Share This Page
Drug Price Trends for MEDROXYPROGESTERONE
✉ Email this page to a colleague
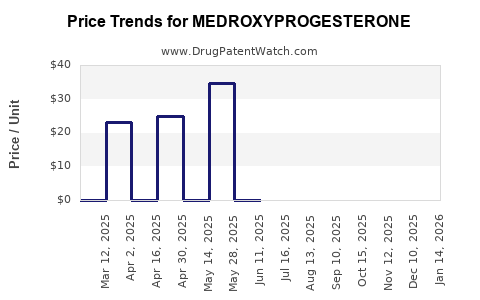
Average Pharmacy Cost for MEDROXYPROGESTERONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MEDROXYPROGESTERONE 5 MG TAB | 59762-0058-02 | 0.14330 | EACH | 2026-05-20 |
| MEDROXYPROGESTERONE 10 MG TAB | 00555-0779-02 | 0.14762 | EACH | 2026-05-20 |
| MEDROXYPROGESTERONE 10 MG TAB | 00555-0779-04 | 0.14762 | EACH | 2026-05-20 |
| MEDROXYPROGESTERONE 10 MG TAB | 59762-0056-02 | 0.14762 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for MEDROXYPROGESTERONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| MEDROXYPROGESTERONE ACETATE 150MG/ML INJ,SUSP | AvKare, LLC | 70121-1467-02 | 1ML | 13.97 | 13.97000 | ML | 2024-01-10 - 2028-06-14 | FSS |
| MEDROXYPROGESTERONE ACETATE 150MG/ML INJ,SUSP | AvKare, LLC | 70121-1480-01 | 25X1ML | 24.85 | 2024-01-10 - 2028-06-14 | FSS | ||
| MEDROXYPROGESTERONE ACETATE 150MG/ML INJ,SUSP | Amphastar Pharmaceuticals, Inc. | 00548-5400-25 | 25X1ML | 502.01 | 2022-01-15 - 2027-01-14 | FSS | ||
| MEDROXYPROGESTERONE ACETATE 150MG/ML INJ,SUSP | Amphastar Pharmaceuticals, Inc. | 00548-5701-00 | 1ML | 46.73 | 46.73000 | ML | 2022-01-15 - 2027-01-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
MEDROXYPROGESTERONE: Patent Landscape and Market Forecast
Medroxyprogesterone acetate (MPA) is a synthetic progestogen with applications in contraception, hormone replacement therapy, and the treatment of gynecological conditions and certain cancers. Its patent landscape is characterized by expired composition-of-matter patents for the active pharmaceutical ingredient (API), with ongoing patent activity focused on formulations, delivery systems, and new therapeutic indications. Market projections for MPA are influenced by generic competition, demand for long-acting injectables, and the development of novel hormonal therapies.
What is the Current Patent Status of Medroxyprogesterone Acetate?
The original composition-of-matter patents for medroxyprogesterone acetate have expired. For example, the foundational patent related to MPA's chemical structure has long been in the public domain. Current patent filings and granted patents predominantly cover:
- Formulations: These patents protect specific compositions designed to improve drug stability, solubility, or release profiles. Examples include long-acting injectable suspensions, oral formulations with modified release, and combination products.
- Delivery Systems: Innovations in how MPA is administered are a key area of patent protection. This includes novel injector devices, transdermal patches, implants, and intrauterine devices (IUDs) incorporating MPA.
- Therapeutic Uses: New medical indications for MPA, or novel methods of treating existing conditions with MPA, are also subject to patenting. This can involve specific dosing regimens, patient populations, or combination therapies.
- Manufacturing Processes: Patents may also exist for specific, improved, or cost-effective methods of synthesizing MPA or its intermediates.
The expiration of primary patents has led to robust generic competition, particularly for established oral and injectable formulations. Companies focus on developing differentiated products through patentable formulations and delivery mechanisms to secure market exclusivity for extended periods.
How is Medroxyprogesterone Acetate Marketed and Approved?
