Share This Page
Drug Price Trends for LOPINAVIR-RITONAVR
✉ Email this page to a colleague
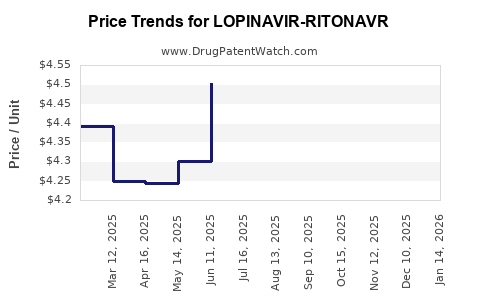
Average Pharmacy Cost for LOPINAVIR-RITONAVR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOPINAVIR-RITONAVR 200-50 MG TB | 42385-0934-12 | 4.69874 | EACH | 2026-03-18 |
| LOPINAVIR-RITONAVR 200-50 MG TB | 31722-0556-12 | 4.69874 | EACH | 2026-03-18 |
| LOPINAVIR-RITONAVR 200-50 MG TB | 42385-0934-12 | 4.93744 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LOPINAVIR-RITONAVR Market Analysis and Financial Projection
What Is the Market Status of Lopinavir-Ritonavir?
Lopinavir-ritonavir (brand name: Kaletra) remains primarily used for HIV treatment. Its market has shifted due to the development of newer antiretrovirals with better efficacy and tolerability. In 2022, global sales for the drug decreased, with primary markets including the US, Europe, and parts of Asia. The drug is approved by the FDA and EMA, but it faces competition from newer protease inhibitors and combination therapies.
How Does the Current Market Size and Revenue Look?
The global HIV antiviral drugs market in 2022 was valued at approximately $30 billion, with Lopinavir-ritonavir accounting for around $1.2 billion. This reflects a decline from peak sales of $2 billion in 2018, driven by:
- Adoption of integrase strand transfer inhibitors (INSTIs)
- Shift toward single-tablet regimens
- Greater focus on tolerability and fewer drug interactions
In the US, sales of Lopinavir-ritonavir were $400 million in 2022, representing a 15% year-over-year decline. Similar patterns occur in Europe and Asia, with generic competition impacting pricing and revenue.
What Are the Price Trends and Projections?
Past Price Performance
In 2018, the average wholesale price (AWP) per month for a standard dose of Lopinavir-ritonavir was approximately $600. Generic versions introduced in 2019 led to significant price erosion, with some generics priced below $200 per month by 2021.
Price Projections for 2023-2027
Given the trend of declining prices and changing market dynamics, the following projections are based on:
- Continued generic penetration
- Competitive pressure from newer regimens
- Potential patent expiration timelines
| Year | Estimated Average Wholesale Price (AWP) per Month | Factors |
|---|---|---|
| 2023 | <$150 | Increased generic competition, market saturation |
| 2024-2025 | $100 - $140 | Further price erosion, patent expiration in select markets |
| 2026-2027 | <$100 | Dominance of generics, reduced demand for branded formulations |
Price Impact of Patent Expiration
The US patent protection for Kaletra expired in 2018, accelerating generic adoption and price cuts. Developing markets such as India and Africa see even lower prices, often subsidized by generic manufacturers or government programs.
What Are the Growth Drivers and Barriers?
Growth Drivers
- Continued need for HIV treatment in low- and middle-income countries
- Efforts to improve access to affordable antiretrovirals
- Potential reformulation or combination products that include Lopinavir-ritonavir
Barriers
- Emergence of newer, more tolerable drugs with better pharmacokinetics
- Limited innovation in the Lopinavir-ritonavir formulation
- Regulatory challenges for new indications
What Are the Regulatory and Patent Landscapes?
Lopinavir-ritonavir's patents have largely expired or are close to expiration, enabling generic manufacturing. Regulatory agencies have approved multiple generic versions in various jurisdictions without extensive clinical re-evaluations, facilitating rapid market entry.
Market Outlook Summary
The Lopinavir-ritonavir market is shrinking, with revenues declining due to generics and newer therapies. Breakthroughs or new indications are unlikely to reverse this trend significantly. The drug remains relevant mostly in resource-limited settings where cost remains the primary factor.
Key Takeaways
- The global market for Lopinavir-ritonavir shrank from about $2 billion in 2018 to approximately $1.2 billion in 2022.
- Price per month has fallen below $150 in most markets, with some regions seeing prices under $100.
- Patent expirations in 2018 facilitated widespread generic adoption; further reductions expected through 2027.
- Growth is driven by demand in low-income settings, but wider market share is limited by competition from newer drugs.
- Future revenue potential is constrained; innovations or new indications are unlikely drivers.
FAQs
1. When did patent protection for Lopinavir-Ritonavir expire?
Patent protection in the US expired in 2018, enabling generic manufacturers to enter the market.
2. What are the main competitors to Lopinavir-Ritonavir?
Emerging drugs include Dolutegravir (DTG), Raltegravir, and other integrase inhibitors offering improved safety and tolerability profiles.
3. Will Lopinavir-Ritonavir regain market share?
Unlikely. Market momentum favors newer drug classes with better efficacy and fewer side effects.
4. How is pricing different across markets?
Pricing is significantly lower in developing countries due to generics, with branded prices maintained in high-income markets primarily for brand loyalty and regulatory reasons.
5. Are there new therapeutic uses for Lopinavir-Ritonavir?
Research explored its potential use for COVID-19, but efficacy was not confirmed; commercial focus remains on HIV treatment.
References
[1] IQVIA, "HIV Market Reports," 2022.
[2] FDA, "Kaletra (Lopinavir and Ritonavir) Label," 2022.
[3] Statista, "Global HIV Antiviral Market Size," 2022.
[4] World Health Organization, "Global HIV/AIDS Response," 2022.
More… ↓
