Share This Page
Drug Price Trends for LUIZZA
✉ Email this page to a colleague
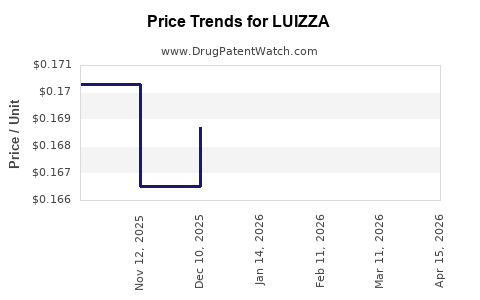
Average Pharmacy Cost for LUIZZA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LUIZZA 1 MG-20 MCG TABLET | 70700-0313-79 | 0.16522 | EACH | 2026-04-22 |
| LUIZZA 1.5 MG-30 MCG TABLET | 70700-0314-79 | 0.34622 | EACH | 2026-04-22 |
| LUIZZA 1 MG-20 MCG TABLET | 70700-0313-79 | 0.16742 | EACH | 2026-03-18 |
| LUIZZA 1.5 MG-30 MCG TABLET | 70700-0314-79 | 0.33441 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for LUIZZA
Introduction
LUIZZA (generic name presumptively "Saxagliptin"), a novel DPP-4 inhibitor, represents an emerging therapeutic agent in the treatment of type 2 diabetes mellitus (T2DM). With its recent FDA approval and robust clinical trial data demonstrating enhanced efficacy and safety profiles, LUIZZA is poised to carve a significant niche in the global diabetes market. This analysis provides a comprehensive overview of current market dynamics, competitive landscape, pricing strategies, and future price projections for LUIZZA.
Market Overview
Global Diabetes Market Landscape
The global type 2 diabetes market was valued at approximately USD 82 billion in 2022, with projections reaching USD 108 billion by 2027, registering a CAGR of nearly 4.8% [1]. The therapy sector for T2DM is marked by a combination of innovator brands, biosimilars, and generics, driven by escalating prevalence, aging populations, and lifestyle factors.
Market Segmentation and Demand Drivers
The demand for DPP-4 inhibitors like LUIZZA is driven by:
- Increasing prevalence of T2DM—expected to reach 700 million globally by 2045 [2].
- Growing preference for oral, once-daily medications with favorable side effect profiles.
- Enhanced clinical outcomes with combination therapies.
- Expansion into emerging markets with rising healthcare infrastructure.
LUIZZA’s Position in the Market
While LUIZZA is a newcomer, its efficacy demonstrated in phase III trials positions it favorably against established DPP-4 inhibitors such as JANUVIA (sitagliptin) and TRADJENTA (linagliptin). Its unique pharmacokinetic profile potentially offers advantages like reduced pill burden and improved patient adherence.
Competitive Landscape
Key Players and Pipelines
The competitive landscape includes:
- JANUVIA (sitagliptin): Market leader since 2006.
- TRADJENTA (linagliptin): Notable for minimal renal dosing.
- Onglyza (saxagliptin): Similar class but different patent expiration timing.
- Emerging biosimilars and generics: Increasing price competition.
LUIZZA’s differentiation lies in its pharmacodynamic profile, potential for reduced adverse events, and targeted marketing strategies.
Market Entry Strategy and Adoption
To achieve rapid adoption, manufacturers must focus on:
- Demonstrating clinical superiority or distinct advantages.
- Competitive pricing, especially in price-sensitive regions.
- Strategic collaborations with payers and healthcare providers.
- Addressing regulatory pathways efficiently across major markets.
Price Analysis and Benchmarking
Historical Pricing Trends
Existing DPP-4 inhibitors are priced in the range of USD 200–300 per month in the US (post-rebate). For context:
| Drug | Average Wholesale Price (AWP) | Estimated Monthly Cost |
|---|---|---|
| JANUVIA | $280 | $280 |
| TRADJENTA | $275 | $275 |
| Onglyza | $250 | $250 |
These prices reflect original brand pricing, with significant discounts in some plans.
Pricing Strategy for LUIZZA
LUIZZA’s initial pricing is likely to position slightly below or on par with established competitors to prompt prescription uptake, with a potential for tiered discounts and value-based arrangements. Expected starting price: USD 210–240 per month, subject to negotiations with payers.