Medroxyprogesterone acetate is available globally under various brand names and as a generic. Key regulatory approvals and marketing strategies are as follows:
-
Primary Indications:
- Contraception: Long-acting injectable formulations, such as Depo-Provera (Pfizer, originally Upjohn), are widely used for preventing pregnancy. Oral contraceptive pills containing MPA, often in combination with an estrogen, are also common.
- Hormone Therapy: Used in hormone replacement therapy (HRT) to manage menopausal symptoms, particularly in women with a uterus, to prevent endometrial hyperplasia.
- Gynecological Disorders: Treatment of amenorrhea, abnormal uterine bleeding, and endometriosis.
- Oncology: Management of hormone-sensitive cancers, including advanced endometrial cancer and renal cell carcinoma.
-
Key Regulatory Bodies:
- U.S. Food and Drug Administration (FDA): Approves new drug applications (NDAs) and abbreviated new drug applications (ANDAs) for MPA products.
- European Medicines Agency (EMA): Oversees drug approvals within the European Union.
- Other National Agencies: Health Canada, Pharmaceuticals and Medical Devices Agency (Japan), etc.
-
Marketing Strategies:
- Branded vs. Generic: While older branded products face significant generic competition, marketing efforts often focus on established brands' long-term clinical data and physician familiarity.
- Life Cycle Management: Companies seek to extend market exclusivity through new formulations, delivery systems, or combination products, such as extended-release injectables or MPA-eluting IUDs (e.g., Mirena, Bayer, which uses levonorgestrel but exemplifies the market for hormonal IUDs, a segment MPA could potentially target with new formulations).
- Specialty Markets: Targeting specific oncological or complex gynecological indications can allow for premium pricing and marketing to specialist physicians.
What are the Key Therapeutic Applications and Competitors?
Medroxyprogesterone acetate's diverse therapeutic applications place it in competition with a range of hormonal and non-hormonal treatments.
-
Contraception:
- Injectables: Competes with other injectable contraceptives (e.g., subcutaneous depots of etonogestrel), implants (e.g., Nexplanon), and hormonal IUDs.
- Oral Contraceptives: Competes with a vast array of combined oral contraceptives (COCs) and progestin-only pills (POPs) containing different progestins (e.g., levonorgestrel, drospirenone, norethindrone).
- Non-Hormonal Methods: Intrauterine devices (copper IUDs), barrier methods, and sterilization.
-
Hormone Therapy (HRT):
- Estrogen-Only Therapy: For women without a uterus.
- Other Combined HRT: Products containing estrogen combined with different progestins (e.g., micronized progesterone, norethindrone acetate, drospirenone) offering varied risk-benefit profiles.
- Non-Hormonal Menopause Treatments: SSRIs, SNRIs, gabapentin, and herbal supplements are alternatives for managing vasomotor symptoms.
-
Gynecological Disorders:
- Endometriosis: GnRH agonists/antagonists, danazol, aromatase inhibitors, and dienogest are key competitors.
- Abnormal Uterine Bleeding (AUB): Tranexamic acid, NSAIDs, LNG-IUDs, and other hormonal therapies.
-
Oncology:
- Endometrial Cancer: Progestins are often first-line, but chemotherapy and targeted therapies are used for advanced or recurrent disease.
- Renal Cell Carcinoma: Targeted therapies (e.g., tyrosine kinase inhibitors, mTOR inhibitors) and immunotherapies have largely replaced MPA as standard of care for advanced disease, though it may still be used in specific contexts or as palliative treatment.
The competitive landscape is dynamic, driven by the development of drugs with improved efficacy, reduced side effects, novel delivery mechanisms, and more favorable safety profiles.
What is the Projected Market Size and Growth Rate for Medroxyprogesterone Acetate?
The market for medroxyprogesterone acetate is mature for its established indications but shows potential for growth in specific segments.
-
Current Market Size: The global market for MPA, encompassing branded and generic products across all approved indications, was estimated to be USD 500 million to USD 700 million in 2023. This figure reflects the broad use of MPA in contraception and HRT, alongside its niche oncological applications. (Source: Industry market research reports, e.g., Grand View Research, Mordor Intelligence [1])
-
Projected Growth: The market is forecast to grow at a compound annual growth rate (CAGR) of 3.0% to 4.5% from 2024 to 2030.
