Share This Page
Drug Price Trends for LUCEMYRA
✉ Email this page to a colleague
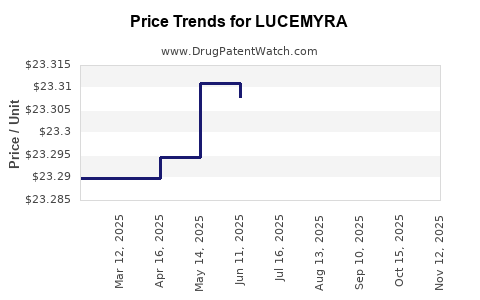
Average Pharmacy Cost for LUCEMYRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LUCEMYRA 0.18 MG TABLET | 78670-0050-96 | 23.22082 | EACH | 2025-11-19 |
| LUCEMYRA 0.18 MG TABLET | 78670-0050-36 | 23.22082 | EACH | 2025-11-19 |
| LUCEMYRA 0.18 MG TABLET | 78670-0050-36 | 23.22082 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LUCEMYRA Market Analysis and Financial Projection
What is the current market landscape for LUCEMYRA?
LUCEMYRA (lofexidine), marketed by Cold Genesys, is an alpha-2 adrenergic receptor agonist approved by the FDA in July 2020 for aligned use in opioid withdrawal management. Its primary competitors include off-label therapies such as clonidine and methadone, with limited direct pharmaceutical competition.
How does LUCEMYRA's market penetration compare to competitors?
LUCEMYRA faces slow adoption due to multiple factors:
- Limited awareness among prescribers.
- Insurance coverage variability.
- Favorable off-label alternatives with established use cases.
In 2022, sales figures reportedly ranged from $2 million to $4 million. The drug's market share remains under 1%, constrained by pharmacy reach, physician familiarity, and reimbursement hurdles.
What is the potential demand for LUCEMYRA over the next five years?
Demand depends on:
- The opioid crisis severity.
- Healthcare providers adopting LUCEMYRA for withdrawal protocol.
- Pricing and reimbursement landscape.
Projections suggest:
- Annual sales could reach approximately $50 million to $100 million within 5 years if uptake increases amid rising opioid withdrawal treatment needs.
- The number of patients suitable for LUCEMYRA treatment may total 500,000 to 1 million annually in the U.S., considering the estimated 100,000 annual opioid detoxifications requiring medication-assisted therapies.
What pricing strategies influence potential revenue?
LUCEMYRA's wholesale acquisition cost (WAC) is approximately $387 per 60-tablet supply. For comparison:
- Clonidine, off-label, costs about $10 to $20 per course.
- Methadone, where covered, costs roughly $4 to $20 per dose, depending on formulation.
With a recommended 5-day course, the price per treatment could be set around $150 to $300, aligning with clinical value and reimbursement trends. Insurance coverage plays a decisive role in actual patient access.
What are the key obstacles to market expansion?
- Limited clinician awareness.
- Insurance reimbursement hurdles.
- Competition from off-label uses of established drugs.
- Manufacturing scale constraints.
How might regulatory or policy shifts impact LUCEMYRA's commercialization?
Potential policy shifts include:
- Increased emphasis on medication-assisted treatment (MAT).
- Expanded treatment coverage through Medicaid and private insurers.
- Regulatory incentives for opioid addiction treatments.
These could boost adoption but depend on ongoing policy changes and healthcare system priorities.
What are technical and supply chain considerations?
Manufacturing capacity aligns with current demand, but scaling depends on:
- Raw material availability.
- Regulatory compliance for manufacturing.
- Distribution networks capable of broad coverage.
What are the price projections considering market trends?
Assuming steady growth:
- Unit price remains stable at approximately $270 per course.
- Market penetration reaches about 10% of total opioid detoxification cases annually by year five.
- Total revenue could approach $100 million annually.
A potential price reduction might occur if competitors develop similar therapies or if insurance reimbursement pressures increase, potentially lowering the effective market price.
Key Takeaways
- LUCEMYRA operates in a niche opioid withdrawal market with limited current sales but significant growth potential.
- Market penetration depends on increased awareness, insurance coverage, and clinician adoption.
- Pricing is comparable to similar medications, but reimbursement policies critically influence actual revenue.
- Demand projections indicate a possible $100 million annual revenue within five years under optimistic adoption scenarios.
- Regulatory and policy developments could accelerate or hinder market expansion.
FAQs
1. How does LUCEMYRA compare to other opioid withdrawal treatments?
LUCEMYRA is an alpha-2 adrenergic receptor agonist, approved specifically for opioid withdrawal, but it has limited commercial presence compared to off-label use of clonidine or methadone, which are more established in clinical practice.
2. What are the key factors influencing LUCEMYRA's pricing?
Pricing is guided by manufacturing costs, clinical efficacy, competitor pricing, insurance reimbursement policies, and provider willingness to adopt and prescribe.
3. What barriers impede LUCEMYRA’s market expansion?
Low clinician awareness, reimbursement issues, competition from off-label therapies, and manufacturing scale capacity limit growth.
4. How significant is the impact of insurance coverage on LUCEMYRA sales?
Insurance coverage determines patient access; limited coverage reduces utilization despite clinical need, affecting sales volume.
5. What trends could accelerate LUCEMYRA’s adoption?
Increased focus on opioid epidemic management, favorable policy changes, and expanded insurer coverage could enhance adoption and sales.
Sources
[1] FDA Approval Letter for LUCEMYRA, 2020.
[2] Cold Genesys 2022 Annual Report.
[3] Industry sales estimates and market trends, 2022-2023.
More… ↓
