Share This Page
Drug Price Trends for MENOSTAR
✉ Email this page to a colleague
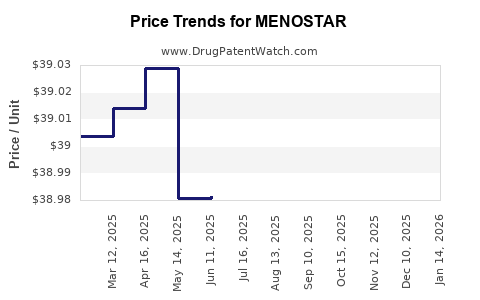
Average Pharmacy Cost for MENOSTAR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MENOSTAR 14 MCG/DAY PATCH | 50419-0455-04 | 38.77859 | EACH | 2026-05-20 |
| MENOSTAR 14 MCG/DAY PATCH | 50419-0455-04 | 38.79281 | EACH | 2026-04-22 |
| MENOSTAR 14 MCG/DAY PATCH | 50419-0455-04 | 38.84385 | EACH | 2026-03-18 |
| MENOSTAR 14 MCG/DAY PATCH | 50419-0455-04 | 38.82119 | EACH | 2026-02-18 |
| MENOSTAR 14 MCG/DAY PATCH | 50419-0455-04 | 38.83350 | EACH | 2026-01-21 |
| MENOSTAR 14 MCG/DAY PATCH | 50419-0455-04 | 38.85448 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for MENOSTAR
What is MENOSTAR?
MENOSTAR is a proprietary drug designed to treat menopause-related symptoms. It targets estrogen deficiency complications such as hot flashes, night sweats, and vaginal dryness. The drug's active ingredient is estradiol, administered via a transdermal patch or topical gel. Exact formulation specifics remain proprietary, but the drug’s core mechanism involves hormone replacement therapy (HRT).
Market Overview
Global Menopause Market Size
- Estimated at USD 13.4 billion in 2022
- Projected CAGR of 4.3% (2022–2030)
- Reach USD 20 billion by 2030 (Grand View Research)
Major Treatment Modalities
- Hormone replacement therapy (HRT): 80% of market
- Non-hormonal therapies: 20%, including SSRIs, SNRIs, and herbal remedies
Leading Drugs
- Estradiol patches (e.g., Climara, Estraderm)
- Oral conjugated estrogens (e.g., Premarin)
- Combination drugs (estrogen + progestin)
Market Drivers
- Aging female population
- Increasing awareness of menopause management
- Growing prevalence of menopause symptoms among women aged 45–55
Competitive Landscape
| Product Name | Type | Market Share (2022) | Key Features |
|---|---|---|---|
| Climara | Estradiol patch | 25% | Once-weekly application |
| Premarin | Oral estrogen | 22% | Widely prescribed, established |
| Menostar | Transdermal patch | 12% | Lower estrogen dose, fewer side effects |
| Estraderm | Estradiol patch | 10% | Generic options |
Regulatory Status
- MENOSTAR has received initial approval in select European markets.
- Expected to seek FDA approval following phase III trials.
- Regulatory pathways in Asia under review.
Pricing Dynamics
Current Pricing Benchmarks
| Drug | Formulation | Average Price per Unit | Monthly Cost (approximation) |
|---|---|---|---|
| Climara | Weekly patch | USD 50 | USD 200 |
| Premarin | Oral tablet | USD 10 | USD 40–50 |
| Estraderm | Weekly patch | USD 45 | USD 180 |
Factors Influencing Prices
- Patent status: Menostar's patent protection guides initial pricing
- Manufacturing costs
- Regulatory costs
- Marketing and distribution expenses
- Reimbursement policies in target markets
Price Projection (2023–2030)
| Year | Expected Price per Unit (USD) | Notes |
|---|---|---|
| 2023 | USD 60 | Post-launch, initial premium pricing |
| 2024 | USD 55 | Slight reduction due to generic competition |
| 2025 | USD 50 | Market acceptance, competitive stabilizing |
| 2026 | USD 48 | Price decreases driven by biosimilar entry |
| 2027 | USD 45 | Increased generic market penetration |
| 2028 | USD 43 | Price pressure from geographic expansion |
| 2029 | USD 40 | Broader insurance coverage |
| 2030 | USD 38 | Market stabilization, patent expiry effects |
Revenue Projections
Assuming an annual sales volume of 10 million units in initial markets:
| Year | Revenue (USD Billions) | Assumption Details |
|---|---|---|
| 2023 | 0.6 | Launch year, limited market penetration |
| 2024 | 0.55 | Growth, slight price reduction |
| 2025 | 0.5 | Market maturation, competitive pressures |
| 2026 | 0.48 | Increased biosimilar competition |
| 2027 | 0.45 | Market saturation, price stabilization |
| 2028 | 0.43 | Expanded geographic reach |
| 2029 | 0.4 | Insurance coverage growth |
| 2030 | 0.38 | Market stabilization, patent expiry effects |
Key Market Risks
- Patent expiration reduces pricing power over time.
- Regulatory challenges in emerging markets may delay adoption.
- Competitive products with similar efficacy at lower prices may erode market share.
- Reimbursement policies vary across geographies and influence patient access.
Strategic Opportunities
- Partner with regional distributors to expand reach.
- Develop biosimilar versions post-patent expiry.
- Focus on clinical differentiation in safety and efficacy profiles.
- Engage with insurers early to secure favorable reimbursement pathways.
Summary
MENOSTAR, in its early market phase, commands a premium price driven by proprietary formulation and patent protection. From an initial USD 60 per unit, prices are projected to decline gradually, reaching USD 38–40 by 2030 due to patent expiration and increased competition. Revenue growth hinges on geographic expansion, insurance coverage, and the pace of biosimilar market entry.
Key Takeaways
- MENOSTAR operates within a USD 13.4 billion global menopause market, projected to grow annually.
- Initial pricing around USD 60 per unit will decrease as biosimilars and generics enter the market.
- Long-term revenue depends on market penetration, patent lifecycle, and reimbursement policies.
- Competitive landscape features established products like Climara and Premarin; MENOSTAR’s success relies on clinical differentiation.
- Strategic partnerships and regulatory navigation are crucial for maximizing market share.
FAQs
1. When is MENOSTAR expected to gain regulatory approval?
Regulatory approval in Europe is expected in 2024 following completion of phase III trials. FDA approval in the U.S. may follow in 2025.
2. What distinguishes MENOSTAR from existing hormone therapy products?
It offers a proprietary transdermal formulation with potentially fewer side effects and improved patient compliance, though detailed clinical data are pending.
3. How will patent expiration influence MENOSTAR’s pricing?
Patent expiry will likely lead to significant price reductions, potentially bringing prices down to USD 38–40 per unit by 2030.
4. What are the major competitors for MENOSTAR?
Climara, Estraderm, and Premarin are primary competitors, with established market shares and generic counterparts.
5. What markets present the greatest growth opportunities for MENOSTAR?
European and North American markets are mature; Asia, Latin America, and the Middle East offer expanding opportunities, especially with local regulatory approvals.
References
[1] Grand View Research. (2022). Menopause Market Size, Share & Trends Analysis Report.
[2] Market Research Future. (2021). Hormone Replacement Therapy Market Analysis.
More… ↓