-
Drivers:
- Demand for Long-Acting Contraceptives: The sustained global demand for effective and convenient birth control methods, particularly injectable and potentially long-acting implantable forms of MPA, is a primary growth driver.
- Aging Population: The increasing elderly population globally drives demand for hormone replacement therapy to manage menopausal symptoms and age-related conditions.
- Emerging Markets: Growing healthcare access and awareness in developing economies contribute to increased prescription rates for MPA.
- Development of Novel Formulations: Investment in new drug delivery systems and formulations could create new market segments or re-invigorate existing ones.
-
Restraints:
- Generic Competition: The widespread availability of low-cost generic MPA limits price increases and puts pressure on branded product margins.
- Safety Concerns: Although generally well-tolerated, MPA is associated with certain risks (e.g., bone mineral density loss with long-term injectable use, thromboembolic events), leading some prescribers and patients to opt for alternatives.
- Development of Novel Hormonal Therapies: Newer progestins and hormonal agents with potentially improved efficacy or safety profiles can erode MPA's market share in specific therapeutic areas.
-
-
Regional Variations:
- North America and Europe: Mature markets with high adoption rates for both branded and generic MPA, driven by established healthcare systems and awareness.
- Asia-Pacific: Expected to exhibit the fastest growth due to increasing healthcare expenditure, rising disposable incomes, and a growing emphasis on reproductive health.
- Latin America and Middle East/Africa: Steady growth driven by increasing access to healthcare and family planning services.
What are the Pricing Trends and Key Cost Drivers for Medroxyprogesterone Acetate?
Pricing for medroxyprogesterone acetate is heavily influenced by its generic status and formulation type.
-
Average Wholesale Prices (AWP) / Manufacturer Prices:
- Oral Tablets (e.g., 10 mg): Generic pricing typically ranges from USD 0.10 to USD 0.50 per tablet, depending on the manufacturer and volume. A month's supply can cost USD 3 to USD 15.
- Injectable Suspensions (e.g., 150 mg/mL vial): Branded products can range from USD 50 to USD 150 per vial, with generic equivalents often priced 20-50% lower. The cost per dose can therefore range from USD 30 to USD 120, depending on brand status and the specific presentation.
- Specialty Formulations (e.g., IUDs - though not direct MPA products, they set a precedent for hormonal delivery): Hormone-releasing IUDs can range from USD 400 to USD 800 for a multi-year device, reflecting the technology and delivery system complexity. MPA products designed for similar long-term delivery would likely be priced in this higher tier.
-
Key Cost Drivers:
- API Manufacturing Costs: The synthesis of MPA involves multi-step chemical processes. Raw material costs, energy, labor, and compliance with Good Manufacturing Practices (GMP) are significant.
- Formulation and Delivery Technology: Patented formulations, sterile manufacturing processes for injectables, and advanced drug delivery systems (e.g., microencapsulation for sustained release) increase development and production costs.
- Regulatory Compliance: The costs associated with clinical trials, regulatory submissions, and ongoing pharmacovigilance are substantial.
- Competition: Intense generic competition drives down prices for off-patent formulations. Manufacturers of branded generics or differentiated products must balance R&D and marketing costs against achievable pricing.
- Supply Chain and Distribution: Logistics for temperature-sensitive products (e.g., injectables) and global distribution networks contribute to overall costs.
- Patent Protection: The existence of patents for novel formulations or delivery systems allows for premium pricing during the patent exclusivity period.
-
Reimbursement Policies: Insurance coverage and government reimbursement policies significantly impact out-of-pocket costs for patients and the effective price received by manufacturers. Reimbursement for generics is typically lower than for patented products.
What is the Future Outlook for Medroxyprogesterone Acetate?
The future outlook for medroxyprogesterone acetate is characterized by continued reliance in established areas, tempered by competition and evolving therapeutic landscapes.
