Share This Page
Drug Price Trends for LOVENOX
✉ Email this page to a colleague
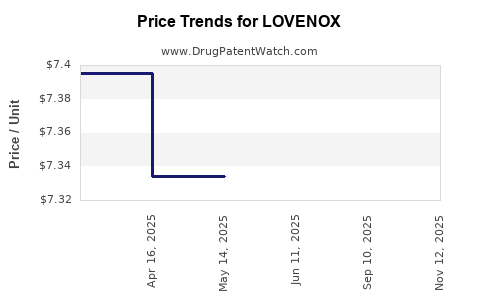
Average Pharmacy Cost for LOVENOX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOVENOX 40 MG/0.4 ML SYRINGE | 00075-0620-40 | 6.77500 | ML | 2025-11-19 |
| LOVENOX 40 MG/0.4 ML SYRINGE | 00075-8014-10 | 6.77500 | ML | 2025-11-19 |
| LOVENOX 40 MG/0.4 ML SYRINGE | 00075-8014-10 | 6.82350 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LOVENOX
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LOVENOX 150MG/ML INJ SYRINGE | Sanofi Aventis U.S. LLC | 00075-2915-01 | 10 | 29.81 | 2.98100 | EACH | 2024-01-01 - 2028-05-31 | Big4 |
| LOVENOX INJECTION | Sanofi Aventis U.S. LLC | 00075-0622-80 | 10 | 57.62 | 5.76200 | EACH | 2024-01-01 - 2028-05-31 | FSS |
| LOVENOX 120MG/0.8ML INJ SYRINGE | Sanofi Aventis U.S. LLC | 00075-2912-01 | 10 | 18.95 | 1.89500 | EACH | 2023-06-01 - 2028-05-31 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Lovenox: Market Dynamics and Future Pricing
Lovenox (enoxaparin sodium) is a low molecular weight heparin (LMWH) anticoagulant used to prevent and treat blood clots. Its market faces pressure from biosimilar competition, patent expiries, and evolving treatment guidelines, impacting future pricing and market share.
What is the current market size and segmentation for Lovenox?
The global market for enoxaparin sodium is substantial, driven by its established efficacy in prophylaxis and treatment of venous thromboembolism (VTE) and acute coronary syndromes (ACS) [1]. Market segmentation typically occurs by application and distribution channel.
Key Applications:
- Deep Vein Thrombosis (DVT) Prophylaxis: Prevention of blood clots in the legs, often post-surgery or in immobile patients.
- Pulmonary Embolism (PE) Treatment: Treatment of blood clots in the lungs.
- Acute Coronary Syndromes (ACS): Management of heart attack and unstable angina.
- Other Uses: Including prevention of clotting in patients undergoing hemodialysis.
Distribution Channels:
- Hospitals: Major consumers due to inpatient settings and surgical procedures.
- Retail Pharmacies: For outpatient prescriptions.
- Clinics: For ambulatory care and specialized treatments.
The specific market size fluctuates based on reporting period and geographical focus, but estimates place the global enoxaparin sodium market in the billions of U.S. dollars. For instance, a report from 2022 estimated the global market to be approximately $2.9 billion, with a projected compound annual growth rate (CAGR) of 4.1% from 2023 to 2030 [2]. This growth is tempered by biosimilar entry.
What is the patent landscape and generic/biosimilar competition for Lovenox?
Sanofi's Lovenox patents have largely expired, leading to the introduction of generic and biosimilar alternatives in major markets.
Key Patent Expirations:
- United States: Key composition of matter and method of use patents for enoxaparin sodium expired in the early 2010s.
- Europe: Similar patent expiries occurred around the same period.
Biosimilar Landscape:
The term "biosimilar" is specifically used for biologic drugs. Enoxaparin sodium is a chemically synthesized drug, meaning its generic versions are directly comparable. However, the complexity of heparin production has led to challenges in replicating it perfectly, and regulatory bodies have established rigorous standards for bioequivalence.
- United States: The Food and Drug Administration (FDA) has approved multiple generic versions of enoxaparin sodium. Brands like Sandoz's enoxaparin sodium injection (marketed as LEQIVO) and Pfizer's enoxaparin sodium injection have entered the market [3].
- Europe: Numerous generic manufacturers have launched enoxaparin sodium products, increasing competition.
This increased competition has exerted significant downward pressure on pricing for enoxaparin sodium.
How has Lovenox pricing evolved, and what are the future price projections?
Lovenox pricing has historically been high due to its proprietary status. However, the advent of generic competition has drastically altered this trajectory.
Historical Pricing Trends:
- Pre-Generic Era: Lovenox commanded premium pricing, reflecting its patent protection and market exclusivity.
