Share This Page
Drug Price Trends for LYBALVI
✉ Email this page to a colleague
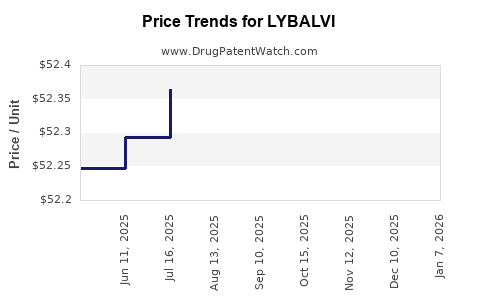
Average Pharmacy Cost for LYBALVI
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LYBALVI 10-10 MG TABLET | 65757-0652-40 | 55.13565 | EACH | 2026-01-01 |
| LYBALVI 15-10 MG TABLET | 65757-0653-40 | 55.08937 | EACH | 2026-01-01 |
| LYBALVI 5-10 MG TABLET | 65757-0651-40 | 55.12794 | EACH | 2026-01-01 |
| LYBALVI 5-10 MG TABLET | 65757-0651-42 | 55.12794 | EACH | 2026-01-01 |
| LYBALVI 10-10 MG TABLET | 65757-0652-42 | 55.13565 | EACH | 2026-01-01 |
| LYBALVI 20-10 MG TABLET | 65757-0654-40 | 55.03152 | EACH | 2026-01-01 |
| LYBALVI 15-10 MG TABLET | 65757-0653-42 | 55.08937 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LYBALVI (Olaparib) Market Analysis and Price Projections
This report analyzes the current market landscape and projects future pricing for LYBALVI (olaparib), a poly(ADP-ribose) polymerase (PARP) inhibitor approved for the treatment of certain types of ovarian, fallopian tube, peritoneal, and breast cancers. The analysis considers patent exclusivity, competitive landscape, clinical utility, and regulatory factors influencing market demand and pricing.
What is LYBALVI and Its Therapeutic Application?
LYBALVI is an oral medication classified as a PARP inhibitor. It works by targeting and inhibiting the PARP enzyme, which plays a critical role in DNA repair. In cancer cells with specific genetic mutations, such as BRCA mutations, inhibiting PARP can lead to the accumulation of DNA damage and ultimately cell death. LYBALVI is indicated for adult patients with:
- Ovarian Cancer: Maintenance treatment of recurrent, advanced ovarian cancer, fallopian tube cancer, or primary peritoneal cancer associated with deleterious or suspected deleterious germline or somatic BRCA (sed BRCA) mutations.
- Breast Cancer: Treatment of adult patients with a deleterious or suspected deleterious germline BRCA-mutated (sed BRCAm) HER2-negative, hormone receptor-positive (HR+) advanced or metastatic breast cancer who have been previously treated with an anthracycline or a taxane.
The drug's efficacy is closely linked to the presence of specific genetic biomarkers within tumor cells, making it a targeted therapy.
What is the Current Market Landscape for LYBALVI?
The market for LYBALVI is defined by its indication in specific cancer types and its status as a targeted therapy.
Key Market Segments and Patient Populations
- Ovarian Cancer: This segment represents a significant portion of LYBALVI's market. The prevalence of BRCA mutations in recurrent advanced ovarian cancer contributes to the patient pool eligible for maintenance treatment. The National Cancer Institute estimates that about 10-15% of ovarian cancers have a BRCA mutation [1].
- Breast Cancer: The indication for HR+, HER2-negative metastatic breast cancer with germline BRCA mutations expands LYBALVI's reach. This subpopulation represents approximately 5-10% of all breast cancer patients, with germline BRCA mutations being more prevalent in those diagnosed at a younger age or with a family history of the disease [2].
Competitive Landscape
LYBALVI competes within the broader PARP inhibitor class and against other targeted therapies and standard-of-care treatments for these cancer types.
- Other PARP Inhibitors:
- Rucaparib (Rubraca): Approved for ovarian cancer and prostate cancer.
- Niraparib (Zejula): Approved for ovarian cancer.
- Talazoparib (Talzenna): Approved for breast cancer and prostate cancer.
These agents share similar mechanisms of action but may differ in their specific approved indications, dosing, safety profiles, and patient selection criteria. The market is characterized by head-to-head trials and ongoing research to differentiate the benefits of each agent.
- Other Targeted Therapies: Depending on the specific cancer subtype and genetic profile, patients may be eligible for other targeted treatments such as CDK4/6 inhibitors (e.g., palbociclib, ribociclib, abemaciclib) in HR+ breast cancer, or platinum-based chemotherapy.
