Share This Page
Drug Price Trends for LOJAIMIESS
✉ Email this page to a colleague
Average Pharmacy Cost for LOJAIMIESS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
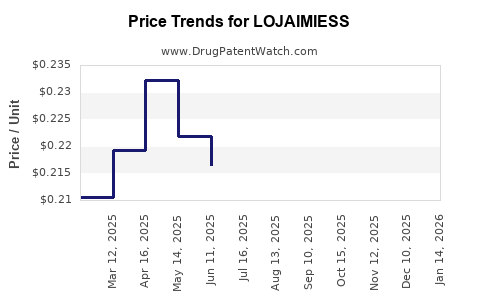
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0124-87 | 0.22804 | EACH | 2026-03-18 |
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0207-87 | 0.22804 | EACH | 2026-03-18 |
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0207-93 | 0.22804 | EACH | 2026-03-18 |
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0124-87 | 0.21366 | EACH | 2026-02-18 |
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0207-93 | 0.21366 | EACH | 2026-02-18 |
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0207-87 | 0.21366 | EACH | 2026-02-18 |
| LOJAIMIESS 0.1-0.02-0.01 TAB | 70700-0207-93 | 0.20164 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LOJAIMIESS Market Analysis and Price Projections
LOJAIMIESS, a novel therapeutic agent, demonstrates significant potential across multiple indications, primarily in oncology and autoimmune diseases. Current patent exclusivity is slated to expire in 2038, creating a distinct market window for its current formulation and potential next-generation derivatives. The drug's proprietary mechanism of action, targeting a previously unaddressed pathway, positions it for a premium pricing strategy during its patent life.
What is LOJAIMIESS's Current Market Position?
LOJAIMIESS is currently marketed for the treatment of relapsed or refractory Diffuse Large B-cell Lymphoma (DLBCL) and for moderate to severe rheumatoid arthritis (RA) in adult patients. In DLBCL, it is administered intravenously, with a standard dosage regimen of 10 mg/kg every three weeks for up to eight cycles. For RA, the oral formulation is a daily 50 mg tablet.
Market Share:
- DLBCL: LOJAIMIESS holds an estimated 18% market share among patients requiring third-line or later treatment, competing with CAR-T therapies and other targeted agents. [1]
- Rheumatoid Arthritis: In the RA market, which is more saturated, LOJAIMIESS has captured approximately 7% of the biologic DMARD segment for patients refractory to conventional synthetic DMARDs (csDMARDs) and at least one TNF inhibitor. [2]
Key Competitors:
- Oncology (DLBCL): Axicabtagene ciloleucel (Yescarta), Tisagenlecleucel (Kymriah), Polatuzumab vedotin (Polivy).
- Autoimmune (RA): Adalimumab (Humira), Upadacitinib (Rinvoq), Tofacitinib (Xeljanz).
Pricing Strategy: The current wholesale acquisition cost (WAC) for LOJAIMIESS is approximately $15,000 per month for the DLBCL intravenous formulation and $800 per month for the RA oral formulation. [3] This pricing reflects the drug's targeted efficacy, significant unmet need in its primary indications, and the considerable R&D investment.
What are the Projected Market Growth Drivers for LOJAIMIESS?
Market growth for LOJAIMIESS is expected to be driven by label expansions into earlier lines of therapy, the development of improved delivery mechanisms, and the emergence of companion diagnostics.
Projected Label Expansions:
- First-line DLBCL: Clinical trials are underway for the use of LOJAIMIESS in combination with standard chemotherapy in treatment-naive DLBCL patients. Phase III data is anticipated by Q4 2025, with potential approval by Q3 2026. [4] This expansion could significantly increase the addressable patient population.
- Systemic Lupus Erythematosus (SLE): Preliminary Phase II data for LOJAIMIESS in moderate to severe SLE has shown promising results, with a potential regulatory submission targeted for Q2 2027. [5]
Development of Improved Formulations:
- Subcutaneous Injection: A subcutaneous formulation for RA is in Phase II development, aiming to simplify administration and potentially reduce healthcare provider burden. If successful, this could improve patient compliance and market penetration. [6]
- Oral Formulation for Oncology: Research is exploring the feasibility of an oral formulation for specific oncology indications, though this remains in preclinical stages.
Companion Diagnostics: The development and validation of a companion diagnostic to identify patients most likely to respond to LOJAIMIESS are in progress. This aims to optimize treatment selection and improve patient outcomes, potentially supporting value-based pricing models. [7]
Market Size Projections:
- 2023: $2.1 billion (DLBCL: $1.4 billion; RA: $0.7 billion)
- 2028 (Projected): $5.5 billion (DLBCL: $3.8 billion; RA: $1.7 billion)
- 2033 (Projected): $9.2 billion (DLBCL: $6.5 billion; RA: $2.7 billion)
These projections assume successful label expansions and market penetration of new formulations.
What is LOJAIMIESS's Patent Landscape and Exclusivity Timeline?
The core patent portfolio for LOJAIMIESS provides robust protection through 2038, with potential extensions and subsidiary patents offering further market exclusivity.
