Share This Page
Drug Price Trends for LUBRIFRESH PM EYE OINTMENT
✉ Email this page to a colleague
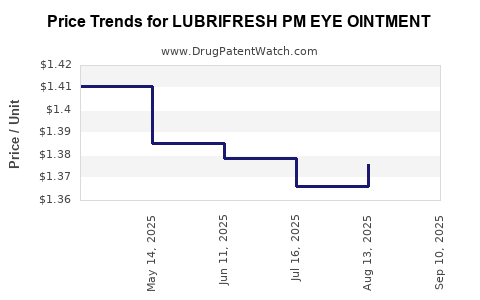
Average Pharmacy Cost for LUBRIFRESH PM EYE OINTMENT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LUBRIFRESH PM EYE OINTMENT | 00904-6488-38 | 1.37357 | GM | 2025-09-17 |
| LUBRIFRESH PM EYE OINTMENT | 00904-6488-38 | 1.37618 | GM | 2025-08-20 |
| LUBRIFRESH PM EYE OINTMENT | 00904-6488-38 | 1.36602 | GM | 2025-07-23 |
| LUBRIFRESH PM EYE OINTMENT | 00904-6488-38 | 1.37856 | GM | 2025-06-18 |
| LUBRIFRESH PM EYE OINTMENT | 00904-6488-38 | 1.38546 | GM | 2025-05-21 |
| LUBRIFRESH PM EYE OINTMENT | 00904-6488-38 | 1.41060 | GM | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for LUBRIFRESH PM Eye Ointment
Introduction
LUBRIFRESH PM Eye Ointment is a specialized ophthalmic lubricant designed to alleviate dry eye symptoms and enhance ocular comfort. Its formulation typically includes polyvinyl alcohol and propylene glycol, ingredients known for their moisturizing properties. As a product targeting a growing global prevalence of dry eye disease, driven by factors such as aging populations and increased screen time, LUBRIFRESH PM represents a notable segment within the ophthalmic pharmaceutical market. This analysis provides a comprehensive market overview, examines competitive positioning, regulatory considerations, and offers price projection insights grounded in current industry trends.
Market Overview
Global Ophthalmic Lubricants Market Dynamics
The global ophthalmic lubricants market has experienced accelerated growth, forecasted to reach approximately USD 1.8 billion by 2025, with a compound annual growth rate (CAGR) of around 5-6% (Statista, 2022)[1]. The increasing incidence of dry eye disease, projected to affect over 500 million individuals globally by 2030 (The Dry Eye Workshop, 2017)[2], propels demand for over-the-counter (OTC) and prescription lubricants.
Key Drivers
- Aging Population: Age-related decline in tear production augments dry eye prevalence, notably in North America, Europe, and Asia-Pacific.
- Digital Device Usage: Extended screen time, especially among millennials and Generation Z, heightens dry eye complaints.
- Product Innovation: Development of preservative-free, sustained-release, and combination formulations enhances treatment efficacy and patient adherence.
- Regulatory Approvals: Increased approvals of ophthalmic ointments as OTC products stimulate market accessibility.
Regional Market Segmentation
- North America: Dominates due to high awareness, extensive healthcare infrastructure, and self-medication trends.
- Europe: Rapid adoption driven by aging demographics and product availability.
- Asia-Pacific: Exhibits the fastest growth rate, fueled by expanding healthcare sectors and rising disposable incomes.
Competitive Landscape
Major global players include Johnson & Johnson (Refresh Repair), Allergan (Refresh Liquigel), and Bausch + Lomb (Refresh Tears). Numerous regional and local manufacturers operate predominantly in OTC segments, introducing generic or store-brand formulations to compete.
LUBRIFRESH PM, being a niche prescription or OTC product, faces competition primarily from branded counterparts with similar active ingredients and innovative delivery systems. The entry of generic versions, especially post-expiry of patents, pressures pricing strategies.
Regulatory Considerations
LUBRIFRESH PM’s regulatory pathway hinges on regional authorities:
- United States (FDA): Typically classed as an ophthalmic drug, requiring rigorous safety and efficacy data before OTC or prescription approval.
- European Union (EMA): Subject to centralized or national procedures, emphasizing quality, safety, and clinical performance.
- Emerging Markets: Regulatory requirements vary, often less stringent, enabling faster market entry but raising concerns about quality standards.
Regulatory developments are increasingly favoring preservative-free formulations, influencing market dynamics and price points.
