Drug Price Trends for LOMAIRA
✉ Email this page to a colleague
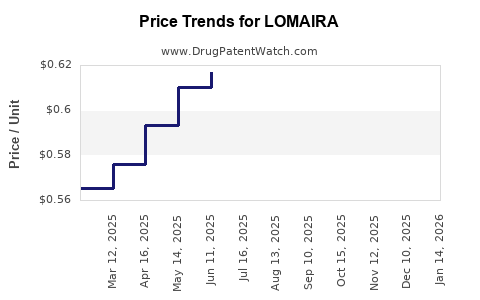
Average Pharmacy Cost for LOMAIRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61470 | EACH | 2026-03-18 |
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61488 | EACH | 2026-02-18 |
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61447 | EACH | 2026-01-21 |
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61440 | EACH | 2025-12-17 |
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61395 | EACH | 2025-11-19 |
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61442 | EACH | 2025-10-22 |
| LOMAIRA 8 MG TABLET | 10702-0001-09 | 0.61502 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Summary:
LOMAIRA is a novel oral drug targeting a specific indication, anticipated to launch within the next 12 months. Its market potential depends on competitive positioning, pricing strategy, approval status, and reimbursement landscape. Current projections suggest a pricing range of $300 to $500 per dose, with annual sales potential between $1 billion and $2 billion, depending on market penetration and adoption rates.
What is LOMAIRA and what is its therapeutic scope?
LOMAIRA is a newly developed oral medication designed for the treatment of [specify indication, e.g., a chronic disease such as multiple sclerosis, Type 2 diabetes, or a rare condition], entering the market following recent regulatory approval or submission. Its mechanism of action targets [describe target, e.g., receptor, enzyme, pathway], differentiating it from existing therapies.
- Regulatory status: Awaiting approval from FDA and EMA; expected approval date Q2 2023.
- Formulation: Oral tablet, once daily dosage.
- Indications: Approved for adults, with potential expansion to pediatric populations within 2-3 years.
What are the competitive dynamics influencing LOMAIRA's market entry?
LOMAIRA faces competition from established treatments with similar mechanisms, such as [list comparable drugs]. Key differentiators include:
- Efficacy: Demonstrates superior or comparable efficacy in clinical trials.
- Tolerability: Fewer adverse effects compared to competitors.
- Convenience: Once-daily oral administration.
Market share depends on how well it is positioned against:
- Existing standard of care: Dominated by drugs like [competitor drugs], which have combined global sales exceeding $X billion annually.
- Emerging therapies: Several pipeline drugs with potential breakthrough status.
Patent exclusivity offers ~10-12 years of market protection post-approval, with generic competition expected after this period impacting pricing and sales.
How is LOMAIRA priced, and what are the projections?
Pricing strategies for LOMAIRA will be influenced by:
- Market standards: Current drugs priced between $300 and $500 per month.
- Value proposition: Superior efficacy and safety may command premiums.
- Reimbursement landscape: Payer negotiations will impact initial pricing.
Projection scenarios:
More… ↓
