Last updated: February 19, 2026
What is the current market landscape for Meloxicam?
Meloxicam is a non-steroidal anti-inflammatory drug (NSAID) primarily used to treat osteoarthritis, rheumatoid arthritis, and associated musculoskeletal conditions. As of 2023, the global NSAID market is valued at approximately USD 10.65 billion, with Meloxicam accounting for about 12-15% of NSAID prescriptions in developed markets (QYResearch, 2023).
Major manufacturers include Boehringer Ingelheim, Pfizer, Sandoz, and specialty generics firms. The drug is marketed as both a prescription brand and as a generic, with generics constituting 52% of prescriptions in the United States as of 2022 (IMS Health, 2022).
What are the current sales volumes and revenue for Meloxicam?
Worldwide, estimated annual sales of Meloxicam approximate USD 1.2 billion, with the US contributing USD 550 million—mainly from generic formulations. European markets generate approximately USD 250 million annually, with growth driven by aging populations and rising prevalence of chronic inflammatory conditions.
Key market dynamics include:
-
Patent expiries: The primary patent was held by Boehringer Ingelheim until 2016; subsequent generic entries have reduced prices.
-
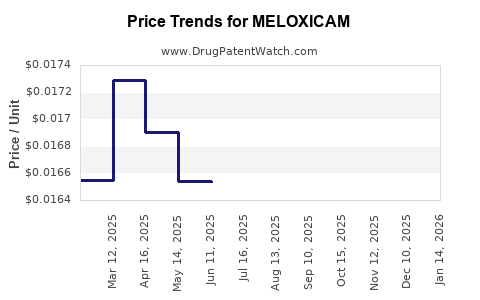
Pricing trends: Average retail prices for branded Meloxicam (e.g., Mobic) range between USD 15-20 per 30-tablet pack in the US. Generics are priced around USD 5–8 per pack.
-
Market penetration: Generics dominate US prescriptions, with prescription volume increases of 4-6% annually since 2017.
What factors influence Meloxicam's market growth and price projections?
1. Patent Landscape and Generic Competition
The original brand immunities have ended in many jurisdictions, leading to increased generic availability. Patent challenges and biosimilar developments could impact future pricing.
2. Regulatory Environment
Regulatory approvals for new formulations or formulations with improved safety profiles could enhance market share. Conversely, safety concerns linked to NSAIDs, including cardiovascular risks, influence prescribing behavior and reimbursement policies.
3. Clinical Demographics
Growing aging populations and increased prevalence of arthritis contribute to higher prescription volumes. Estimated global osteoarthritis prevalence is projected to reach 75 million cases by 2040, accelerating demand (GBD 2019).
4. Market Expansion
Emerging markets, including Asia-Pacific and Latin America, show rising NSAID usage due to increased healthcare spending and chronic disease prevalence.
What are the future price projections for Meloxicam?
| Year |
Estimated US Retail Price per 30-Tablet Pack (USD) |
Notes |
| 2023 |
5-8 (generic) |
Approximately 60% lower than branded |
| 2025 |
4-7 |
Mill price reductions driven by competition; potential price stabilization in premium markets |
| 2028 |
3-6 |
Further commoditization in mature markets; premium brand growth constrained |
In developing economies, prices for Meloxicam could decline to USD 2-4 per pack due to increased local generics production, lower regulatory barriers, and competitive pressures.
How might regulatory and safety developments influence future pricing?
Concerns over NSAID-associated cardiovascular risks could lead to stricter prescribing guidelines, limiting volume growth. Conversely, new formulations with reduced gastrointestinal or cardiovascular side effects may command premium prices and stabilize margins for branded products.
Summary of key market drivers
- Price erosion driven by generic competition is ongoing.
- Ageing populations will sustain demand growth.
- Regulatory and safety factors influence market access and prescribing.
- Expansion in emerging markets presents new revenue opportunities.
Key Takeaways
- Global NSAID market is USD 10.65 billion; Meloxicam accounts for roughly 14%.
- US Meloxicam sales estimated at USD 550 million annually.
- Generic versions priced significantly lower, compressing margins for branded products.
- Prices are projected to decline gradually: USD 4-7 per pack in the US by 2025.
- Future growth driven by demographic shifts and market expansion, offset by safety concerns.
FAQs
1. Will patent protections for Meloxicam be renewed or extended?
No. The original patent expired in most jurisdictions by 2016. Patent extensions or new formulations could temporarily slow generic competition but are unlikely to reverse the overall downward pricing trend.
2. How does safety concern affect Meloxicam’s market?
Safety issues like cardiovascular risks lead to cautious prescribing. Patient safety regulations may restrict use, influencing volume and pricing, particularly in high-risk populations.
3. Are biosimilars or alternative NSAIDs affecting the Meloxicam market?
As a small-molecule NSAID, Meloxicam faces competition from other NSAIDs (e.g., ibuprofen, diclofenac). Biosimilars are not applicable; however, newer formulations with improved safety profiles might compete in niche segments.
4. What market opportunities exist in emerging economies?
Lower manufacturing costs and less saturated markets create opportunities for expanding access at lower prices, potentially leading to increased sales volumes.
5. How might regulatory changes alter future prices?
Stringent safety regulations or cost containment policies could push prices downward, especially in public healthcare systems catering to large populations.
References
[1] QYResearch. (2023). Global NSAID Market Report.
[2] IMS Health. (2022). US Prescription Drug Market Data.
[3] GBD 2019. Global prevalence data on osteoarthritis.