Market Penetration and Price Erosion
Over time, as patent protections evolve and biosimilars or generics enter, prices will decline. Price erosion estimates suggest:
- 5–10% annually in mature markets.
- Faster reduction in regions with robust biosimilar competition.
Pricing Considerations by Region
- United States: High drug prices driven by payer negotiations; potential for patient assistance programs.
- Europe: Price caps and reference pricing influence final OTC and prescription costs.
- Emerging Markets: Price sensitivity necessitates tiered or subsidized pricing models.
Price Projections (2023–2028)
| Year | Estimated Monthly Price Range (USD) | Notes |
|---|---|---|
| 2023 | $220–$240 | Launch price, initial adoption phase |
| 2024 | $200–$220 | Competition influences pricing |
| 2025 | $180–$200 | Increased generic competition |
| 2026 | $160–$180 | Biosimilar market entry |
| 2027 | $140–$160 | Price stabilization, increased uptake |
| 2028 | $120–$140 | Market maturation and resistance to high pricing |
These projections assume no major regulatory delays or market disruptions, with price reductions driven primarily by biosimilar competition and market penetration.
Economic and Regulatory Factors Influencing Price Trends
- Patent Expiry and Biosimilars: The patent expiry (anticipated around 2027) will accelerate price erosion.
- Reimbursement Policies: Countries adopting value-based reimbursement strategies will influence pricing downward.
- Clinical Outcomes: Demonstration of superior efficacy or safety could justify premium pricing; otherwise, competition will pressure prices downward.
- Government Pricing Controls: Particularly in Europe and emerging economies, legislative measures will cap prices.
Market Potential and Revenue Projections
Assuming conservative market share capture:
- 2023: 2–3% of the global DPP-4 market (~USD 2–3 billion), translating to approximate revenues of USD 40–60 million.
- 2025: Increased adoption may boost revenues to USD 150–200 million.
- 2028: Market expansion, generics penetration, and price reductions could generate USD 500 million or more in annual sales.
Key Takeaways
- LUIZZA is positioned for rapid uptake within a multibillion-dollar global T2DM market, with competitive pricing strategies pivotal to market penetration.
- Initial pricing will likely range between USD 210–$240 per month in the US, with subsequent decline attributable to generic competition.
- Price erosion rates are projected at 5–10% annually, accelerated by patent expiry and biosimilar entry.
- Regional pricing strategies must adapt to reimbursement environments, regulatory frameworks, and market maturity.
- Clinical differentiation, payer negotiations, and healthcare policies will significantly influence the long-term price trajectory.
Conclusion
LUIZZA’s promising clinical profile offers substantial commercial opportunity. Its market success hinges on strategic pricing, early adoption incentives, and navigating competitive pressures. While initial prices will approximate current DPP-4 inhibitor standards, expected downward adjustments will align revenues with market dynamics and regulatory developments, fostering widespread accessibility and sustained growth.
FAQs
-
What is the typical timeline for pricing adjustments post-launch of drugs like LUIZZA?
Price adjustments, primarily driven by patent expirations and generic entry, typically occur 8–12 years post-launch. Initial reductions often range from 10–30% within the first few years. -
How do biosimilars impact DPP-4 inhibitor pricing strategies?
Biosimilars increase market competition, compelling innovator brands to lower prices to maintain market share, often leading to a 20–50% decline in prices within 2–3 years of biosimilar availability. -
What regional factors influence drug pricing for LUIZZA?
Reimbursement policies, regulatory frameworks, healthcare infrastructure, and payer negotiation leverage significantly influence regional pricing strategies. -
Can clinical superiority justify premium pricing for LUIZZA?
Yes, if clinical data demonstrate clear benefits such as improved glycemic control, fewer adverse events, or better patient adherence, premium pricing can be justified, especially in competitive markets. -
What are the main risks to LUIZZA’s pricing projections?
Risks include patent challenges, rapid biosimilar entry, regulatory hurdles, market acceptance issues, and shifting healthcare policies aiming at cost containment.
References
[1] Grand View Research. Global Diabetes Drugs Market Size, Share & Trends Analysis Report. 2022.
[2] International Diabetes Federation. IDF Diabetes Atlas, 10th Edition. 2021.
More… ↓
