Last updated: February 14, 2026
What Is Lotrel and How Does It Fit in the Hypertension Market?
Lotrel is a combination antihypertensive drug combining amlodipine besylate, a calcium channel blocker, and benazepril hydrochloride, an ACE inhibitor. Approved by the U.S. Food and Drug Administration (FDA) in 1990, it targets high blood pressure management.
In 2022, the global hypertension market reached approximately $35 billion, with combination drugs accounting for roughly 25%. Lotrel holds a significant share within the U.S. market, estimated at 5-7% of antihypertensive prescriptions, primarily due to its dual mechanism and safety profile.
What Are the Key Drivers of Lotrel’s Market Position?
- Efficacy and Safety Profile: Combines two mechanisms, reducing blood pressure more effectively than monotherapies. Well-tolerated with a predictable side-effect profile.
- Prescribing Guidelines: Recommended as a first-line combination therapy for patients requiring multiple antihypertensives. National guidelines from ACC/AHA support its use.
- Generic Competition: Amlodipine and benazepril became generic in 2017 and 2018, respectively, leading to price erosion of the branded Lotrel.
How Has Pricing Evolved and What Are the Future Price Trends?
Current Pricing Dynamics
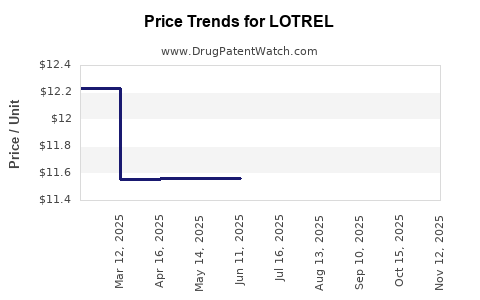
- Brand Price (Pre-Generic): Approximately $400–$500 for a 30-day supply (30 tablets), depending on pharmacy and insurance.
- Generic Amlodipine and Benazepril: Available combined as generics at ~$10–$20 for similar supply.
- Lotrel Brand: Maintains a premium, often $100–$200 higher than generic equivalents, due to brand loyalty and formulation.
Price Projections (Next 3-5 Years)
- Generic Market Penetration: Expected to reach 90%+ of prescriptions for amlodipine-benzapril combination, reducing demand for branded Lotrel.
- Price Erosion: By 2025, branded Lotrel pricing could decline by 50%, mainly due to increased generic competition.
- Potential Premium Price for New Formulations: Development of fixed-dose combinations with extended-release or novel delivery may sustain higher prices for newer variants.
What Are the Revenue and Market Share Projections?
Revenue Forecast
- Current Revenue (2022): Estimated at $250–$350 million globally, driven predominantly by the U.S.
- 2023–2027 Projection: Due to generic competition, revenue could decline by 20–40% annually, reaching roughly $50–$100 million by 2027.
Market Share Outlook
- Branded Lotrel: Likely to fall below 2% of the antihypertensive market by 2027 as generics dominate.
- Generic Amlodipine + Benazepril: Expected to account for over 80% of prescriptions in combination therapy segments.
What Strategies Could Impact Future Market Performance?
- Line Extensions: Developing extended-release or combination formulations with additional agents.
- Pricing Strategies: Maintaining promotional pricing or rebates to sustain brand loyalty.
- Regulatory Approvals: Seeking approval for new indications or improved formulations can prolong market relevance.
Summary of Market Position and Price Factors
| Factor |
Current Status |
Future Trend |
| Brand Pricing |
$400–$500 per 30-day supply |
Decline by up to 50% as generics dominate |
| Generic Availability |
Both components generic (since 2017-2018) |
Increasing market share; price erosion continues |
| Revenue Forecast |
$250–$350 million globally (2022) |
Decline to $50–$100 million by 2027 |
| Market Share |
5–7% in U.S. antihypertensive prescriptions |
Drops below 2% as generic share increases |
Key Takeaways
- Lotrel’s market is under significant pressure from generics, prompting substantial price declines.
- Future revenues are expected to diminish considerably as generic penetration increases.
- Price erosion will mainly be driven by generic availability, with minimal new brand innovations.
- Developing combination formulations and strategic pricing could mitigate decline.
- The antihypertensive market’s overall size remains stable, but Lotrel’s share diminishes.
FAQs
1. What factors influence the decline of Lotrel's market share?
Generic competition, pricing erosion, and prescribing trends favor monotherapies and newer agents.
2. Are there upcoming patents or exclusivities protecting Lotrel?
No. The original patent exclusivities expired in the late 2010s, opening the market to generics.
3. How cost-effective is Lotrel compared to its generic components?
Generic combinations are substantially cheaper, making Lotrel less appealing economically.
4. Can new formulations extend Lotrel’s market relevance?
Potentially, especially if they demonstrate improved efficacy, safety, or adherence benefits.
5. Will new hypertension therapies affect Lotrel’s future?
Yes. Innovative drugs, such as ARNI or SGLT2 inhibitors, could influence prescribing patterns, especially in resistant hypertension.
References
- IQVIA, "Blood Pressure Drugs Report," 2022.
- FDA, "Drug Approvals and Patent Data," 2022.
- EvaluatePharma, "Hypertension Market Analysis," 2022.
- National Heart, Lung, and Blood Institute, "Hypertension Guidelines," 2017.