Share This Page
Drug Price Trends for LORYNA
✉ Email this page to a colleague
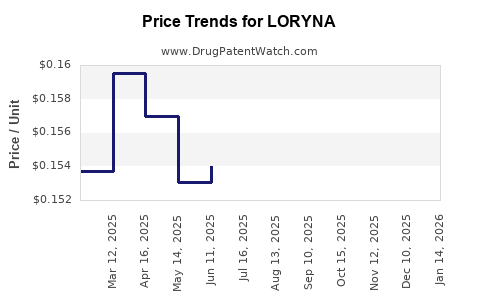
Average Pharmacy Cost for LORYNA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LORYNA 3 MG-0.02 MG TABLET | 70700-0114-85 | 0.17961 | EACH | 2026-05-20 |
| LORYNA 3 MG-0.02 MG TABLET | 70700-0114-84 | 0.17961 | EACH | 2026-05-20 |
| LORYNA 3 MG-0.02 MG TABLET | 70700-0114-85 | 0.16558 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is Loryna?
Loryna is an oral contraceptive containing 20 mcg of ethinyl estradiol and 3 mg of drospirenone. It is marketed as a hormonal birth control pill used for contraception, acne treatment, and regulation of menstrual cycles. Manufactured by Bayer, Loryna was approved by the FDA in 2014.
What Is the Market Size for Loryna?
The oral contraceptive market in the United States exceeds $7 billion annually, with popular brands such as Yaz, Yasmin, and Yasminelle accounting for significant shares. The segment driven by drospirenone-based pills is estimated to be around $2.8 billion.
Specifically, drospirenone-based formulations, including Loryna, target women aged 15–44. The user population in the U.S. is approximately 56 million women of reproductive age, with an estimated 70% using prescription contraception. Around 14 million women use drospirenone-based pills.
Estimated Market Penetration: Loryna's market share remains modest, approximately 2–3% of the drospirenone segment, translating to annual sales estimates of $60–85 million.
What Are the Key Drivers and Barriers?
Drivers:
- Growing awareness of oral contraceptives’ non-contraceptive benefits such as acne reduction.
- Increased adoption among women seeking hormonal regulation for menstrual symptoms.
- Bayer’s ongoing marketing efforts and clinician awareness.
Barriers:
- Competition from established brands like Yaz, Yasmin, and generic alternatives.
- FDA warnings concerning certain drospirenone formulations linked to thromboembolic risks.
- The shift toward non-hormonal or lighter hormonal options.
What Are the Price Dynamics?
Current Pricing:
- Loryna's wholesale acquisition cost (WAC): approximately $60–$70 per cycle (28 days).
- Cash price for consumers: ranges from $75 to $100 per cycle depending on discounts and pharmacy.
Pricing Compared to Competitors:
| Product | Typical Price per Cycle | Active Ingredients | Market Position |
|---|---|---|---|
| Loryna | $60–$70 | Ethinyl estradiol (20 mcg) + drospirenone (3 mg) | Niche-focus, branded |
| Yaz | $50–$60 | Ethinyl estradiol (20 mcg) + drospirenone (3 mg) | Leading brand |
| Yasminelle | $35–$45 | Ethinyl estradiol (30 mcg) + drospirenone (3 mg) | Generic alternative |
Price Trends:
- Loryna's price has remained relatively stable since launch.
- Price competition from generics has pressured branded drospirenone products.
- Insurance coverage reduces out-of-pocket costs, affecting retail prices.
What Are the Future Price Projections?
Market Trends:
- The U.S. oral contraceptive market is expected to grow at a compound annual growth rate (CAGR) of about 3% over the next five years, reaching nearly $8 billion by 2028.
- Price pressure from generics will intensify, reducing margins for branded drugs like Loryna.
- The average retail price for branded estrogen-progestin pills is projected to decline by 2–3% annually, driven by increased generic access.
Projected Pharmaceutical Revenue:
- Loryna's sales are, conservatively, projected to remain within the $60–$85 million annual range through 2025, assuming stable market share.
- Price per cycle may decrease by 5–10% due to generic competition and formulary negotiations.
Outlook:
- Without new indications or substantial market expansion, Loryna’s price will face downward pressure.
- Likelihood of price stability exists if Bayer can differentiate through marketing or exclusive formulations.
How Do Regulatory and Policy Factors Affect Pricing?
- FDA warnings and increased scrutiny of drospirenone formulations may impact prescribing patterns and pricing.
- Healthcare reforms and insurance coverage policies influence retail prices.
- Patent protections for drospirenone formulations expired or will expire, increasing generic competition.
Key Takeaways
- Loryna's U.S. market is part of a $2.8 billion segment, with a modest market share.
- Current prices hover around $60–$70 per cycle, with competitive pressures expected to lower prices.
- Future revenue depends on maintaining market share amidst increasing generic options and regulatory shifts.
- The overall market is projected to grow modestly, but branded contraceptive pricing faces ongoing downward pressure.
5 FAQs
1. Will Loryna gain market share against generics?
Likely small gains if Bayer maintains marketing efforts; however, generic competition limits significant growth prospects.
2. How will regulatory issues impact Loryna?
FDA warnings may restrict prescribing, reducing sales volume unless Bayer manages to distinguish Loryna through safety data or new indications.
3. What are the main price factors for Loryna?
Pricing is influenced by manufacturing costs, competition, insurer negotiations, and patient out-of-pocket expenses.
4. Could Loryna's price increase due to new indications?
Potentially, if Bayer secures approvals for additional uses, which could justify higher pricing and expanded market share.
5. How does insurance coverage affect retail prices?
Insurance reduces out-of-pocket costs, making actual consumer prices lower and limiting price escalation potential.
References
[1] IQVIA, "U.S. Prescription Market Reports," 2022.
[2] Bayer, "Loryna Product Information," 2014.
[3] Statista, "Oral Contraceptive Market in the U.S.," 2022.
[4] FDA, "Drug Safety Communications," 2019.
[5] IMS Health, "Prescription Drug Market Trends," 2022.
More… ↓