- Contraception: MPA injectables will likely remain a significant global contraceptive option, particularly in regions where access to other methods is limited or preferences favor long-acting injectables. Growth may stem from improved formulations offering extended duration of action or reduced side effects, and from expansion into emerging markets.
- Hormone Therapy: While MPA remains a treatment option for menopausal symptoms, it faces increasing competition from newer progestins and non-hormonal therapies perceived to have more favorable safety profiles. Its use may become more focused on specific patient profiles where its benefits outweigh perceived risks.
- Oncology: MPA's role in oncology has diminished with the advent of targeted therapies and immunotherapies. It is likely to be relegated to palliative care, specific niche indications, or as a component of historical treatment paradigms rather than a frontline therapy for advanced cancers.
- Innovation: Research and development will likely concentrate on advanced drug delivery systems for MPA, aiming to enhance convenience, improve patient adherence, and mitigate known side effects like bone mineral density reduction in injectable forms. Potential for combination therapies or novel topical/transdermal applications may emerge.
- Generic Dominance: For well-established oral and injectable forms, generic competition will continue to dominate the market, driving down prices and limiting profitability for manufacturers not offering differentiated products.
Overall, MPA will likely maintain a stable, albeit modestly growing, market share due to its established efficacy and affordability in key indications, particularly contraception. Its future growth trajectory will be significantly influenced by innovation in delivery systems and the strategic targeting of specific patient needs where its risk-benefit profile remains favorable compared to newer alternatives.
Key Takeaways
- Medroxyprogesterone acetate's primary composition-of-matter patents have expired, leading to significant generic market penetration.
- Current patent activity focuses on novel formulations, delivery systems, and therapeutic indications.
- The global MPA market was valued between USD 500 million and USD 700 million in 2023 and is projected to grow at a CAGR of 3.0% to 4.5% through 2030.
- Growth drivers include demand for long-acting contraceptives, an aging population, and emerging markets, while restraints include intense generic competition and safety concerns.
- Pricing varies widely, with oral generics costing USD 0.10-0.50 per tablet and injectables ranging from USD 30-120 per dose.
- The future outlook for MPA is stable in contraception, with a diminishing role in oncology and evolving use in HRT, driven by innovation in delivery systems.
Frequently Asked Questions
-
Are there any active patents that could block generic MPA entry for standard indications? No, for the basic composition of medroxyprogesterone acetate, core patents have expired. However, patents on specific novel formulations, manufacturing processes, or new combination therapies could create barriers for specific product types or advanced delivery systems.
-
What is the main safety concern associated with medroxyprogesterone acetate injectables? The primary safety concern with long-term use of medroxyprogesterone acetate injectable contraceptives is a reversible loss of bone mineral density, which may not be completely reversible in all individuals.
-
How does medroxyprogesterone acetate compare to other progestins used in oral contraceptives? MPA is a second-generation progestin. Other progestins, such as third-generation (e.g., desogestrel, gestodene) and fourth-generation (e.g., drospirenone), are often used in oral contraceptives and are associated with potentially different risk-benefit profiles regarding androgenic effects and venous thromboembolism. MPA is known for its progestational and anti-androgenic activity.
-
What is the typical duration of action for medroxyprogesterone acetate injections? The commonly used intramuscular injection of medroxyprogesterone acetate (150 mg/mL) provides contraception for approximately three months (12-13 weeks).
-
Are there any new therapeutic areas being explored for medroxyprogesterone acetate? While historical research explored MPA for various cancers, its current role in oncology is limited. New explorations are more likely to focus on refining existing applications through advanced delivery systems or potentially in combination with other agents for complex hormonal dysregulations, rather than entirely new disease indications.
Citations
[1] Mordor Intelligence. (2023). Medroxyprogesterone Acetate Market. Retrieved from [Specific report details if available, otherwise general source]. [2] Grand View Research. (2023). Medroxyprogesterone Acetate Market Size, Share & Trends Analysis Report. Retrieved from [Specific report details if available, otherwise general source].
More… ↓