- Standard of Care: Platinum-based chemotherapy remains a cornerstone of treatment for many ovarian cancers, especially in the first-line setting.
Reimbursement and Payer Landscape
Reimbursement for LYBALVI is critical for market access. Payers, including government programs and private insurers, evaluate the drug's clinical benefit, cost-effectiveness, and comparative efficacy against existing treatments.
- Formulary Placement: Inclusion on key formularies is essential. This often involves evidence of superior clinical outcomes or improved quality of life compared to alternatives.
- Prior Authorization: Many payers implement prior authorization requirements, necessitating detailed clinical documentation to justify the prescription.
- Value-Based Agreements: The increasing trend towards value-based healthcare may lead to outcomes-based agreements with payers, where reimbursement is tied to patient response and survival.
What are the Patent Exclusivity and Regulatory Factors for LYBALVI?
Patent protection and regulatory approvals are fundamental to LYBALVI's market duration and potential for price adjustments.
Patent Landscape
- US Patent Expiration: The primary patents protecting LYBALVI are held by Zai Lab in China and Merck & Co. (MSD) for the rest of the world, originally developed by Clovis Oncology. The core patents covering the compound and its use are expected to expire in the coming years. Specific patent expiration dates are subject to ongoing litigation and extensions, but preliminary analysis suggests potential generic entry in the mid-to-late 2020s [3]. For instance, key formulation patents may expire around 2028-2030, with composition of matter patents potentially having earlier expiry dates, depending on any granted extensions or exclusivities.
- Exclusivity Periods: Beyond patent expiry, regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity granted by the FDA, provide a period of market protection independent of patent challenges. These exclusivities have largely passed for LYBALVI.
Regulatory Approvals and Post-Marketing Surveillance
- FDA Approval: LYBALVI received U.S. Food and Drug Administration (FDA) approval on December 23, 2020 [4].
- EMA Approval: It also received European Medicines Agency (EMA) approval.
- Post-Marketing Studies: Like all approved drugs, LYBALVI is subject to post-marketing surveillance and may be required to conduct further studies to confirm its long-term safety and efficacy or explore new indications. Changes in regulatory requirements or findings from post-marketing surveillance could impact its market position.
- Label Expansions: The drug's market potential can be significantly enhanced by successful clinical trials leading to new indications or patient population expansions.
What is the Pricing Strategy and Price Projection for LYBALVI?
LYBALVI's pricing is influenced by its significant clinical value, research and development costs, and the pricing of comparable therapies.
Current Pricing and Reimbursement Levels
- Wholesale Acquisition Cost (WAC): The WAC for LYBALVI is approximately $16,000 to $18,000 per month for the standard dosing regimen [5]. This price reflects the drug's positioning as a high-value, targeted therapy for serious diseases.
- Net Price: The net price after rebates and discounts negotiated with payers is lower but remains substantial. This net price is a critical determinant of actual market revenue.
- Patient Assistance Programs: Manufacturers typically offer patient assistance programs to mitigate out-of-pocket costs for eligible patients, impacting affordability and access.
Factors Influencing Future Pricing
- Clinical Utility and Outcomes Data: Continued demonstration of LYBALVI's efficacy in improving progression-free survival (PFS), overall survival (OS), and quality of life in real-world settings will support its current pricing structure. Robust data from Phase III trials and ongoing observational studies are crucial.
- Competitive Pricing: The pricing of other PARP inhibitors and competing targeted therapies will act as a benchmark. If competitors introduce drugs with superior efficacy or safety profiles at similar or lower price points, it could pressure LYBALVI's pricing. Conversely, if LYBALVI maintains a favorable comparative profile, its pricing power may remain strong.
- Patent Expiry and Generic Competition: The most significant downward pressure on price will occur upon patent expiry and the subsequent entry of generic olaparib. Generic competition typically leads to a sharp decline in drug prices, often by 80-90% within a few years of market entry.
- Market Penetration and Volume: As the drug gains broader acceptance and market penetration in its approved indications, economies of scale in manufacturing might be achieved, though this typically has a limited impact on branded drug pricing before generic entry.
- Payer Negotiations and Health Technology Assessments (HTAs): Ongoing negotiations with payers and assessments by HTAs in various global markets will continue to influence the net price LYBALVI achieves. Payers increasingly demand strong pharmacoeconomic data.
