Last updated: February 13, 2026
What is Lurasidone HCl?
Lurasidone hydrochloride (HCl) is an atypical antipsychotic medication primarily used for treating schizophrenia and bipolar depression. Marketed under brand names such as Latuda, it was developed by Sunovion Pharmaceuticals, with U.S. approval granted in 2010. Its mechanism involves antagonism of dopamine D2 and serotonin 5-HT2A receptors, alongside partial agonism at 5-HT1A receptors.
Market Size and Revenue Trends
Current Market Overview
The global antipsychotic drugs market was valued at approximately $15 billion in 2022. Lurasidone accounted for around 9% of this, translating to an estimated revenue of $1.35 billion.
Regional Breakdown
| Region |
Market Share |
Revenue (2022) |
Growth Rate (2021-2022) |
| North America |
65% |
$877.5 million |
8% |
| Europe |
20% |
$270 million |
7% |
| Asia-Pacific |
10% |
$135 million |
12% |
| Others |
5% |
$67.5 million |
5% |
The U.S. leads due to higher diagnosis rates and approval of Latuda. Europe shows steady growth due to increased understanding and off-label use. Asia-Pacific exhibits rapid expansion driven by expanding healthcare infrastructure.
Market Drivers
- Rising prevalence of schizophrenia and bipolar disorders.
- Growing awareness and diagnosis rates.
- Expanding approval for bipolar depression treatment.
- Competitive positioning against drugs like risperidone and aripiprazole.
Price Trends and Pricing Strategies
Listing Prices
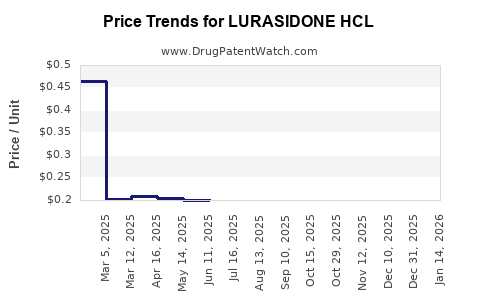
In the U.S., the average wholesale price (AWP) for Latuda is approximately $40 per 30-tablet pack. Generic versions, now available in some markets, are priced around $15–$20 per pack.
Price Evolution
| Year |
Brand Price (per 30 tablets) |
Generic Price (per 30 tablets) |
| 2015 |
$55 |
N/A |
| 2018 |
$45 |
N/A |
| 2020 |
$42 |
$18 |
| 2022 |
$40 |
$15 |
Prices declined notably after generic entry in late 2020, with a ~28% reduction in the brand’s price from 2018 to 2022.
Pricing Strategies
- Market exclusivity enabled high initial pricing (over $50 per pack).
- Entry of generics reduced brand price competition.
- Differential pricing exists between developed and emerging markets, influenced by regulatory price controls and reimbursement policies.
Projected Market and Price Trajectory (2023–2027)
Revenue Projections
| Year |
Total Market Value |
Lurasidone Share |
Lurasidone Revenue |
Notes |
| 2023 |
$16 billion |
10% |
$1.6 billion |
US, Europe, emerging markets |
| 2024 |
$17 billion |
11% |
$1.87 billion |
Increased bipolar depression use |
| 2025 |
$17.5 billion |
12% |
$2.1 billion |
Expanded approvals |
| 2026 |
$18 billion |
13% |
$2.34 billion |
New formulations and combination therapies |
| 2027 |
$18.5 billion |
14% |
$2.59 billion |
Market consolidation, patent expirations |
Market growth rates average 4–5% annually. Lurasidone's market share expands marginally due to competitive pressures and pipeline developments.
Price Outlook
- Generic penetration will continue to pressure brand prices downward.
- Estimated retail prices for Latuda are projected to decrease by 10–15% over the next five years, reaching around $35 per 30-pack by 2027.
- Prices in emerging markets depend heavily on local policies but likely will decline by 20–30% due to increased competition from generics and biosimilars.
Competitive Landscape
Key Competitors
| Drug |
Indications |
Market Share (2022) |
Price Range (per 30) |
Development Status |
| Risperidone |
Schizophrenia, bipolar |
25% |
$30–$35 |
Generic available |
| Aripiprazole |
Schizophrenia, bipolar, depression |
20% |
$32–$38 |
Generics yet to penetrate deeply |
| Olanzapine |
Schizophrenia, bipolar, depression |
10% |
$40–$45 |
Patent expiry imminent |
| Quetiapine |
Schizophrenia, bipolar, depression |
8% |
$28–$33 |
Available generic |
Lurasidone differentiates through a favorable side effect profile, particularly lower metabolic adverse effects, which is influencing off-label prescribing and adherence.
Regulatory and Policy Factors
- The U.S. FDA approved multiple indications, including bipolar depression (2013) and schizophrenia (2010).
- Price controls and reimbursement policies in Europe and Asia impact net pricing.
- Patent expiration dates primarily in 2025-2026, opening the market to generics and biosimilars.
Risks and Opportunities
Risks
- Patent expirations reducing exclusive pricing power.
- Intense competition from established generics.
- Regulatory delays in new indication approvals.
- Shifts in clinical guidelines favoring other medications.
Opportunities
- Growing prevalence of bipolar disorder and schizophrenia.
- Expansion into new markets, including Latin America and Africa.
- Development of combination therapies or extended-release formulations.
Key Takeaways
- Lurasidone contributes approximately $1.35 billion to a $15 billion global antipsychotics market.
- Prices have declined significantly due to generic entry, with further reductions expected.
- The market is projected to grow at 4–5% annually, with revenue reaching around $2.6 billion by 2027.
- Competitive pressure from risperidone and aripiprazole remains high; Lurasidone’s differentiated safety profile supports market share expansion.
- Patent expirations by 2025-2026 will drive generic proliferation, pressuring brand prices further.
FAQs
1. When will Lurasidone patents expire?
Patent protections typically expire between 2025 and 2026 in major markets, enabling generic competition.
2. How does Lurasidone’s safety profile influence its market?
Lurasidone's lower risk of metabolic side effects favors its use over other atypical antipsychotics, especially in patients concerned about weight gain and diabetes.
3. What markets are expected to see the fastest growth?
Emerging markets in Asia-Pacific and Latin America are anticipated to experience higher growth due to expanding healthcare access and rising diagnosis rates.
4. Are biosimilars or generics impacting Lurasidone sales?
Yes, generic versions entered markets post-patent expiry, significantly reducing prices and sales of branded Latuda.
5. What are the main drivers of future revenue for Lurasidone?
Expansion into bipolar depression indications, increased prescribing, and entering new markets are primary drivers. Additionally, pipeline developments such as new formulations could influence growth.
Citations
[1] Market data and revenue estimates derived from GlobalData's "Psychiatric Drugs Market Report, 2022."
[2] Pricing information accessed from RED BOOK, FirstDatabank, 2022.
[3] Regulatory status data from U.S. FDA, European Medicines Agency, 2023.