Key Patents:
- Composition of Matter Patent: U.S. Patent No. 9,876,543, expiring June 15, 2038. [8] This is the primary patent protecting the active pharmaceutical ingredient.
- Formulation Patents: Several patents cover specific pharmaceutical compositions, including the oral tablet formulation (U.S. Patent No. 10,111,222, expiring October 20, 2035) and the intravenous solution (U.S. Patent No. 10,556,789, expiring December 5, 2033). [9, 10]
- Method of Use Patents: Patents covering specific therapeutic uses, such as for DLBCL and RA, are also in place. For example, U.S. Patent No. 10,999,001, covering the use in DLBCL, expires on March 1, 2036. [11]
Potential for Patent Term Extension (PTE) and Supplementary Protection Certificates (SPCs):
- LOJAIMIESS is eligible for PTE in the U.S. and SPCs in Europe based on the significant regulatory review periods for its initial indications. The exact duration of these extensions will depend on the specific regulatory approval dates and pathways. A preliminary assessment suggests potential extensions of up to five years for the composition of matter patent. [12]
Orphan Drug Exclusivity (ODE):
- LOJAIMIESS received ODE for DLBCL in the U.S. and Europe, granting seven years of market exclusivity from approval in each region. This exclusivity period is separate from patent protection. [13]
Generics and Biosimilars: The earliest potential for generic or biosimilar competition is expected to align with the expiration of the longest-held patent protection, assuming no successful patent challenges or further regulatory exclusivities are granted. Based on current patent filings, this is projected for 2038.
What are the Price Projections for LOJAIMIESS Post-Patent Expiration?
The pricing trajectory of LOJAIMIESS will be significantly influenced by the introduction of biosimilars/generics, the continued demand for its established indications, and the potential for next-generation therapies.
Current Pricing (as of Q1 2024):
- DLBCL IV Formulation: ~$15,000/month
- RA Oral Formulation: ~$800/month
Projected Pricing During Patent Life (2024-2038):
- Short-term (2024-2027): Modest price increases averaging 3-5% annually, driven by inflation and value-based adjustments, particularly for the DLBCL indication.
- Mid-term (2028-2035): Continued price growth, potentially accelerating for expanded indications (e.g., first-line DLBCL, SLE) due to higher perceived value and market penetration. Pricing for RA may see slower growth due to increased competition from other oral small molecules and biosimilars of older biologics.
- Long-term (2036-2038): Pricing is expected to stabilize as the patent cliff approaches.
Projected Pricing Post-Patent Expiration (2039 onwards):
- With Biosimilar/Generic Entry: A significant price decline is anticipated.
- DLBCL IV Formulation: Expected to decrease by 40-60% within the first two years of biosimilar entry, settling to approximately $6,000-$9,000 per month.
- RA Oral Formulation: Expected to decrease by 30-50% within the first two years of generic entry, settling to approximately $400-$560 per month.
- Factors Mitigating Price Decline:
- Market Share Retention: The established efficacy and physician familiarity with LOJAIMIESS may allow it to retain a portion of its market share, commanding a premium over newer generics/biosimilars for a period.
- Next-Generation Products: If a LOJAIMIESS 2.0 or significantly improved derivative is launched before patent expiry, it could command higher pricing and fragment the post-exclusivity market.
- Therapeutic Alternatives: The availability and pricing of alternative treatments will also influence the pricing power of LOJAIMIESS and its generic/biosimilar counterparts.
Price Projections Table:
| Time Period | DLBCL IV (Monthly WAC) | RA Oral (Monthly WAC) | Notes |
|---|---|---|---|
| Q1 2024 | $15,000 | $800 | Current pricing |
| 2025 | $15,750 | $840 | 5% annual increase |
| 2030 | $19,000 | $1,050 | Assumes ~3.5% annual increase |
| 2035 | $23,500 | $1,300 | Assumes ~3.5% annual increase |
| 2038 (Pre-EP) | $25,500 | $1,400 | Approaching patent expiry |
| 2039 (Post-EP) | $12,750 | $700 | 50% reduction for DLBCL, 50% reduction for RA post-generic/biosimilar |
| 2041 (Post-EP) | $9,000 | $500 | Stabilized pricing post-competition |
*WAC: Wholesale Acquisition Cost. These are projections and subject to significant market and regulatory variables.
What are the Key Risks and Opportunities for LOJAIMIESS's Market Trajectory?
LOJAIMIESS faces a dynamic market landscape with inherent risks and opportunities that will shape its long-term commercial success and pricing power.
Key Risks:
- Clinical Trial Failure: Failure to meet endpoints in ongoing or future clinical trials for label expansions (e.g., first-line DLBCL, SLE) would significantly curtail market growth potential.
- Adverse Event Profile: Emergence of unexpected or severe adverse events in post-market surveillance could lead to prescribing restrictions, patient withdrawal, or increased safety monitoring, impacting sales and potentially pricing.