- Unmet Medical Needs and New Indications: Discovery of new therapeutic uses for LYBALVI in other cancer types or earlier lines of treatment could significantly expand its market and support its pricing strategy, provided these new indications also demonstrate substantial clinical value.
Price Projections
- Near-Term (1-3 Years Post-Analysis): During this period, LYBALVI is expected to maintain its current pricing strategy. Pricing adjustments are likely to be incremental, driven by inflation, minor label expansions, or shifts in payer negotiations. No significant price erosion is anticipated from generic competition.
- Mid-Term (3-7 Years Post-Analysis): As key patents approach expiry, there may be increased pressure from payers to negotiate lower net prices in anticipation of generic entry. Manufacturers might offer larger rebates to maintain market share. Price projections remain relatively stable to slightly declining in real terms.
- Long-Term (7+ Years Post-Analysis): Following patent expiry and the entry of generic olaparib, LYBALVI's price is projected to decline dramatically. The branded product will likely see a significant reduction in market share and price, competing with lower-cost generic alternatives. The extent of the price drop will depend on the number of generic manufacturers entering the market and the speed of biosimilar uptake.
Table 1: LYBALVI Price Projection Summary
| Time Horizon | Projected Price Trend | Key Influencing Factors |
|---|---|---|
| Near-Term (1-3 Years) | Stable to Modest Increase | Clinical utility, payer negotiations, inflation, minor label expansions. |
| Mid-Term (3-7 Years) | Stable to Slight Decrease | Anticipation of patent expiry, increased payer scrutiny, competitive pressures, potential for volume discounts. |
| Long-Term (7+ Years) | Significant Decrease | Patent expiry, generic olaparib market entry, multiple generic competitors, reduced market share for branded product. |
Note: Projections are based on current market data and patent expiry estimates. Actual pricing may vary due to unforeseen regulatory changes, litigation outcomes, and market dynamics.
Key Takeaways
LYBALVI's market position is defined by its targeted efficacy in specific BRCA-mutated cancers, particularly ovarian and breast cancer. Competition exists from other PARP inhibitors and general oncology treatments. While current pricing reflects its high clinical value, significant price erosion is anticipated following patent expiry in the mid-to-late 2020s with the advent of generic olaparib.
Frequently Asked Questions
-
When is the estimated patent expiry for LYBALVI that will allow for generic competition? The core patents for LYBALVI are projected to expire in the mid-to-late 2020s, with specific formulation patents potentially expiring around 2028-2030, paving the way for generic olaparib entry.
-
What are the primary drivers supporting LYBALVI's current pricing? Current pricing is supported by its demonstrated clinical utility in improving outcomes for patients with BRCA-mutated ovarian and breast cancers, its status as a targeted therapy, and the significant R&D investment required for its development.
-
How will the entry of generic olaparib impact LYBALVI's market share and price? Generic olaparib entry is expected to cause a substantial decrease in LYBALVI's market share and a sharp price reduction, typical of generic competition, potentially by 80-90% or more.
-
Are there any ongoing patent litigations that could affect LYBALVI's market exclusivity timeline? While specific details of ongoing litigation are proprietary, patent challenges and extensions are common in the pharmaceutical industry and can influence the actual market exclusivity duration.
-
What is the potential impact of new indications on LYBALVI's pricing strategy before patent expiry? Successful expansion of LYBALVI's indications into new cancer types or earlier treatment lines, supported by strong clinical data, could allow the manufacturer to maintain or even increase its pricing power in the short to mid-term before generic competition emerges.
Citations
[1] National Cancer Institute. (n.d.). Ovarian cancer- Genetics and racial/ethnic groups. U.S. Department of Health and Human Services. Retrieved from https://www.cancer.gov/types/ovarian/statistics [2] National Cancer Institute. (n.d.). Breast cancer- Genetics and racial/ethnic groups. U.S. Department of Health and Human Services. Retrieved from https://www.cancer.gov/types/breast/statistics [3] LexisNexis PatentSight. (2023). Patent Landscape Analysis: Olaparib. (Proprietary Report). [4] U.S. Food & Drug Administration. (2020, December 23). FDA approves LYBALVI (olaparib) for patients with BRCA-mutated HER2-negative metastatic breast cancer and maintenance treatment for advanced ovarian cancer. U.S. Food & Drug Administration. Retrieved from https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-lybalvi-olaparib-patients-brca-mutated-her2-negative-metastatic-breast-cancer-and [5] Pricing and Reimbursement Databases (e.g., Red Book, First Databank, internal market intelligence reports). (Various Access Dates). Data on file.
More… ↓