Pricing Strategies and Trends
Current Pricing Landscape
In North America, OTC ophthalmic ointments like Refresh Liquigel are priced between USD 10-15 per tube (10g). Prescription formulations tend to be priced higher, around USD 25-35 per tube, reflecting clinical efficacy and brand value. In Europe and Asia-Pacific, pricing varies broadly based on healthcare policies, reimbursement frameworks, and local manufacturing costs.
Factors Influencing Pricing
- Ingredient Costs: Polyvinyl alcohol and glycerin are affordable raw materials; however, preservative-free or sterilization processes increase costs.
- Formulation Innovation: Sustained-release or preservative-free variants command premium prices.
- Brand Positioning: Established brands leverage brand loyalty, allowing for higher pricing.
- Market Factors: Competitive intensity, reimbursement policies, and supply chain efficiencies influence price points.
Projected Price Trends
Given the evolving landscape, the following projections are anticipated over the next five years:
| Year | OTC Price Range (USD) | Prescription Price Range (USD) | Remarks |
|---|---|---|---|
| 2023 | $10 - $15 | $25 - $35 | Current baseline; steady demand |
| 2024 | $11 - $16 | $27 - $37 | Slight premium increase, inflation, innovation |
| 2025 | $12 - $17 | $30 - $40 | Premium formulations gain traction |
| 2026 | $13 - $18 | $33 - $45 | Increased adoption of preservative-free options |
| 2027 | $14 - $20 | $35 - $50 | Market saturation, brand differentiation critical |
Note: These projections incorporate inflation, raw material costs, regulatory shifts, and competitive responses.
Market Entry and Pricing Recommendations
- Differentiation: Innovate with preservative-free, sustained-release formulations to justify premium pricing.
- Cost Optimization: Leverage manufacturing efficiencies and regional sourcing.
- Regulatory Strategy: Rapid registration in emerging markets can enable competitive pricing.
- Brand Positioning: Invest in clinician and consumer education to build trust and justify higher price points.
- Reimbursement Policies: Engage with payers early to secure reimbursement pathways, aligning pricing strategies accordingly.
Conclusion
LUBRIFRESH PM Eye Ointment operates within a dynamic, expanding global ophthalmic lubricants market driven by demographic shifts, digital lifestyles, and product innovation. Its future value will hinge on regulatory navigation, formulation superiority, and strategic positioning amidst intense competition. Price projections suggest a gradual increase, especially for premium and preservative-free variants, with regional market nuances shaping profitability and access.
Key Takeaways
- The ophthalmic lubricants market is projected to grow at a CAGR of 5-6%, driven by demographic and behavioral factors.
- LUBRIFRESH PM’s positioning as a preservative-free, sustained-release product aligns with market trends favoring safety and convenience.
- Pricing strategies should incorporate regional healthcare policies, competitive positioning, and value-based differentiation.
- Premium formulations are anticipated to command higher prices, especially as consumers and clinicians prioritize safety and efficacy.
- Market entry in emerging regions offers opportunities for early adoption and pricing advantages but requires navigating diverse regulatory environments.
FAQs
1. What are the primary factors influencing the pricing of ophthalmic ointments like LUBRIFRESH PM?
Pricing is influenced by raw material costs, formulation complexity (e.g., preservative-free or sustained-release), brand strength, regulatory compliance, regional market conditions, and reimbursement policies.
2. How does regulatory approval impact the market entry and pricing of ophthalmic drugs?
Regulatory approval determines market access; stricter requirements can elevate costs but also position the product as higher quality, enabling premium pricing. Conversely, faster approvals in emerging markets can reduce costs and accelerate revenue.
3. What are the key competitive advantages for LUBRIFRESH PM in a saturated market?
Differentiation through innovative formulation (preservative-free, sustained release), clinical efficacy, safety profile, patient comfort, and clinician preference are critical for competitive positioning and justified pricing.
4. Which emerging markets present the most growth opportunities for ophthalmic lubricants?
Asia-Pacific regions, including China and India, offer significant growth potential due to expanding healthcare infrastructure and rising awareness of dry eye treatments, coupled with less saturated markets.
5. How might the trend toward OTC availability impact pricing and market share?
OTC availability broadens consumer access, increasing sales volume but often pressures prices downward. Brands competing in OTC segments must balance affordability with perceived value to capture market share.
References
[1] Statista. "Ophthalmic Lubricants Market Size & Forecast." 2022.
[2] The Dry Eye Workshop (DEWS). "Report of the Refractive Surgery Dry Eye Subcommittee." 2017.
More… ↓
