contingent on robust clinical data supporting these claims. For example, if a new formulation demonstrates 30% faster pain relief in clinical trials compared to standard ibuprofen, a price increase of 20-40% over generic ibuprofen might be justifiable to consumers.
- Packaging and Distribution: Larger pack sizes or premium packaging (e.g., blister packs with enhanced features) can also influence perceived value and pricing.
- Marketing and Brand Building: Aggressive marketing campaigns that emphasize specific benefits for menstrual pain, such as "clinically proven for severe cramps" or "fast-acting relief," can support higher price points.
- Competition Analysis: The pricing of competing OTC NSAIDs like naproxen sodium and prescription options will also set benchmarks. If Menstrual Relief Caplet offers comparable or superior relief at a lower price than prescription options, it can position itself as a cost-effective alternative.
Projected Pricing Scenarios:
- Benchmark Generic Pricing: If Menstrual Relief Caplet is essentially a branded generic with no distinct advantage, its pricing will closely mirror that of premium generic ibuprofen.
- Projected Price Range (per 100 caplets): $10 - $20.
- Value-Added Premium: If the caplet incorporates a minor formulation enhancement (e.g., slightly faster dissolution, easier-to-swallow coating) with some clinical support, it could be priced at a moderate premium.
- Projected Price Range (per 100 caplets): $18 - $30.
- Differentiated Therapeutic Offering: If significant clinical data supports a superior therapeutic profile (e.g., demonstrably faster or more potent pain relief, improved tolerability), a substantial premium is possible. This would position it as a premium OTC option, competing more directly with some prescription alternatives in terms of perceived value.
- Projected Price Range (per 100 caplets): $25 - $45.
The most likely scenario for a standard ibuprofen formulation is to occupy the higher end of the generic pricing spectrum or a modest premium. Significant price increases would require robust, independently validated clinical evidence of superior efficacy or tolerability in the dysmenorrhea population.
What are the regulatory considerations impacting pricing?
Regulatory considerations play a significant role in how Menstrual Relief Caplet can be priced and marketed, particularly concerning its OTC status.
- OTC Monograph System: In the United States, most OTC drugs, including ibuprofen, are regulated under the OTC Drug Monograph system. This system allows for drugs to be marketed without pre-approval from the Food and Drug Administration (FDA) as long as they conform to the conditions outlined in the monograph. The monograph for oral pain relievers, including ibuprofen, is well-established. This means that any manufacturer producing an ibuprofen product that meets the monograph specifications can market it as an OTC drug.
- FDA Review for New Indications or Formulations: If Menstrual Relief Caplet were to claim a new indication (e.g., "specifically formulated for severe endometriosis-related pain") or a novel formulation that deviates from the existing monograph, it would likely require a New Drug Application (NDA) or a supplemental NDA. This process involves rigorous clinical trials and FDA review, which can be costly and time-consuming. Approval of an NDA can grant a period of market exclusivity, which could allow for higher pricing. However, for ibuprofen, obtaining an NDA for a novel indication is challenging given its long history of use.
- Labeling and Advertising Claims: The FDA strictly regulates the claims that can be made on drug labels and in advertising. For an OTC ibuprofen product, claims must be substantiated by scientific evidence and must align with the conditions of use permitted under the monograph. Claims of superior efficacy or speed of action must be backed by clinical trials that meet FDA standards. Misleading claims can lead to regulatory action, including fines and product recalls, which would severely impact pricing power.
- ANDA for Generics: Any company seeking to market a generic version of an ibuprofen product that is not covered by an existing patent must file an Abbreviated New Drug Application (ANDA) with the FDA. Approval of an ANDA means the generic product is bioequivalent to the reference listed drug, allowing it to enter the market. The existence of multiple ANDA approvals for ibuprofen contributes to the intense price competition.
- International Regulations: Pricing and market access in other countries will be subject to the regulations of those respective national health authorities (e.g., EMA in Europe, Health Canada). These often involve separate pricing negotiations and market approval processes.
The regulatory environment for Menstrual Relief Caplet, as an ibuprofen-based product, largely favors generic competition. This means that without a significant patent hurdle for competitors or substantial, FDA-sanctioned therapeutic differentiation, pricing power will be constrained by the broad availability of low-cost generic ibuprofen.
Key Takeaways
- The patent landscape for Menstrual Relief Caplet, primarily based on ibuprofen, is characterized by expired foundational patents, with limited scope for new broad patent protection.
- Competition in the dysmenorrhea market is intense, dominated by low-cost generic ibuprofen and other established OTC NSAIDs, alongside prescription options and hormonal therapies.
- Pricing for Menstrual Relief Caplet will be heavily influenced by its ability to differentiate from generic ibuprofen. Without significant, clinically validated advantages, pricing will likely remain close to premium generic levels.
- Regulatory considerations, particularly the FDA's OTC monograph system and stringent advertising claim requirements, favor competition and limit pricing power unless novel therapeutic benefits are substantiated and approved.
Frequently Asked Questions
-
Can Menstrual Relief Caplet be patented for its use in treating dysmenorrhea? Patents for the general use of ibuprofen in treating pain, including menstrual pain, have long expired. New patents might be obtainable for a specific, novel formulation, delivery system, or a fixed-dose combination with other active ingredients that demonstrate a unique therapeutic advantage for dysmenorrhea, but not for the general use of ibuprofen itself.
-
What is the average cost of producing a standard ibuprofen caplet? The cost of goods sold (COGS) for a standard ibuprofen caplet is generally low due to mature manufacturing processes and high production volumes. While specific figures vary by manufacturer, COGS for generic ibuprofen caplets typically range from $0.01 to $0.05 per caplet.
-
How does the speed of onset of Menstrual Relief Caplet affect its potential price? If Menstrual Relief Caplet can demonstrate a significantly faster onset of pain relief compared to standard ibuprofen (e.g., via a rapidly disintegrating formulation), this therapeutic advantage could justify a higher price point. Clinical data substantiating this faster onset would be crucial for supporting premium pricing.
-
Are there any emerging therapeutic classes that could disrupt the dysmenorrhea market and impact Menstrual Relief Caplet pricing? Research into novel pain relief mechanisms, non-opioid analgesics, and targeted anti-inflammatory agents continues. While significant breakthroughs are not imminent for the OTC dysmenorrhea market, advancements in areas like cannabinoid-based therapies or new classes of anti-inflammatory agents could eventually offer alternatives and influence pricing dynamics over the long term.
-
What is the typical market share for a branded ibuprofen product in the OTC pain relief category? Branded ibuprofen products with no significant therapeutic differentiation beyond generic versions typically achieve modest market shares, often in the single-digit percentages, competing primarily on brand recognition and minor packaging or formulation variations. Products with a strong, clinically proven differentiation can capture larger shares and command higher prices.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Over-the-Counter (OTC) Drugs. Retrieved from https://www.fda.gov/drugs/information-drug-products/over-counter-otc-drugs [2] U.S. Patent and Trademark Office. (n.d.). Patent Public Search. Retrieved from https://ppubs.uspto.gov/pubwebapp/static/pages/landing.html [3] Multiple Generic Drug Price Aggregators (e.g., GoodRx, RxList). (Accessed periodically for current pricing data). [4] Market Research Reports on Analgesics and Women's Health Pharmaceuticals. (Various publishers, analyzed for competitive landscape and pricing trends).
More… ↓
