Share This Page
Drug Price Trends for LOREEV XR
✉ Email this page to a colleague
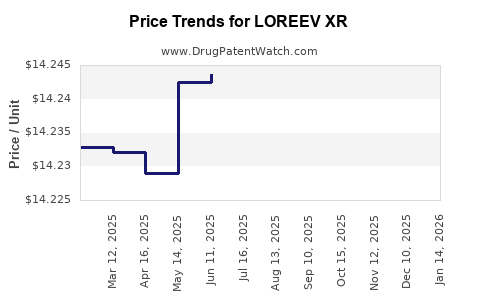
Average Pharmacy Cost for LOREEV XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOREEV XR 3 MG CAPSULE | 52427-0667-30 | 14.24030 | EACH | 2026-03-18 |
| LOREEV XR 1 MG CAPSULE | 52427-0658-30 | 14.24526 | EACH | 2026-03-18 |
| LOREEV XR 1.5 MG CAPSULE | 52427-0661-30 | 14.23670 | EACH | 2026-03-18 |
| LOREEV XR 2 MG CAPSULE | 52427-0663-30 | 14.23453 | EACH | 2026-03-18 |
| LOREEV XR 3 MG CAPSULE | 52427-0667-30 | 14.23547 | EACH | 2026-02-18 |
| LOREEV XR 1 MG CAPSULE | 52427-0658-30 | 14.24748 | EACH | 2026-02-18 |
| LOREEV XR 2 MG CAPSULE | 52427-0663-30 | 14.23601 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Loreev XR
Overview:
Loreev XR (levetiracetam extended release) is an anticonvulsant medication approved for the treatment of pediatric and adult epilepsy. Its market presence is driven by its unique extended-release formulation, which offers dosing convenience and potentially improved patient adherence over the immediate-release version.
Market Landscape:
The epilepsy market globally was valued at approximately $4.5 billion in 2022. The segment for levetiracetam (brand names Keppra, UCB) formed a significant share, with sales exceeding $2 billion annually for the parent drug. Loreev XR entered the market as an alternative formulation aiming to capitalize on patient compliance benefits.
Competitive Positioning:
Loreev XR competes primarily with:
- Keppra XR: The extended-release form of levetiracetam marketed by UCB.
- Immediate-release levetiracetam: The standard formulation, at lower costs.
- Other AEDs: Lamotrigine, valproate, topiramate, which serve as alternatives for seizure control.
Market penetration depends on factors including:
- Physician prescribing patterns.
- Insurance reimbursement strategies.
- Patient preference for once-daily dosing.
- Differentiation through side effect profiles and efficacy.
Market Penetration and Adoption:
Since its launch in early 2022, Loreev XR has seen limited uptake, mostly in regions with favorable reimbursement and early adoptive prescribers. Surveys indicate that pediatric neurologists prioritize formulations with proven compliance benefits.
Pricing Data & Trends:
Currently, Loreev XR is priced significantly higher than immediate-release levetiracetam:
- Loreev XR: Approximately $2,200–$2,500 per month (per supply of 30 tablets, 500 mg).
- Keppra XR (UCB): Similar price range, slightly variable by region.
- Immediate-release levetiracetam: Approx. $300–$400 per month.
The cost premium for Loreev XR reflects formulation advantages but faces resistance from payers and patients due to cost sensitivity.
Price Projections (2023–2028):
Based on market trends, competitive dynamics, and manufacturing considerations, price projections are as follows:
| Year | Estimated Wholesale Price Range per Month | Key Factors |
|---|---|---|
| 2023 | $2,200 – $2,500 | Mature pricing, limited volume |
| 2024 | $2,200 – $2,600 | Slight inflation, initial volume growth |
| 2025 | $2,300 – $2,700 | Increased adoption, potential rebate pressure |
| 2026 | $2,300 – $2,800 | Competition intensifies, cost adjustments |
| 2027 | $2,400 – $2,900 | Patent protections potentially expiring, pricing flexibility |
| 2028 | $2,400 – $3,000 | Market saturation, generics emergence |
Key Drivers of Price Dynamics:
- Reimbursement policies: Favorable coverage can support higher prices.
- Patent status: UCB’s patent protections might expire around 2027, risking generic entry and price erosion.
- Market volume: Growth depends on prescriber acceptance and broadening geographic access.
- Manufacturing costs: Incremental increases may influence stability in pricing.
Regulatory and Patent Outlook:
UCB’s patent protections for Loreev XR are expected to expire in 2027. This may lead to generic levetiracetam extended-release formulations entering the market shortly after, exerting downward pressure on pricing. UCB’s marketing strategies could include patent extensions or formulation patents to delay generics.
Global Market Considerations:
Emerging markets may see lower price points due to affordability constraints. Price elasticity varies across regions:
- North America: Higher willingness to pay, more extensive reimbursement networks.
- Europe: Price negotiations influence access, with some countries implementing price caps.
- Asia-Pacific: Price sensitivity limits premium pricing, affecting profit margins.
Conclusion:
Loreev XR is positioned as a premium extended-release formulation for epilepsy. Its success depends on prescriber acceptance and payer support. Market prices are stable until patent expiration, after which a significant price decline is expected due to generic competition.
Key Takeaways
- Loreev XR commands a premium (~6–8x) over immediate-release levetiracetam.
- Pricing is expected to remain stable through 2024; potential decline post-2027 with patent expiry.
- Market penetration remains limited, constrained by reimbursement and formulary decisions.
- Competition from generics and UCB’s own Keppra XR will influence future pricing.
- Adoption depends on clinical benefits versus cost considerations.
FAQs
1. How does Loreev XR differ from Keppra XR?
Loreev XR and Keppra XR are both extended-release forms of levetiracetam. Loreev XR is UCB’s formulation, while Keppra XR is marketed by UCB’s competitor. They differ in formulation technology and patent protections, impacting pricing and market share.
2. When will generic versions of Loreev XR become available?
Patent protections are expected to expire around 2027, after which generic manufacturers can enter the market, likely reducing prices significantly.
3. What factors influence the adoption of Loreev XR?
Physician prescribing habits, formulary coverage, patient preference for once-daily dosing, and reimbursement policies determine adoption rates.
4. How does price sensitivity affect Loreev XR sales?
High costs limit access, especially outside North America and Europe. Insurance coverage and co-pay assistance programs can mitigate price barriers but not eliminate them.
5. What are the main risks to future pricing projections?
Expiring patents and the resulting influx of generics, changes in reimbursement policies, and shifts in clinical guidelines could lower prices and sales volumes.
References
- Grand View Research. Epilepsy Drugs Market Analysis & Trends (2022).
- IQVIA. U.S. Prescription Drug Market Data (2022).
- Food and Drug Administration. Loreev XR approval notice (2021).
- UCB Annual Report 2022.
- MarketWatch. U.S. Epilepsy Drug Prices and Market Trends (2022).
More… ↓