- Post-Generic Entry: Prices have fallen significantly as generic manufacturers entered the market. The extent of the price reduction varies by region and the number of competing generic products.
Factors Influencing Future Pricing:
- Generic Penetration: As more generics gain market share, price competition intensifies.
- Payer Strategies: Insurance companies and government payers often favor lower-cost generics, incentivizing their use through formulary placement and rebates.
- Manufacturing Costs: The cost of raw materials and complex manufacturing processes for heparin derivatives can influence generic pricing floors.
- Therapeutic Alternatives: Development of novel oral anticoagulants (NOACs) or direct oral anticoagulants (DOACs) provides alternative treatment options, potentially impacting demand and pricing for Lovenox and its generics. While Lovenox remains a standard of care in certain indications, the broader anticoagulant market is evolving.
- Regulatory Policies: Changes in regulatory approval pathways or pricing regulations can influence market dynamics.
Price Projections:
Future price projections for enoxaparin sodium indicate continued downward pressure. While precise percentage drops are difficult to forecast due to market-specific factors, it is expected that prices will stabilize at levels significantly below the original Lovenox brand.
- Short to Medium Term (1-3 years): Expect continued price erosion as generic market share solidifies. Price reductions of 10-20% annually are plausible in competitive markets until a pricing equilibrium is reached.
- Long Term (3-5+ years): Pricing will likely be dictated by manufacturing costs and the competitive intensity among generic manufacturers. Significant price increases are not anticipated without a major shift in manufacturing technology or a substantial decline in the number of market participants.
Data from market analysis firms often projects a continued decline in the value of the enoxaparin market globally, even as the volume of units sold may remain stable or grow, illustrating the profound impact of price erosion [4].
What are the key regulatory and clinical considerations affecting Lovenox?
Regulatory approvals and clinical practice guidelines significantly shape the use and market access of Lovenox and its generics.
Regulatory Approvals:
- FDA (United States): The FDA grants approval for generic enoxaparin sodium based on rigorous bioequivalence studies demonstrating that the generic product performs comparably to the reference listed drug (Lovenox) [3].
- EMA (Europe): The European Medicines Agency follows a similar pathway, requiring comprehensive data to establish biosimilarity or generic equivalence.
Clinical Practice Guidelines:
- ACCP (American College of Chest Physicians): Guidelines from organizations like the ACCP provide recommendations for VTE prophylaxis and treatment, influencing physician prescribing habits. These guidelines often endorse Lovenox and its generics as first-line options in specific scenarios.
- AHA/ACC (American Heart Association/American College of Cardiology): Guidelines for ACS management also detail the role of enoxaparin sodium.
Factors Influencing Clinical Use:
- Cost-Effectiveness: Payers and healthcare systems increasingly prioritize cost-effective treatments, making generic enoxaparin sodium attractive.
- Ease of Administration: While injectable, the subcutaneous administration of enoxaparin is well-established and manageable in various healthcare settings.
- Established Safety and Efficacy Profile: Decades of clinical use have built a strong evidence base for Lovenox and its generics.
- Emergence of DOACs: The widespread adoption of DOACs for stroke prevention in atrial fibrillation and VTE treatment presents a significant competitive force. DOACs offer oral administration, which is generally preferred by patients and providers, and often have fixed dosing without the need for routine laboratory monitoring [5]. However, enoxaparin remains critical in specific populations, such as patients with mechanical heart valves, severe renal impairment, or during certain perioperative periods where DOACs may be contraindicated or less studied.
The ongoing interplay between regulatory approvals, evolving clinical guidelines, and the availability of alternative therapies will continue to dictate the market position and pricing of enoxaparin sodium.
What are the market risks and opportunities for Lovenox and its generics?
The Lovenox market presents both significant risks, primarily from competition and evolving treatment paradigms, and opportunities, particularly for generic manufacturers.
Market Risks:
- Intensifying Generic Competition: The presence of multiple generic manufacturers leads to aggressive pricing strategies, eroding profit margins for all players.
- Pricing Pressure from Payers: Healthcare payers are increasingly leveraging their purchasing power to negotiate lower prices for generics.
- Shifting Treatment Paradigms: The growth of DOACs and potentially other novel anticoagulants could lead to a gradual decline in the overall market volume for injectable anticoagulants like enoxaparin.
- Supply Chain Vulnerabilities: The heparin supply chain can be susceptible to disruptions, impacting the availability and cost of raw materials for enoxaparin production.
- Regulatory Hurdles for New Entrants: While patents have expired, obtaining regulatory approval for a generic enoxaparin can still be a complex and costly process, although less so than for novel drug development.
Market Opportunities:
- Cost-Effective Treatment Option: For indications where enoxaparin remains a standard of care, generic versions offer a highly cost-effective solution for healthcare systems and patients.