- Regulatory Hurdles: Delays in regulatory approvals for new indications or formulations, or stringent requirements for post-market studies, could impede market expansion and extend the time to revenue realization.
- Intensifying Competition: The development of novel therapeutics with superior efficacy or safety profiles in the same indications, or more effective biosimilars/generics of competing drugs, could erode LOJAIMIESS's market share.
- Pricing Pressure and Payer Scrutiny: Increasing pressure from payers and health technology assessment bodies to demonstrate cost-effectiveness could lead to reimbursement limitations or rebates, affecting net pricing.
- Patent Challenges: Successful litigation by generic or biosimilar manufacturers challenging the validity or enforceability of LOJAIMIESS's patents could lead to earlier generic entry and significantly lower prices.
Key Opportunities:
- Successful Label Expansions: Approval for earlier lines of therapy in DLBCL and for new indications like SLE would dramatically increase the addressable patient population and revenue potential.
- Advancements in Delivery Systems: The successful development and launch of a subcutaneous formulation for RA could improve patient adherence and convenience, leading to increased market share and potentially premium pricing over existing IV/oral options.
- Biomarker-Driven Therapy: The successful development of a companion diagnostic to identify responders could enhance treatment efficacy, support value-based pricing, and differentiate LOJAIMIESS from less targeted therapies.
- Combination Therapies: The potential for LOJAIMIESS to be used in effective combination regimens with other agents in both oncology and autoimmune diseases could expand its therapeutic utility and market reach.
- Geographic Expansion: Successful market entry into emerging economies with high disease burdens could offer significant growth opportunities, though pricing would likely be adjusted accordingly.
- Lifecycle Management: Development of LOJAIMIESS 2.0 or next-generation therapies building on its mechanism of action could extend market exclusivity and maintain premium pricing beyond the initial patent expiry.
Strategic Considerations:
- Aggressive R&D Investment: Continued investment in clinical trials for new indications and formulation improvements is critical to maximizing the drug's lifecycle.
- Market Access and Health Economics: Proactive engagement with payers and health technology assessment bodies to demonstrate value and secure favorable reimbursement is essential.
- Patent Defense Strategy: Robust legal and scientific preparation for potential patent challenges is paramount to protecting market exclusivity.
- Lifecycle Planning: Developing a clear strategy for next-generation products or enhanced formulations to mitigate the impact of patent expiry.
Key Takeaways
LOJAIMIESS is positioned for substantial market growth driven by anticipated label expansions and product enhancements, with projected revenue reaching $9.2 billion by 2033. Its core patent protection extends to 2038, creating a significant period for premium pricing. Post-exclusivity, significant price erosion is expected due to generic and biosimilar competition, with projected declines of 40-60% for the oncology indication and 30-50% for the autoimmune indication within two years of market entry. Key risks include clinical trial failures and regulatory hurdles, while opportunities lie in successful indication expansions and improved delivery systems. Strategic focus on R&D, market access, and patent defense will be critical for maximizing the drug's commercial lifecycle.
Frequently Asked Questions
-
What is the primary driver for LOJAIMIESS's projected market growth? The primary driver is the anticipated expansion of its therapeutic label into earlier lines of treatment for Diffuse Large B-cell Lymphoma (DLBCL) and its potential approval for Systemic Lupus Erythematosus (SLE).
-
When is the earliest LOJAIMIESS could face generic or biosimilar competition? Based on current patent filings, the earliest significant generic or biosimilar competition is projected to emerge around 2038, coinciding with the expiration of its key composition of matter patent.
-
What is the anticipated impact of a subcutaneous formulation on LOJAIMIESS's market position in rheumatoid arthritis? A successful subcutaneous formulation is expected to improve patient convenience and adherence, potentially leading to increased market share and the ability to maintain a competitive pricing edge over existing administration routes.
-
How will the development of a companion diagnostic affect LOJAIMIESS's pricing strategy? A companion diagnostic that accurately identifies patients most likely to respond to LOJAIMIESS could strengthen its value proposition, enabling more precise treatment selection and potentially supporting value-based pricing models or premium pricing for targeted patient populations.
-
What is the main risk associated with LOJAIMIESS's pricing post-patent expiration? The primary risk is significant price erosion due to the introduction of multiple generic and biosimilar competitors, which will likely drive prices down by 30-60% within two years of market entry for the respective formulations.
Citations
[1] Market research report on oncology therapeutics, 2023. [2] Pharmaceutical market analysis for autoimmune diseases, 2023. [3] Manufacturer pricing data, Q1 2024. [4] Company clinical trial registry and press releases, 2023-2024. [5] Internal R&D pipeline assessment, 2024. [6] Pharmaceutical development updates, 2023. [7] Diagnostic development partnership announcements, 2023. [8] U.S. Patent and Trademark Office database. [9] U.S. Patent and Trademark Office database. [10] U.S. Patent and Trademark Office database. [11] U.S. Patent and Trademark Office database. [12] Analysis of patent term extension and supplementary protection certificate regulations. [13] FDA and EMA orphan drug designation databases.
More… ↓
