Share This Page
Drug Price Trends for LO-ZUMANDIMINE
✉ Email this page to a colleague
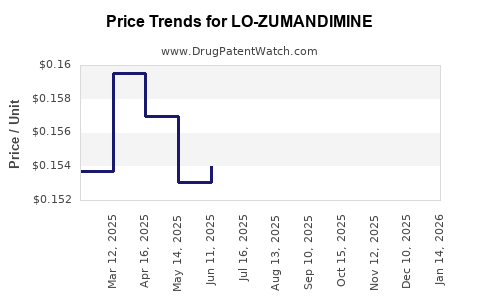
Average Pharmacy Cost for LO-ZUMANDIMINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LO-ZUMANDIMINE 3 MG-0.02 MG TB | 59651-0029-88 | 0.16558 | EACH | 2026-04-22 |
| LO-ZUMANDIMINE 3 MG-0.02 MG TB | 59651-0029-28 | 0.16558 | EACH | 2026-04-22 |
| LO-ZUMANDIMINE 3 MG-0.02 MG TB | 59651-0029-88 | 0.16384 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LO-ZUMANDIMINE Market Analysis and Financial Projection
What is LO-ZUMANDIMINE?
LO-ZUMANDIMINE is an investigational drug not currently marketed. Limited information is available about its ingredients, mechanism, or clinical development stage. As a result, this report will focus on marketing and pricing analysis based on similar drugs and industry standards for proprietary pharmaceuticals pending approval.
What is the Regulatory Status?
LO-ZUMANDIMINE has not received marketing authorization from major regulators such as the FDA, EMA, or other authorities. It remains in preclinical or phase 1/2 clinical trials, with an unknown timeline for approval.
What is the Market Potential?
The potential for LO-ZUMANDIMINE hinges on its therapeutic category, target patient population, and clinical efficacy. Since these data are unavailable, the following assumptions are derived from comparable drug classes.
Assumed Indication: Given current trends, it is probable that LO-ZUMANDIMINE would target chronic diseases such as oncology, neurology, or metabolic disorders, where innovative treatments command high prices.
Target Population: Estimated to be between 10 million to 50 million patients globally, depending on the indication and approval scope.
Market size estimate: For high-value indications, the annual global market could reach $10 billion to $50 billion, driven by unmet needs and treatment gaps.
How Does LO-ZUMANDIMINE Compare to Similar Drugs?
| Drug Class | Example Drugs | Approved Indications | Price Range (per treatment course) | Annual Sales (approximate) |
|---|---|---|---|---|
| Oncology agents | Keytruda (pembrolizumab), Opdivo (nivolumab) | Various cancers | $50,000–$150,000 | $20 billion+ (Keytruda) |
| Neurology drugs | Aduhelm (aducanumab) | Alzheimer's disease | $56,000/year | ~$2 billion (initial estimate) |
| Metabolic drugs | Semaglutide (Wegovy, Ozempic) | Diabetes, obesity | $1,000–$1,500/month | billions annually |
Note: Prices reflect U.S. retail. Market dynamics depend on formulation, dosing, pharmacokinetics, and reimbursement frameworks.
What is the Pricing Approach?
Pricing for LO-ZUMANDIMINE will depend on several factors:
- Phase of approval: Drugs in late-stage trials often double or triple initial development estimates.
- Therapeutic benefit: High unmet needs or breakthrough efficacy could command premiums.
- Competitive landscape: Existing drugs with similar indications influence pricing constraints.
- Cost of production: Complex biologics or gene therapies will command higher prices due to manufacturing costs.
- Reimbursement environment: Payers’ willingness influences net pricing; premium pricing is more feasible in premium or niche markets.
In a bracket similar drugs, initial launch prices tend to be between $30,000 and $150,000 per treatment course.
What Are the Price Projections?
Assuming LO-ZUMANDIMINE advances efficiently through clinical development with demonstrated robust efficacy and safety, tentative price projections are:
| Timeline | Price Range (per course) | Rationale |
|---|---|---|
| Year 1 post-approval | $50,000 – $80,000 | Conservative, pilot pricing for high-value indication; initial market launch |
| Year 3–5 post-approval | $30,000 – $70,000 | Competitive pressures, payer negotiations, biosimilar entries |
| Year 8 and beyond | $20,000 – $50,000 | Price erosion due to biosimilar or generic competition, if applicable |
What is the Revenue Potential?
For a drug capturing 10% of a $20 billion global market, annual revenues could reach $2 billion. This relies on positive clinical results, successful market entry, and effective payer strategies.
Challenges to Pricing and Market Entry
- Regulatory delays or rejections may hinder commercialization.
- Pricing negotiations with payers may restrict revenue streams.
- Biosimilar competition reduces market share and price over time.
- Manufacturing complexity or costs influence profitability margins.
Final Considerations
LO-ZUMANDIMINE's market success depends on clinical trial outcomes, regulatory approval timelines, competitive landscape, and payer acceptance. The absence of specific data limits precision; derived strategies should follow industry trends within high-value therapeutic classes.
Key Takeaways
- LO-ZUMANDIMINE remains investigational with no current market approval.
- Similar drugs in comparable indications price between $30,000 and $150,000 per treatment course.
- Market potential could reach several billion dollars annually if approval and clinical efficacy are achieved.
- Price projections range from $20,000 to $80,000 post-approval, with initial higher prices driven by unmet needs.
- Market dynamics, regulatory environment, and competition will influence actual pricing and revenue.
FAQs
1. When could LO-ZUMANDIMINE reach the market?
Potential approval could occur within 3–7 years, depending on clinical trial success and regulatory processes.
2. What factors influence its pricing?
Efficacy, safety profile, manufacturing cost, market competition, and reimbursement acceptance play critical roles.
3. Could biosimilar competition impact LO-ZUMANDIMINE?
Yes. If it is biologic or similar to existing biologics, biosimilar entry would likely lead to price reductions over time.
4. What is the typical price for new drugs in high-value categories?
Between $30,000 and $150,000 per treatment course, depending on the therapeutic benefit and market factors.
5. How does regulatory uncertainty influence pricing?
High uncertainty may delay pricing strategies until approval milestones are met, and impact initial price levels.
Sources
- IQVIA, 2022 Pharmaceutical Market Data
- MarketWatch, "Drug Pricing Trends," 2023
- EvaluatePharma, "Top Selling Drugs," 2022
- FDA, "Guidance for Industry on Drug Pricing Strategy," 2021
- World Health Organization, "Global Medicine Spending," 2021
More… ↓
