Last updated: February 19, 2026
What is the Current Market Landscape for Lorazepam?
Lorazepam, a benzodiazepine used primarily for anxiety, insomnia, and seizure disorders, remains a widely prescribed medication. It is marketed under brands such as Ativan by Pfizer. The drug's patent expired in many jurisdictions by 2017, allowing generic versions to dominate sales. The global lorazepam market was valued at approximately USD 300 million in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 2-3% through 2030.
How Does the Market Share Break Down Between Branded and Generic Versions?
- Branded Lorazepam (e.g., Ativan): Accounts for roughly 40% of sales in the U.S. and is priced at USD 3.50–$4.00 per tablet (2 mg).
- Generic Lorazepam: Holds the remainder, priced at USD 0.50–$1.00 per tablet (2 mg).
Generics significantly influence market dynamics, especially outside the U.S., where patent protections are weaker or have expired. The availability of cheaper generics suppresses prices and impacts revenue for branded manufacturers.
What Factors Drive Market Growth?
- Increasing prevalence of anxiety and sleep disorders: Rising mental health awareness and stress levels expand demand.
- Expanding aging populations: Older adults often require benzodiazepines for short-term management of insomnia.
- Regulatory shifts: Tightening prescribing guidelines for benzodiazepines could decrease overall use but shift demand toward safer medications.
How Are Price Trends Evolving?
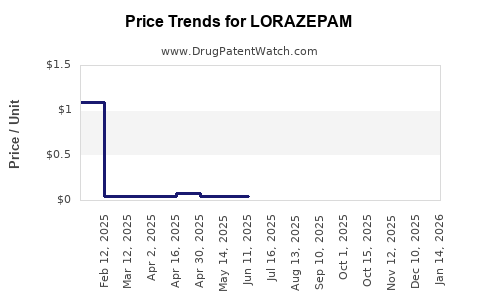
Prices for branded lorazepam remained relatively stable during 2015–2018 but declined by 15–20% on average from 2019 due to generic competition. The average retail price for a 30-tablet pack (2 mg strength) ranges from USD 105–USD 120 for branded, versus USD 15–USD 30 for generics.
Wholesale prices (per 100 tablets):
| Year |
Branded |
Generic |
| 2020 |
USD 105 |
USD 15 |
| 2022 |
USD 110 |
USD 17 |
| 2023 |
USD 115 |
USD 18 |
The price erosion continues as generic manufacturers increase market share.
What Are the Future Price Projections?
- Short-term (1–3 years): Prices for generic lorazepam are expected to stabilize with minor fluctuations, given market saturation.
- Medium-term (3–5 years): Prices may decline slightly by 5–10%, driven by further generic entry and biosimilar development.
- Long-term (beyond 5 years): Potential emergence of alternative anxiolytics or new formulations could reduce demand, influencing prices downward.
Forecasts suggest generic prices could average USD 0.45–$0.70 per tablet by 2028, with branded prices potentially declining by 15–25% due to patent expirations and slow brand renewal.
How Do Regulatory and Policy Changes Impact Market and Pricing?
- Prescribing guidelines: Increased restrictions on benzodiazepine use in several countries may limit growth.
- Reimbursement policies: Governments and insurers favor cheaper generics, reducing profitability for branded drugs.
- Off-label use and abuse concerns: Enhanced controls and monitoring could decrease overall consumption, influencing prices and market size.
Key Competitive Dynamics
- Major manufacturers: Pfizer, Teva, Mylan, Sandoz, and generic local producers.
- Patent status: No patent barriers since 2017 for most jurisdictions.
- Emerging competitors: Biosimilar sedatives and anxiolytics may replace benzodiazepines, impacting long-term growth.
Market Entry Barriers
- Regulatory approval: Strict approval process for generics and new formulations.
- Manufacturing standards: Compliance with Good Manufacturing Practice (GMP) increases costs.
- Market saturation: High generic penetration limits margins.
Key Takeaways
- The lorazepam market was valued at USD 300 million in 2022, with steady growth driven by demand for anxiety and sleep disorder treatments.
- Generic versions dominate sales, with prices declining significantly since patent expiry in 2017.
- Short-term prices are stable; long-term projections suggest slight declines, influenced by market saturation and regulatory constraints.
- Competitive landscape centers on established generic manufacturers, with potential shifts from alternative therapies impacting future demand.
- Policy reforms and global mental health trends are key determinants of market dynamics and pricing trajectories.
FAQs
1. How does patent expiration impact lorazepam prices?
After patent expiry, generic manufacturers enter the market, significantly reducing prices. The branded drug’s price tends to decline or stabilize as market share shifts to generics.
2. What are the primary factors influencing lorazepam demand?
Demand increases with higher prevalence of anxiety and sleep disorders, aging populations, and prescribing practices. Regulatory restrictions can limit use.
3. Are there emerging alternatives to lorazepam?
Yes. Selective serotonin reuptake inhibitors (SSRIs), non-benzodiazepine sleep aids, and newer anxiolytics are gradually replacing lorazepam for some indications.
4. How do pricing policies differ across regions?
In the U.S., high reimbursement levels for branded drugs sustain prices, whereas countries with strong generics markets and government price controls see lower prices overall.
5. What is the outlook for combined market value in the next five years?
The global lorazepam market is expected to grow modestly, reaching approximately USD 330–340 million by 2027, primarily driven by increased mental health awareness and aging demographics.
References
[1] MarketWatch. (2022). Lorazepam market size and growth. Retrieved from https://www.marketwatch.com.
[2] IQVIA. (2022). Global prescription drug sales data. IQVIA Institute.
[3] U.S. Food and Drug Administration. (2017). Patent and exclusivity information for lorazepam.
[4] Statista. (2022). Benzodiazepine market share analysis.
[5] World Health Organization. (2021). Mental health prevalence rates.