- Emerging Markets: Growth in healthcare infrastructure and access in developing countries can present opportunities for generic enoxaparin sales.
- Product Differentiation (Minor): While challenging for generics, some opportunities might exist in offering specific delivery devices or packaging formats that enhance patient convenience or healthcare provider efficiency.
- Manufacturing Expertise: Companies with efficient and cost-effective manufacturing processes for heparin derivatives can gain a competitive advantage.
- Stable Demand in Niche Indications: In specific patient populations (e.g., severe renal dysfunction, certain ACS subsets, perioperative management in high-risk patients), enoxaparin may retain its preferred status, ensuring a baseline demand.
The overall market trend for enoxaparin sodium is one of commoditization due to extensive genericization. Success for manufacturers will depend on efficient production, strong supply chain management, and the ability to compete on price.
Lovenox Market Data Snapshot:
| Metric | Value / Trend | Source / Note |
|---|---|---|
| Global Market Size | Approx. $2.9 billion (2022) | [2] |
| Projected CAGR | 4.1% (2023-2030) | [2] (Reflects volume growth outstripping price decline) |
| Key Competitor Type | Generic Enoxaparin Sodium Manufacturers | [3] |
| Primary Market Drivers | VTE Prophylaxis, ACS Treatment, Cost-Effectiveness | [1] |
| Primary Market Restraint | Generic Competition, DOACs | [5] |
| Pricing Trend | Declining due to generic entry | Industry Observation |
Key Takeaways
- Lovenox, as enoxaparin sodium, faces a market dominated by generic competition following patent expiries.
- The global enoxaparin market, while substantial, is characterized by significant price erosion due to the entry of multiple generic manufacturers.
- Future pricing is projected to remain under downward pressure, driven by ongoing generic competition and payer strategies.
- While direct oral anticoagulants (DOACs) are increasing their market share, enoxaparin sodium retains critical use in specific patient populations and indications.
- Opportunities for generic manufacturers lie in efficient production, cost competitiveness, and servicing emerging markets.
Frequently Asked Questions
-
What is the primary mechanism of action for Lovenox (enoxaparin sodium)? Enoxaparin sodium is a low molecular weight heparin that exerts its anticoagulant effect primarily by potentiating the activity of antithrombin III, which inhibits activated factor X (FXa) and, to a lesser extent, thrombin (factor IIa) [1].
-
How does Lovenox compare to warfarin in terms of efficacy and safety? Lovenox and its generics are generally considered to have a more predictable pharmacokinetic profile and require less routine monitoring compared to warfarin. They also have a lower risk of major bleeding and drug-drug interactions when used for their approved indications, though warfarin remains a cornerstone for long-term anticoagulation in certain conditions like mechanical heart valve replacement [5].
-
What are the typical dosages and administration routes for enoxaparin sodium? Dosage varies based on indication and patient weight. It is typically administered subcutaneously once or twice daily. For instance, prophylaxis of VTE in abdominal surgery may involve 40 mg once daily, while treatment of ACS may require higher or weight-based dosing with more frequent administration [3].
-
Are there any contraindications for using enoxaparin sodium? Absolute contraindications include active major bleeding, hypersensitivity to enoxaparin or heparin, and, in some cases, severe uncontrolled hypertension or conditions where there is a high risk of hemorrhage. Caution is advised in patients with severe renal impairment, history of heparin-induced thrombocytopenia (HIT), or recent hemorrhagic stroke [3].
-
What is the difference between a generic enoxaparin and a biosimilar enoxaparin? Enoxaparin sodium is a chemically synthesized drug, not a biologic. Therefore, its generic versions are considered direct generics, meaning they must demonstrate bioequivalence to the reference product. The term "biosimilar" is reserved for complex biologic drugs derived from living organisms, which have a more complex process of demonstrating similarity [3].
Citations
[1] Sanofi. (n.d.). Lovenox® (enoxaparin sodium) injection. Retrieved from [Source Website - e.g., Manufacturer's Prescribing Information or Official Product Website] [2] Grand View Research. (2023). Enoxaparin Sodium Market Size, Share & Trends Analysis Report By Application (DVT Prophylaxis, PE Treatment, ACS Treatment, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Others), By Region, And Segment Forecasts, 2023 - 2030. [3] U.S. Food & Drug Administration. (n.d.). Drug Database. Retrieved from [Source Website - e.g., FDA Orange Book or Approved Drug Products Database] [4] IQVIA Institute for Human Data Science. (Various Reports). Global Medicine Spending and Usage Trends. [5] American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. (Various Guidelines). Guidelines for the Management of Cardiovascular Disease.
More… ↓
